
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Abdulmajeed Alotaibi | + 1147 word(s) | 1147 | 2021-09-03 08:21:54 | | | |
| 2 | Lindsay Dong | Meta information modification | 1147 | 2021-09-28 04:11:27 | | |
Video Upload Options
Multiple sclerosis (MS) is characterised by widespread damage of the central nervous system that includes alterations in normal-appearing white matter (NAWM) and demyelinating white matter (WM) lesions. Neurite orientation dispersion and density imaging (NODDI) has been proposed to provide a precise characterisation of WM microstructures. NODDI maps can be calculated for the Neurite Density Index (NDI) and Orientation Dispersion Index (ODI), which estimate orientation dispersion and neurite density. Although NODDI has not been widely applied in MS, this technique is promising in investigating the complexity of MS pathology, as it is more specific than diffusion tensor imaging (DTI) in capturing microstructural alterations.
1. Introduction
2. Neurite Orientation Dispersion and Density Imaging in Multiple Sclerosis Imaging
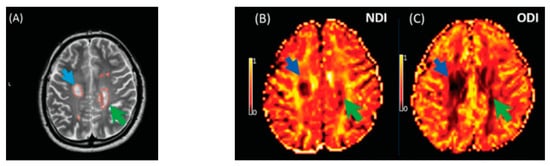
The NDI was reduced in MS lesions and NAWM compared to healthy controls (Figure 1). This significant reduction of the NDI corresponds to reduced intracellular signal fraction and may reflect underlying damage or loss of neurites. Previous work has shown that this reduction is accompanied by higher MD and lower FA [24], and the higher ODI suggests demyelination, axonal loss, and less coherence in the fibre orientation (bending or fanning axons) [23]. This meta-analysis demonstrates a significant reduction of intracellular signal fraction in the NAWM of MS subjects compared with the WM of controls, suggesting early axonal pathology outside demyelination in WM lesions [27][28][29][30]. This consistent reduction among all studies proves that the NDI (through its sensitivity to the intra-axonal compartment) may quantify and differentiate microstructural changes in WM MS lesions, in the NAWM of MS, and in healthy WM. This suggests a loss of axonal density in WM lesions and NAWM in MS compared with healthy controls, which is consistent with previous DTI studies [10][31] reporting altered water diffusion in NAWM and WM MS lesions compared with controlled subjects. Interestingly, the reduction of the NDI in WM lesions and NAWM is in line with previous pathological studies showing axonal loss within WM lesions and a lesser degree in NAWM [17][30][31][32]. This consistent reduction of the NDI reported may further assist researchers in relying on NODDI metrics in biophysically understanding the axonal loss/damage in WM lesions and NAWM.
3. Conclusions
References
- Ellwardt, E.; Zipp, F. Molecular mechanisms linking neuroinflammation and neurodegeneration in MS. Exp. Neurol. 2014, 262, 8–17.
- Mustafi, S.M.; Harezlak, J.; Kodiweera, C.; Randolph, J.S.; Ford, J.C.; Wishart, H.A.; Wu, Y.-C. Detecting white matter alterations in multiple sclerosis using advanced diffusion magnetic resonance imaging. Neural Regen. Res. 2019, 14, 114–123.
- Filippi, M.; Rocca, M.A.; Ciccarelli, O.; De Stefano, N.; Evangelou, N.; Kappos, L.; Rovira, A.; Sastre-Garriga, J.; Tintorè, M.; Frederiksen, J.L.; et al. MRI criteria for the diagnosis of multiple sclerosis: MAGNIMS consensus guidelines. Lancet Neurol. 2016, 15, 292–303.
- Wattjes, M.P.; Rovira, À.; Miller, D.; Yousry, T.A.; Sormani, M.P.; De Stefano, N.; Tintoré, M.; Auger, C.; Tur, C.; Filippi, M.; et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis-Establishing disease prognosis and monitoring patients. Nat. Rev. Neurol. 2015, 11, 597–606.
- De Groot, C.J.A.; Bergers, E.; Kamphorst, W.; Ravid, R.; Polman, C.H.; Barkhof, F.; Van Der Valk, P. Post-mortem MRI-guided sampling of multiple sclerosis brain lesions: Increased yield of active demyelinating and (p)reactive lesions. Brain 2001, 124, 1635–1645.
- Cortese, R.; Collorone, S.; Ciccarelli, O.; Toosy, A.T. Advances in brain imaging in multiple sclerosis. Ther. Adv. Neurol. Disord. 2019.
- Tur, C.; Moccia, M.; Barkhof, F.; Chataway, J.; Sastre-Garriga, J.; Thompson, A.J.; Ciccarelli, O. Assessing treatment outcomes in multiple sclerosis trials and in the clinical setting. Nat. Rev. Neurol. 2018, 14, 75–93.
- Sbardella, E.; Tona, F.; Petsas, N.; Pantano, P. DTI Measurements in Multiple Sclerosis: Evaluation of Brain Damage and Clinical Implications. Mult. Scler. Int. 2013, 2013, 1–11.
- Rovaris, M.; Iannucci, G.; Falautano, M.; Possa, F.; Martinelli, V.; Comi, G.; Filippi, M. Cognitive dysfunction in patients with mildly disabling relapsing-remitting multiple sclerosis: An exploratory study with diffusion tensor MR imaging. J. Neurol. Sci. 2002, 195, 103–109.
- Werring, D.J.; Clark, C.A.; Barker, G.J.; Thompson, A.J.; Miller, D.H. Diffusion tensor imaging of lesions and normal-appearing white matter in multiple sclerosis. Neurology 1999, 52, 1626–1632.
- Filippi, M.; Agosta, F. Diffusion tensor imaging and functional MRI. In Handbook of Clinical Neurology; Elsevier B.V: Amsterdam, The Netherlands, 2016; pp. 1065–1087.
- Ciccarelli, O.; Werring, D.J.; Barker, G.J.; Griffin, C.M.; Wheeler-Kingshott, C.A.M.; Miller, D.H.; Thompson, A.J. A study of the mechanisms of normal-appearing white matter damage in multiple sclerosis using diffusion tensor imaging: Evidence of Wallerian degeneration. J. Neurol. 2003, 250, 287–292.
- Welton, T.; Kent, D.; Constantinescu, C.S.; Auer, D.P.; Dineen, R.A. Functionally Relevant White Matter Degradation in Multiple Sclerosis: A Tract-based Spatial Meta-Analysis. Radiology 2015, 275, 89–96.
- Gallo, A.; Rovaris, M.; Riva, R.; Ghezzi, A.; Benedetti, B.; Martinelli, V.; Falini, A.; Comi, G.; Filippi, M. Diffusion-tensor magnetic resonance imaging detects normal-appearing white matter damage unrelated to short-term disease activity in patients at the earliest clinical stage of multiple sclerosis. Arch. Neurol. 2005, 62, 803–808.
- Preziosa, P.; Rocca, M.A.; Mesaros, S.; Pagani, E.; Stosic-Opincal, T.; Kacar, K.; Absinta, M.; Caputo, D.; Drulovic, J.; Comi, G.; et al. Intrinsic damage to the major white matter tracts in patients with different clinical phenotypes of multiple sclerosis: A voxelwise diffusion-tensor MR study. Radiology 2011, 260, 541–550.
- Budde, M.D.; Xie, M.; Cross, A.H.; Song, S.K. Axial diffusivity is the primary correlate of axonal injury in the experimental autoimmune encephalomyelitis spinal cord: A quantitative pixelwise analysis. J. Neurosci. 2009, 29, 2805–2813.
- Schmierer, K.; Wheeler-Kingshott, C.A.M.; Boulby, P.A.; Scaravilli, F.; Altmann, D.R.; Barker, G.J.; Tofts, P.S.; Miller, D.H. Diffusion tensor imaging of post mortem multiple sclerosis brain. Neuroimage 2007, 35, 467–477.
- Kim, J.H.; Budde, M.D.; Liang, H.F.; Klein, R.S.; Russell, J.H.; Cross, A.H.; Song, S.K. Detecting axon damage in spinal cord from a mouse model of multiple sclerosis. Neurobiol. Dis. 2006, 21, 626–632.
- Alexander, A.L.; Hasan, K.M.; Lazar, M.; Tsuruda, J.S.; Parker, D.L. Analysis of partial volume effects in diffusion-tensor MRI. Magn. Reson. Med. 2001, 45, 770–780.
- Schneider, T.; Brownlee, W.; Zhang, H.; Ciccarelli, O.; Miller, D.H.; Wheeler-Kingshott, C.G. Sensitivity of multi-shell NODDI to multiple sclerosis white matter changes: A pilot study. Funct. Neurol. 2017, 32, 97–101.
- Zhang, H.; Schneider, T.; Wheeler-Kingshott, C.A.; Alexander, D.C. NODDI: Practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 2012, 61, 1000–1016.
- Spanò, B.; Giulietti, G.; Pisani, V.; Morreale, M.; Tuzzi, E.; Nocentini, U.; Francia, A.; Caltagirone, C.; Bozzali, M.; Cercignani, M. Disruption of neurite morphology parallels MS progression. Neurol. Neuroimmunol. NeuroInflamm. 2018, 5, e502.
- Granberg, T.; Fan, Q.; Treaba, C.A.C.A.; Ouellette, R.; Herranz, E.; Mangeat, G.; Louapre, C.; Cohen-Adad, J.; Klawiter, E.C.E.C.; Sloane, J.A.J.A.; et al. In vivo characterization of cortical and white matter neuroaxonal pathology in early multiple sclerosis. Brain 2017, 140, 2912–2926.
- Hagiwara, A.; Kamagata, K.; Shimoji, K.; Yokoyama, K.; Andica, C.; Hori, M.; Fujita, S.; Maekawa, T.; Irie, R.; Akashi, T.; et al. White Matter Abnormalities in Multiple Sclerosis Evaluated by Quantitative Synthetic MRI, Diffusion Tensor Imaging, and Neurite Orientation Dispersion and Density Imaging. Am. J. Neuroradiol. 2019, 40, 1642–1648.
- De Santis, S.; Bastiani, M.; Droby, A.; Kolber, P.; Zipp, F.; Pracht, E.; Stoecker, T.; Groppa, S.; Roebroeck, A. Characterizing Microstructural Tissue Properties in Multiple Sclerosis with Diffusion MRI at 7T and 3T: The Impact of the Experimental Design. Neuroscience 2019, 403, 17–26.
- Sacco, S.; Caverzasi, E.; Papinutto, N.; Cordano, C.; Bischof, A.; Gundel, T.; Cheng, S.; Asteggiano, C.; Kirkish, G.; Mallott, J.; et al. Neurite orientation dispersion and density imaging for assessing acute inflammation and lesion evolution in MS. Am. J. Neuroradiol. 2020, 41, 2219–2226.
- Bonnier, G.; Roche, A.; Romascano, D.; Simioni, S.; Meskaldji, D.; Rotzinger, D.; Lin, Y.C.; Menegaz, G.; Schluep, M.; Du Pasquier, R.; et al. Advanced MRI unravels the nature of tissue alterations in early multiple sclerosis. Ann. Clin. Transl. Neurol. 2014, 1, 423–432.
- Filippi, M. Mri measures of neurodegeneration in multiple sclerosis: Implications for disability, disease monitoring, and treatment. J. Neurol. 2015, 262, 1–6.
- Inglese, M.; Bester, M. Diffusion imaging in multiple sclerosis: Research and clinical implications. NMR Biomed. 2010, 23, 865–872.
- Schmierer, K.; Scaravilli, F.; Altmann, D.R.; Barker, G.J.; Miller, D.H. Magnetization transfer ratio and myelin in postmortem multiple sclerosis brain. Ann. Neurol. 2004, 56, 407–415.
- Klawiter, E.C.; Schmidt, R.E.; Trinkaus, K.; Liang, H.F.; Budde, M.D.; Naismith, R.T.; Song, S.K.; Cross, A.H.; Benzinger, T.L. Radial diffusivity predicts demyelination in ex vivo multiple sclerosis spinal cords. Neuroimage 2011, 55, 1454–1460.
- Bammer, R.; Augustin, M.; Strasser-Fuchs, S.; Seifert, T.; Kapeller, P.; Stollberger, R.; Ebner, F.; Hartung, H.P.; Fazekas, F. Magnetic resonance diffusion tensor imaging for characterizing diffuse and focal white matter abnormalities in multiple sclerosis. Magn. Reson. Med. 2000, 44, 583–591.




