
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Francisco J Romero | + 1992 word(s) | 1992 | 2021-07-28 05:14:22 | | | |
| 2 | Vivi Li | Meta information modification | 1992 | 2021-09-15 04:34:19 | | | | |
| 3 | Conner Chen | Meta information modification | 1992 | 2021-09-22 02:41:53 | | |
Video Upload Options
Nitric oxide (NO) is a gas molecule with diverse physiological and cellular functions. In the eye, NO is used to maintain normal visual function as it is involved in photoreceptor light transduction. In addition, NO acts as a rapid vascular endothelial relaxant, is involved in the control of retinal blood flow under basal conditions and mediates the vasodilator responses of different substances such as acetylcholine, bradykinin, histamine, substance P or insulin. However, the retina is rich in polyunsaturated lipid membranes and is sensitive to the action of reactive oxygen and nitrogen species. Products generated from NO (i.e., dinitrogen trioxide (N2O3) and peroxynitrite) have great oxidative damaging effects. Oxygen and nitrogen species can react with biomolecules (lipids, proteins and DNA), potentially leading to cell death, and this is particularly important in the retina.
1. Introduction
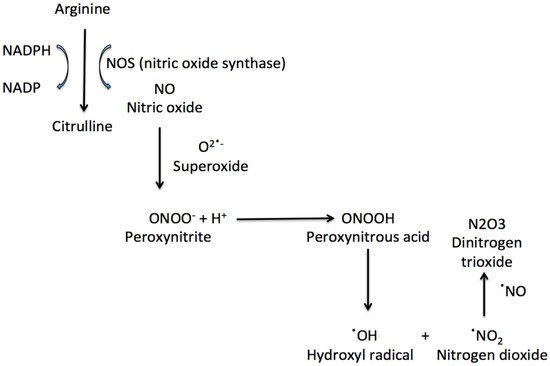
2. Nitric Oxide in the Retina
3. Diabetic Retinopathy
Can NO Inhibition Have a Role in Diabetic Retinopathy Therapy?
References
- Bredt, D.S.; Snyder, S.H. Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc. Natl. Acad. Sci. USA 1990, 87, 682–685.
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric oxide synthases: Structure, function and inhibition. Biochem. J. 2001, 357, 593–615.
- Vielma, A.H.; Retamal, M.A.; Schmachtenberg, O. Nitric oxide signaling in the retina: What have we learned in two decades? Brain Res. 2012, 1430, 112–125.
- Förstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837.
- Knowles, R.G.; Moncada, S. Nitric oxide synthases in mammals. Biochem. J. 1994, 298, 249–258.
- Goldstein, I.M.; Ostwald, P.; Roth, S. Nitric oxide: A review of its role in retinal function and disease. Vision Res. 1996, 36, 2979–2994.
- Cossenza, M.; Socodato, R.; Portugal, C.C.; Domith, I.C.; Gladulich, L.F.; Encarnação, T.G.; Calaza, K.C.; Mendonça, H.R.; Campello-Costa, P.; Paes-de-Carvalho, R. Nitric oxide in the nervous system: Biochemical, developmental, and neurobiological aspects. Vitam. Horm. 2014, 96, 79–125.
- Martínez-Ruiz, A.; Cadenas, S.; Lamas, S. Nitric oxide signaling: Classical, less classical, and nonclassical mechanisms. Free Radic. Biol. Med. 2011, 51, 17–29.
- Knott, A.B.; Bossy-Wetzel, E. Nitric oxide in health and disease of the nervous system. Antioxid. Redox Signal. 2009, 11, 541–554.
- Aslan, M.; Cort, A.; Yucel, I. Oxidative and nitrative stress markers in glaucoma. Free Radic. Biol. Med. 2008, 45, 367–376.
- McBean, G.J.; López, M.G.; Wallner, F.K. Redox-based therapeutics in neurodegenerative disease. Br. J. Pharmacol. 2017, 174, 1750–1770.
- Bradley, S.A.; Steinert, J.R. Nitric Oxide-Mediated Posttranslational Modifications: Impacts at the Synapse. Oxid. Med. Cell. Longev. 2016, 2016, 5681036.
- Lima, M.G.; Maximino, C.; Matos-Oliveira, K.R.; Brasil, A.; Crespo-Lopez, M.E.; Batista-Ede, J.; Rocha, F.A.; Picanço-Diniz, D.L.; Herculano, A.M. Nitric oxide as a regulatory molecule in the processing of the visual stimulus. Nitric Oxide 2014, 36, 44–50.
- Haverkamp, S.; Eldred, W.D. Localization of nNOS in photoreceptor, bipolar and horizontal cells in turtle and rat retinas. Neuroreport 1998, 9, 2231–2235.
- Giove, T.J.; Deshpande, M.M.; Eldred, W.D. Identification of alternate transcripts of neuronal nitric oxide synthase in the mouse retina. J. Neurosci. Res. 2009, 87, 3134–3142.
- Pang, J.J.; Gao, F.; Wu, S.M. Light responses and morphology of bNOS-immunoreactive neurons in the mouse retina. J. Comp. Neurol. 2010, 518, 2456–2474.
- Blom, J.; Giove, T.; Deshpande, M.; Eldred, W.D. Characterization of nitric oxide signaling pathways in the mouse retina. J. Comp. Neurol. 2012, 520, 4204–4217.
- Sennlaub, F.; Courtois, Y.; Goureau, O. Inducible nitric oxide synthase mediates the change from retinal to vitreal neovascularization in ischemic retinopathy. J. Clin. Investig. 2001, 107, 717–725.
- Zhang, M.; Zhou, J.; Marshall, B.; Xin, H.; Atherton, S.S. Lack of iNOS facilitates MCMV spread in the retina. Investig. Ophthalmol Vis. Sci. 2007, 48, 285–292.
- Yang, L.P.; Li, Y.; Zhu, X.A.; Tso, M.O. Minocycline delayed photoreceptor death in rds mice through iNOS-dependent mechanism. Mol. Vis. 2007, 13, 1073–1082.
- McLeod, D.S.; Baba, T.; Bhutto, I.A.; Lutty, G.A. Co-expression of endothelial and neuronal nitric oxide synthases in the developing vasculatures of the human fetal eye. Graefe’s Arch. Clin. Exp. Ophthalmol. 2012, 250, 839–848.
- Tekmen-Clark, M.; Gleason, E. Nitric oxide production and the expression of two nitric oxide synthases in the avian retina. Vis. Neurosci. 2013, 30, 91–103.
- Cheon, E.W.; Park, C.H.; Kang, S.S.; Cho, G.J.; Yoo, J.M.; Song, J.K.; Choi, W.S. Change in endothelial nitric oxide synthase in the rat retina following transient ischemia. Neuroreport 2003, 14, 329–333.
- Waldman, S.A.; Murad, F. Cyclic GMP synthesis and function. Pharmacol. Rev. 1987, 39, 163–196.
- Schmetterer, L.; Polak, K. Role of nitric oxide in the control of ocular blood flow. Prog. Retin. Eye Res. 2001, 20, 823–847.
- Stringham, J.M.; Stringham, N.T. Nitric oxide and lutein: Function, performance, and protection of neural tissue. Foods 2015, 4, 678–689.
- Mahajan, N.; Arora, P.; Sandhir, R. Perturbed biochemical pathways and associated oxidative stress lead to vascular dysfunctions in diabetic retinopathy. Oxid. Med. Cell. Longev. 2019, 2019, 8458472.
- Rossino, M.G.; Casini, G. Nutraceuticals for the treatment of diabetic retinopathy. Nutrients 2019, 11, 771.
- Li, Q.; Verma, A.; Han, P.Y.; Nakagawa, T.; Johnson, R.J.; Grant, M.B.; Campbell-Thompson, M.; Jarajapu, Y.P.; Lei, B.; Hauswirth, W.W. Diabetic eNOS-knockout mice develop accelerated retinopathy. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5240–5246.
- Narayanan, S.P.; Rojas, M.; Suwanpradid, J.; Toque, H.A.; Caldwell, R.W.; Caldwell, R.B. Arginase in retinopathy. Prog. Retin. Eye Res. 2013, 36, 260–280.
- Zheng, L.; Du, Y.; Miller, C.; Gubitosi-Klug, R.A.; Kern, T.S.; Ball, S.; Berkowitz, B.A. Critical role of inducible nitric oxide synthase in degeneration of retinal capillaries in mice with streptozotocin-induced diabetes. Diabetologia 2007, 50, 1987–1996.
- Sharma, S.; Saxena, S.; Srivastav, K.; Shukla, R.K.; Mishra, N.; Meyer, C.H.; Kruzliak, P.; Khanna, V.K. Nitric oxide and oxidative stress is associated with severity of diabetic retinopathy and retinal structural alterations. Clin. Exp. Ophthalmol. 2015, 43, 429–436.
- Kulaksızoglu, S.; Karalezli, A. Aqueous humour and serum levels of nitric oxide, malondialdehyde and total antioxidant status in patients with type 2 diabetes with proliferative diabetic retinopathy and nondiabetic senile cataracts. Can. J. Diabetes 2016, 40, 115–119.
- Abu El-Asrar, A.M.; Meersschaert, A.; Dralands, L.; Missotten, L.; Geboes, K. Inducible nitric oxide synthase and vascular endothelial growth factor are colocalized in the retinas of human subjects with diabetes. Eye (London) 2004, 18, 306–313.
- Kowluru, R.A.; Zhong, Q. Beyond AREDS: Is there a place for antioxidant therapy inthe prevention/treatment of eye disease? Investig. Ophthalmol. Vis. Sci. 2011, 52, 8665–8871.
- Kowluru, R.A.; Kanwar, M.; Chan, P.S.; Zhang, J.P. Inhibition of retinopathy and retinal metabolic abnormalities in diabetic rats with AREDS-based micronutrients. Arch. Ophthalmol. 2008, 126, 1266–1272.
- Arnal, E.; Miranda, M.; Johnsen-Soriano, S.; Alvarez-Nölting, R.; Díaz-Llopis, M.; Araiz, J.; Cervera, E.; Bosch-Morell, F.; Romero, F.J. Beneficial effect of docosahexanoic acid and lutein on retinal structural, metabolic, and functional abnormalities in diabetic rats. Curr. Eye Res. 2009, 34, 928–938.
- Wu, Y.; Tang, L.; Chen, B. Oxidative stress: Implications for the development of diabetic retinopathy and antioxidant therapeutic perspectives. Oxid. Med. Cell. Longev. 2014, 2014, 752387.
- Zheng, L.; Kern, T.S. Role of nitric oxide, superoxide, peroxynitrite and PARP in diabetic retinopathy. Front. Biosci. 2009, 14, 3974–3987.
- Carr, B.C.; Emigh, C.E.; Bennett, L.D.; Pansick, A.D.; Birch, D.G.; Nguyen, C. Towards a treatment for diabetic retinopathy: Intravitreal toxicity and preclinical safety evaluation of inducible nitric oxide synthase nhibitors. Retina 2017, 37, 22–31.
- Hernández-Ramírez, E.; Sánchez-Chávez, G.; Estrella-Salazar, L.A.; Salceda, R. Nitrosative stress in the rat retina at the onset of streptozotocin-induced diabetes. Cell. Physiol. Biochem. 2017, 42, 2353–2363.
- Othman, R.; Vaucher, E.; Couture, R. Bradykinin Type 1 Receptor–Inducible Nitric Oxide Synthase: A New Axis Implicated in Diabetic Retinopathy. Front. Pharmacol. 2019, 10, 300.
- Bailey, A.; Pope, T.W.; Moore, S.A.; Campbell, C.L. The tragedy of TRIUMPH for nitric oxide synthesis inhibition in cardiogenic shock: Where do we go from here? Am. J. Cardiovasc. Drugs 2007, 7, 337–345.




