Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Meagan McMahon | + 2946 word(s) | 2946 | 2021-08-09 10:21:43 | | | |
| 2 | Lily Guo | Meta information modification | 2946 | 2021-08-25 04:38:13 | | | | |
| 3 | Lily Guo | Meta information modification | 2946 | 2021-08-25 04:38:41 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mcmahon, M. Influenza Virus Neuraminidase and Vaccine. Encyclopedia. Available online: https://encyclopedia.pub/entry/13516 (accessed on 08 February 2026).
Mcmahon M. Influenza Virus Neuraminidase and Vaccine. Encyclopedia. Available at: https://encyclopedia.pub/entry/13516. Accessed February 08, 2026.
Mcmahon, Meagan. "Influenza Virus Neuraminidase and Vaccine" Encyclopedia, https://encyclopedia.pub/entry/13516 (accessed February 08, 2026).
Mcmahon, M. (2021, August 24). Influenza Virus Neuraminidase and Vaccine. In Encyclopedia. https://encyclopedia.pub/entry/13516
Mcmahon, Meagan. "Influenza Virus Neuraminidase and Vaccine." Encyclopedia. Web. 24 August, 2021.
Copy Citation
The influenza virus neuraminidase (NA) is primarily involved in the release of progeny viruses from infected cells—a critical role for virus replication.
neuraminidase
antibodies
1. Introduction
Vaccination remains the most effective countermeasure against influenza virus-associated morbidity and mortality [1][2][3][4]. Current seasonal influenza vaccines target the immuno-dominant surface glycoprotein, the hemagglutinin (HA) (Figure 1A) [2][5][6], as HA is responsible for viral attachment to sialic acid receptors on the host cell and fusion of viral and host endosomal membranes [6][7]. However, HA has high plasticity and changes constantly due to polymerase error rate and immune selection pressure, defined as antigenic drift [8]. As a result of this, seasonal vaccine strains must be updated annually, and, occasionally a mismatch between vaccine strains and circulating strains can result in seasonal epidemics [9][10][11]. Despite the necessity for the rapid production of seasonal influenza virus vaccines, the current process is time-consuming and expensive [12]. Hence, the investigation of new viral targets for influenza virus vaccines that are broadly protective, and do not change as frequently as HA, is warranted.
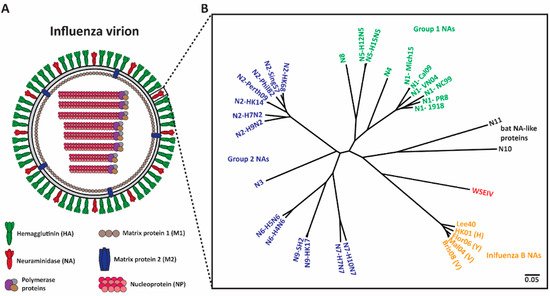
Figure 1. Phylogenetic tree of influenza NAs. (A) Depiction of an influenza virion. There are two major surface influenza glycoproteins: the hemagglutinin (HA) and neuraminidase (NA). (B) Phylogenetic tree of NA subtypes. Influenza A NAs comprise Group 1 (N1, N4, N5, and N8), Group 2 (N2, N3, N6, N7, and N9) and bat-like (N10 and N11) NAs. Influenza B NAs consist of Yamagata-like, Victoria-like and Hong Kong-like lineages. Wuhan spiny eel influenza virus (WSEIV) NA, a close relative of influenza B NAs, is also included in the phylogenetic tree. The scale bar represents a 5% change in amino acids. The phylogenetic tree was built using amino acids in Clustal Omega and then visualized in FigTree.
Neuraminidase (NA) (Figure 1A), the second surface glycoprotein of influenza virus, is a tetrameric type II transmembrane protein that plays several important roles in the viral replication cycle due to its enzymatic activity [13][14]. Initially, when an influenza virion enters a host, the virion needs to penetrate heavily glycosylated mucosal barriers [13][15][16]. These barriers act as decoy receptors for HA binding and neutralize the virion [13][17]. Here, NA assists the virion by releasing the virus particles from the decoy receptors, thus penetrating the mucus layer and gaining access to the underlying respiratory epithelium [13][15][16][17]. Upon entering and successfully replicating in the host cell, NA is crucial for viral detachment from the host cell by cleaving off sialic acid receptors that have adhered to HA [13][18][19]. Additionally, influenza virions are also known to adhere to each other via interactions between HA and sialic acids on glycans of other HAs, and between HA and other glycoproteins in the mucus layer [14][18]. NA prevents this aggregation and allows for the efficient spread of newly produced virions in the host and the subsequent transmission between hosts [14][20]. Interestingly, NA also plays a critical role in virus infectivity and HA-mediated membrane fusion [21].
Shifting the immune response towards the second major glycoprotein, NA, is a promising option for the improvement of seasonal vaccines. NA has a slower rate of antigenic drift, has fewer subtypes (Figure 1B), and lower immune selective pressure [22][23][24]. Hence, NA is an attractive target and anti-NA antibodies can inhibit the enzymatic activity of the virus via direct binding or steric hinderance of the active site [25]. Additionally, animal studies indicate that the induction of an anti-NA antibody response can confer protection [26][27][28]. Human challenge studies performed in the early 1970s revealed that anti-NA antibody titers inversely correlated with virus shedding and disease symptoms [29][30]. Recent studies indicate that NA inhibition (NI) titers independently correlated with protection against influenza virus symptoms and resulted in decreased viral shedding [31][32][33][34]. Understanding the role of anti-NA antibodies in controlling influenza virus infection can be improved through the generation of monoclonal antibodies (mAbs). In this review, we summarize several studies that isolated and characterized anti-NA antibodies from humans, and we discuss how this information will provide supporting evidence for the inclusion of standardized amounts of NA in future vaccine preparations.
2. NA-Based Immunity
Antibody responses towards influenza virus antigens typically target the two major surface glycoproteins, HA and NA (Figure 1A) [35]. Despite the importance of both anti-HA and anti-NA antibodies in preventing and controlling influenza virus infection, HA usually exhibits immunodominance over NA following influenza vaccination [13][36][37]. On the other hand, natural influenza virus infection induces more balanced antibody responses towards HA and NA [37]. Natural infection results in high seroconversion rates against both HA and NA, as measured by enzyme-linked immunosorbent assay (ELISA) [38][39]. A study in H1N1 pandemic influenza virus-infected patients demonstrated that seroconversion to NA could be observed at day 7 and peaked at day 28. However, NA antibodies began to decline by day 90 [39]. In the case of N2 antibodies, one study reported that N2 antibodies began to decline to undetectable levels within 5 months following infection, while another study reported persistence of detectable N2 antibodies up to 4 years after infection [40][41]. It should be noted that, in general, N1 antibody titers are lower than N2 and influenza B NA antibodies [42]. The lower titers of N1 antibodies might be caused by the lower immunogenicity of N1 but could also be an artifact of the reagents used to measure these antibody titers [38][39][42].
Several different types of influenza virus vaccines are currently in use to help protect against influenza virus infections. Immunoglobin responses towards NA after vaccination are substantially reduced when compared to infection [37]. Even though there are several different vaccines against influenza virus, only a handful of the vaccines can induce an immune response against NA, and several of the licensed vaccines contain little to no (e.g., Flucelvax) antigenic NA [43]. Live-attenuated virus vaccines (LAIV), whole inactivated influenza vaccine (IIV) and some split virus vaccines can induce NA antibody responses of varying degrees [34][44][45][46][47]. Similar to infection, antibodies in humans that developed post-vaccination peaked at 2–3 weeks; however, they only persisted for one year [48][49][50][51]. Additionally, route of administration can also have an effect on the humoral response against NA [52][53]. Unlike antibody responses to natural infection, antibody responses to vaccination are short-lived, and antibody titers induced by vaccination may even decline within a given influenza season [44][54][55]. NA-specific human monoclonal antibodies (mAbs) that are induced by natural infection and vaccination will be further discussed in the upcoming sections.
2.1. Human mAbs That Target NA
HA and NA-specific antibodies utilize different modes of action to control influenza virus infection. Anti-HA mAbs predominantly bind to the globular head domain and inhibit virus attachment and entry into the host cell [56][57]. Thus, HA-specific mAbs have potent neutralizing activity [58]. Additionally, some HA head-specific mAbs facilitate Fc receptor-mediated cytotoxicity, such as antibody dependent cellular toxicity (ADCC) [59][60]. Several studies have described human mAbs that are directed against the receptor binding site of HA, which have neutralizing activity and are broadly protective in mice [61][62][63][64]. In contrast to the head-specific mAbs, mAbs that bind to HA stalk inhibit viral-endosomal fusion [65]. Although the titers of stalk binding mAbs in humans are typically low, they bind to HA from different subtypes and have much broader neutralizing capacity and increased Fc-FcR activity when compared to mAbs targeting the head domain [5][65][66][67][68][69]. Different to anti-HA mAbs, anti-NA mAbs play a major role at the later stages of viral replication, specifically when the influenza virion buds off from the infected cells [18]. During the final stages of viral replication, NA enzymatically cleaves off sialic acid residues on the host cell surface, releasing virus progeny [18][19]. It is at this point that most of the anti-NA mAbs inhibit viral egress [13][70]. Since NA mAbs are mostly effective during viral egress, virus titer is not generally affected during infection in an in vitro plaque reduction assay [71][72][73][74]. However, the plaque diameter is significantly reduced in the presence of anti-NA mAbs [72][73][74]. Therefore, most of the mAbs against NA are non-neutralizing but are still able to inhibit the enzymatic activity of NA and prevent virion release and spread from the host cell [25]. Furthermore, some NA-specific mAbs also mediate ADCC, which in turn activates natural killer (NK) cells [20][75][76][77]. Upon activation via effector cells (e.g., NK cells, macrophages), they can produce the antiviral cytokine IFN-γ and degranulate or phagocytose infected cells, aiding in the clearance of virus-infected cells [60][77][78][79]
Influenza virus vaccination and natural infection have the ability to induce a broad immune response against NA glycoprotein. This is demonstrated by the isolation of several human mAbs after both vaccination and natural infection. Even though some of the isolated human mAbs have a narrow reactivity, several of the isolated human mAbs have very broad reactivity spanning across both influenza A and influenza B strains (Figure 2 and Table 1). Below we describe human NA mAbs that have been isolated and their exciting reactivity.
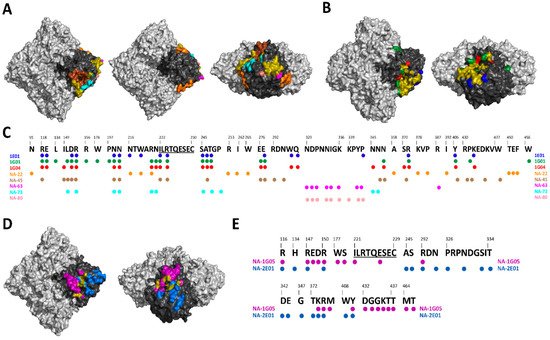
Figure 2. Mapping of NA-specific human monoclonal mAbs with known epitopes. (A) Top, bottom and side views of the A/Hunan/02650/2016 N9 (PDB ID: 6Q1Z) showing the epitopes of NA-22 in orange, NA-45 in brown, NA-63 in pink, NA-73 in teal, and NA-80 in salmon. (B) Top and side views of the A/California/04/2009 N1 (PDB ID: 6Q23) showing the epitopes of 1E01 in blue, 1G01 in green, and 1G04 in red. (C) Alignment of A/Hunan/02650/2016 N9 with the epitopes of 1E01, 1G01, 1G04, NA-22, NA-45, NA-63, NA-73, and NA-80. Universally conserved sequence “ILRTQESEC” is underlined. (D) Top and side views of the B/Perth/211/2001 NA (PDB ID: 3K38) showing the epitopes of NA-1G05 in purple and NA-2E01 in light blue. (E) Alignment of B/Perth/211/2001 with the epitopes of NA-1G05 and NA-2E01. Universally conserved sequence “ILRTQESEC” is underlined. For A, B and D overlapping epitopes between at least two mAbs are show in olive. Light gray denotes the NA tetramer, with the monomer highlighted in black.
Table 1. Summary of NA mAbs isolated from humans.
| Reactivity | Ref. | mAb Name | Induced after |
|---|---|---|---|
| Group 1 NA | [37] | 1000-3C05, 1000-2E06, 1000-3B04, 1000-3B06, EM-2E01, 1000-1D05, 1000-1E02, 1000-1H01, 294-G-1F01, 294-A-1C02, 295-G-2F04, 300-G-2A04, 300-G-2F04, 294-A-1C06 | H1N1 infection |
| [80] | AG7C, AF9C | Seasonal trivalent inactivated vaccine | |
| Group 2 NA | [37] | 229-1D05, 235-1C02, 235-1E06, 294-1A02, 228-1B03, 228-3F04, 2291B05, 229-1F06, 229-1G03, 229-2B04, 229-2C06, 229-2E02 | H3N2 infection |
| [70][81][82] | NA-97 | A/British Columbia/1/2015 (H7N9) natural infection | |
| [70][81][82] | NA-22, NA-45, NA-63, NA-73, NA-80 | A/Shanghai/2/2013 (H7N9) monovalent inactivated influenza vaccine | |
| Influenza B NA | [83] | NA-1A03, NA-1G05, NA-2D10, NA-2E01, NA-2H09, NA-3C01 | Influenza B infection |
| [84] | 1086C12, 1092D4, 1092E10, 1122C7 | Quadrivalent inactivated influenza vaccine | |
| Pan NA | 1G01, 1E01, 1G04 | H3N2 infection |
3. NA Human mAbs Inform Vaccine Design
The development of NA vaccine antigens is complicated by several factors. The skewed antibody response towards HA is mainly due the presence of approximately four times more HA than the NA on the influenza virion surface [85]. As a result of the immunodominance of HA over NA, HAs evolve more quickly than NAs. A H3N2 virus study showed that the globular domain of HA evolves at a rate of 12.9 × 10−3–14.9 × 10−3 amino acid/site/year compared to NA, which evolves at a rate of 9.1 × 10−3 amino acid/site/year [56][86]. While antibody responses against NA are the primary drivers of the antigenic drift, antibody response and altered affinity for NA/HA receptors play a role in NA/HA antigenic drift [33][87]. Furthermore, immunization with the same amount of purified HA and NA resulted in similar increases in antibody titers to each of the antigens, demonstrating that the two antigens have very similar immunogenicity [88]. Due to the lower drift and immunogenic properties of NA, there has been a concerted effort to use NA as a vaccine antigen [20][46][86].
As discussed in the above section, several broadly reactive human NA mAbs have been isolated either after natural infection or post-vaccination. These human NA mAbs display a broad range of protection ranging from homologous protection to different influenza subtypes. For example, human mAbs NA-22, NA-45, NA-63, NA-73 and NA-80 are only active against N9 subtypes [70][81] (Figure 2A). NA-1G05 and NA-2E01 are reactive against all influenza B types [83] (Figure 2D). Lastly, 1E01 and 1G01 are broadly reactive against all influenza A and B types [89] (Figure 2B). The identification of broadly reactive mAbs indicates the presence of conserved epitopes on NA antigen which can be utilized for future NA vaccine candidates (Figure 2C,E). Interestingly, children born after 2006 showed ELISA antibody titers against the ancestral A/South Carolina/1/1918 and B/Lee/1940 influenza virus strains. The ELISA antibody titers correlated positively with NAI titers [42]. Additionally, a recent clinical study in which healthy young adults were challenged with pandemic H1N1 demonstrated differences in the role of HA and NA-specific antibodies. While reduction in virus shedding correlated with HA inhibition titers; fewer symptoms, reduced symptom severity score, reduced duration of symptoms and reduced viral shedding correlated with NAI titers [31]. It has also been shown that NAI titers are independent predictors of immunity against the influenza virus and are an independent correlate of protection [33][34]. These protective mAbs against NA have three different mechanisms of inhibition: (i) direct inhibition of NA catalytic site, (ii) indirect inhibition of NA catalytic site via steric hindrance, and (iii) mAb with little to no NAI activity utilize Fc-FcR-based effector functions [75][89].
Antibodies against NA are not directly involved with preventing virus binding to the host receptors, similar to some anti-HA antibodies. Thus, anti-NA mAbs are not expected to inhibit infection but limit viral spread within the host, reduce morbidity and mortality, decrease viral shedding and reduce transmission to naïve hosts [88][90]. Thus, vaccines containing immunogenic amounts of both HA and NA would be optimal to provide complete protection against influenza virus infection [91]. HA and NA ratios are different for different subtypes and different strains within a subtype [92]. Therefore, NA content and HA:NA ratio in future vaccine candidates need to be standardized. Different assays such as mass spectrometry (MS), isotype dilution MS and capture ELISA to measure the potency of NA in vaccine preparations are under development [92][93][94]. Induction of broadly cross-reactive mAbs has indicated that NA is immunogenic, and that NA antigen contains broadly conserved epitopes.
These studies demonstrate the growing potential of using NA as a vaccine antigen. Advances in emerging platforms (discussed below), a greater understanding of NA structural biology and mAb characterization can inform the design and development of NA vaccine antigens that promote a broad antibody response. Even though the different studies discussed here provide evidence for the use of NA as a vaccine antigen, a slew of questions remain unanswered. The factors that drive long-lasting NA-specific immunity are not well understood. This knowledge could be beneficial in designing NA-based vaccines. What makes natural infection provide a broader and long-lasting antibody response compared to vaccination? Testing of the novel vaccine platforms that use NA as the primary antigen have, so far, been mostly restricted to mice, with only limited platforms assessed in guinea pigs and ferrets (Table 2). Therefore, could a NA vaccine platform that induces robust immune response in mice perform similarly in ferrets and guinea pigs? None of the currently licensed vaccines have standardized amounts of NA. In future vaccine preparations, should NA antigens be standardized to similar amounts or greater amounts than HA to produce a robust immune response? Current studies have shown that NA antigenically drifts at a much slower rate compared to HA. How will the development of a vaccine targeting NA potentially influence the evolution rate of NA? In addition, newly developed assays such as MS, isotype dilution MS and capture ELISA to measure potency of NA in vaccine preparations have been great tools in propelling NA as a vaccine antigen in future vaccine preparations [92][93][94]. Future studies that try to answer the above-mentioned questions along with several others are vital in the development of a future NA-based vaccines.
Table 2. Summary of emerging NA-based vaccine platforms against influenza viruses described in this review. + indicates low immunogenicity, +++ indicates high immunogenicity, N.D. indicates not determined. AA indicates amino acid.
| Platform | NA Antigen Subtype | Animal Model | Immunogenicity | Protection | Ref. |
|---|---|---|---|---|---|
| Inactivated vaccine | 30 AA insertion in seasonal N1 15 AA insertion in N2 |
Mice | +++ +++ |
N.D. | [95] |
| Recombinant NA vaccine | N2 | Human | + | N.D. | [96] |
| Seasonal N1 N2 B/Yamagata/16/88-like B-NA |
Mice | +++ +++ +++ |
Homologous Heterologous |
[27] | |
| Avian N1 Pandemic N1 |
Mice | + | Homologous Partial heterologous |
[97] | |
| N1 | Mice | +++ | Homologous | [98] | |
| N2 | Mice | +++ | Homologous Partial heterologous |
[99] | |
| B-NA | Mice Guinea pigs |
+ | Homologous Heterologous |
[100] | |
| B-NA | Guinea pigs | +++ | Homologous Partial heterologous |
[101] | |
| Virus like particles | Avian N1 | Ferrets | +++ | Homologous | [28] |
| Pandemic N1 | Mice | + | Homologous Heterologous |
[102] | |
| Avian N1 Seasonal N1 |
Mice | +++ | Homologous Heterologous |
[103] | |
| Viral replicon particles | Avian N1 | Chicken | +++ | N.D. | [104] |
| Viral Vector vaccines | Avian N1 Pandemic N1 |
Mice | +++ | Homologous Heterologous Heterosubtypic |
[105] |
| N3 N9 |
Mice | +++ | Homologous | [106] | |
| Nucleic Acid-DNA | Seasonal N1 | Mice | + | Homologous Partial heterologous |
[107] |
| N2 | Mice | + | Homologous Partial heterologous |
[108] | |
| Nucleic Acid-RNA | Seasonal N1 | Mice | +++ | Homologous | [109] |
4. Emerging Platforms for the Development of NA-Based Vaccines
Vaccine candidates that target NA have been frequently revisited since the 1968 Hong Kong influenza A (H3N2) pandemic. The first NA-based inactivated vaccine, which consisted of an irrelevant equine HA and a NA from A/Hong Kong/1/1968 (H3N2), protected against challenge with a virus carrying an antigenically identical NA but a mismatched HA [29]. Despite these encouraging results, NA as a vaccine antigen has only received limited attention in the past. Early immunogenicity studies did not frequently evaluate antibody responses against NA as it was difficult to perform the assay safely, reproducibly and at high throughput [110][111][112]. Furthermore, the amount of NA varied in different viruses and was not easily quantified [20]. Lastly the unstable nature of NAs resulted in conflicting immunogenicity studies [110][113]. As a result, the development of NA-based vaccines using traditional egg-based vaccine platforms has been relatively inactive since 1998 [113]. Emerging vaccine platforms, such as modified inactivated vaccines, recombinant NAs, virus-like particles (VLP), virus replicon particles (VRP), viral vector platforms and nucleic acid vaccines (Table 2), could be used to overcome previously unsuccessful attempts to develop NA as a vaccine antigen. Here we will describe these vaccine platforms and how they have been used in a pre-clinical setting to induce NA antibody responses.
References
- Bernstein, D.I.; Guptill, J.; Naficy, A.; Nachbagauer, R.; Berlanda-Scorza, F.; Feser, J.; Wilson, P.C.; Solorzano, A.; Van der Wielen, M.; Walter, E.B.; et al. Immunogenicity of chimeric haemagglutinin-based, universal influenza virus vaccine candidates: Interim results of a randomised, placebo-controlled, phase 1 clinical trial. Lancet Infect. Dis. 2020, 20, 80–91.
- CDC. Vaccine Effectiveness: How Well Do the Flu Vaccines Work? 2020. Available online: https://www.cdc.gov/flu/vaccines-work/vaccineeffect.htm (accessed on 10 February 2021).
- CDC. Who Is at High Risk for Flu Complications. 2018. Available online: https://www.cdc.gov/flu/highrisk/index.htm (accessed on 1 January 2021).
- WHO. Influenza (Seasonal). 2018. Available online: https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 22 January 2021).
- Nachbagauer, R.; Liu, W.C.; Choi, A.; Wohlbold, T.J.; Atlas, T.; Rajendran, M.; Solorzano, A.; Berlanda-Scorza, F.; Garcia-Sastre, A.; Palese, P.; et al. A universal influenza virus vaccine candidate confers protection against pandemic H1N1 infection in preclinical ferret studies. NPJ Vaccines 2017, 2, 26.
- Shaw, M.L.; Palese, P. Orthomyxoviridae: The Viruses and Their Replication. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 1691–1740.
- Bouvier, N.M.; Palese, P. The Biology of Influenza Viruses. Vaccine 2008, 26 (Suppl. S4), D49–D53.
- Heaton, N.S.; Sachs, D.; Chen, C.J.; Hai, R.; Palese, P. Genome-wide mutagenesis of influenza virus reveals unique plasticity of the hemagglutinin and NS1 proteins. Proc. Natl. Acad. Sci. USA 2013, 110, 20248–20253.
- Xie, H.; Wan, X.F.; Ye, Z.; Plant, E.P.; Zhao, Y.; Xu, Y.; Li, X.; Finch, C.; Zhao, N.; Kawano, T.; et al. H3N2 Mismatch of 2014-15 Northern Hemisphere Influenza Vaccines and Head-to-head Comparison between Human and Ferret Antisera derived Antigenic Maps. Sci. Rep. 2015, 5, 15279.
- De Jong, J.C.; Beyer, W.E.; Palache, A.M.; Rimmelzwaan, G.F.; Osterhaus, A.D. Mismatch between the 1997/1998 influenza vaccine and the major epidemic A(H3N2) virus strain as the cause of an inadequate vaccine-induced antibody response to this strain in the elderly. J. Med. Virol. 2000, 61, 94–99.
- CDC. Past Pandemics. 2018. Available online: https://www.cdc.gov/flu/pandemic-resources/basics/past-pandemics.html (accessed on 21 January 2021).
- Krammer, F.; Palese, P. Advances in the development of influenza virus vaccines. Nat. Rev. Drug. Discov. 2015, 14, 167–182.
- Wohlbold, T.J.; Krammer, F. In the shadow of hemagglutinin: A growing interest in influenza viral neuraminidase and its role as a vaccine antigen. Viruses 2014, 6, 2465–2494.
- Eichelberger, M.C.; Wan, H. Influenza neuraminidase as a vaccine antigen. Curr. Top. Microbiol. Immunol. 2015, 386, 275–299.
- Matrosovich, M.N.; Matrosovich, T.Y.; Gray, T.; Roberts, N.A.; Klenk, H.D. Neuraminidase is important for the initiation of influenza virus infection in human airway epithelium. J. Virol. 2004, 78, 12665–12667.
- Cohen, M.; Zhang, X.Q.; Senaati, H.P.; Chen, H.W.; Varki, N.M.; Schooley, R.T.; Gagneux, P. Influenza A penetrates host mucus by cleaving sialic acids with neuraminidase. Virol. J. 2013, 10, 321.
- Ma, J.; Rubin, B.K.; Voynow, J.A. Mucins, Mucus, and Goblet Cells. Chest 2018, 154, 169–176.
- McAuley, J.L.; Gilbertson, B.P.; Trifkovic, S.; Brown, L.E.; McKimm-Breschkin, J.L. Influenza Virus Neuraminidase Structure and Functions. Front. Microbiol. 2019, 10, 39.
- Shtyrya, Y.A.; Mochalova, L.V.; Bovin, N.V. Influenza virus neuraminidase: Structure and function. Acta Nat. 2009, 1, 26–32.
- Krammer, F.; Fouchier, R.A.M.; Eichelberger, M.C.; Webby, R.J.; Shaw-Saliba, K.; Wan, H.; Wilson, P.C.; Compans, R.W.; Skountzou, I.; Monto, A.S. NAction! How Can Neuraminidase-Based Immunity Contribute to Better Influenza Virus Vaccines? mBio 2018, 9, e02332-17.
- Su, B.; Wurtzer, S.; Rameix-Welti, M.A.; Dwyer, D.; van der Werf, S.; Naffakh, N.; Clavel, F.; Labrosse, B. Enhancement of the influenza A hemagglutinin (HA)-mediated cell-cell fusion and virus entry by the viral neuraminidase (NA). PLoS ONE 2009, 4, e8495.
- Kilbourne, E.D.; Johansson, B.E.; Grajower, B. Independent and disparate evolution in nature of influenza A virus hemagglutinin and neuraminidase glycoproteins. Proc. Natl. Acad. Sci. USA 1990, 87, 786–790.
- Westgeest, K.B.; de Graaf, M.; Fourment, M.; Bestebroer, T.M.; van Beek, R.; Spronken, M.I.; de Jong, J.C.; Rimmelzwaan, G.F.; Russell, C.A.; Osterhaus, A.D.; et al. Genetic evolution of the neuraminidase of influenza A (H3N2) viruses from 1968 to 2009 and its correspondence to haemagglutinin evolution. J. Gen. Virol. 2012, 93, 1996–2007.
- Schulman, J.L.; Kilbourne, E.D. Independent variation in nature of hemagglutinin and neuraminidase antigens of influenza virus: Distinctiveness of hemagglutinin antigen of Hong Kong-68 virus. Proc. Natl. Acad. Sci. USA 1969, 63, 326–333.
- Couzens, L.; Gao, J.; Westgeest, K.; Sandbulte, M.; Lugovtsev, V.; Fouchier, R.; Eichelberger, M. An optimized enzyme-linked lectin assay to measure influenza A virus neuraminidase inhibition antibody titers in human sera. J. Virol. Methods 2014, 210, 7–14.
- Schulman, J.L.; Khakpour, M.; Kilbourne, E.D. Protective effects of specific immunity to viral neuraminidase on influenza virus infection of mice. J. Virol. 1968, 2, 778–786.
- Wohlbold, T.J.; Nachbagauer, R.; Xu, H.; Tan, G.S.; Hirsh, A.; Brokstad, K.A.; Cox, R.J.; Palese, P.; Krammer, F. Vaccination with adjuvanted recombinant neuraminidase induces broad heterologous, but not heterosubtypic, cross-protection against influenza virus infection in mice. mBio 2015, 6, e02556.
- Smith, G.E.; Sun, X.; Bai, Y.; Liu, Y.V.; Massare, M.J.; Pearce, M.B.; Belser, J.A.; Maines, T.R.; Creager, H.M.; Glenn, G.M.; et al. Neuraminidase-based recombinant virus-like particles protect against lethal avian influenza A(H5N1) virus infection in ferrets. Virology 2017, 509, 90–97.
- Couch, R.B.; Kasel, J.A.; Gerin, J.L.; Schulman, J.L.; Kilbourne, E.D. Induction of partial immunity to influenza by a neuraminidase-specific vaccine. J. Infect. Dis. 1974, 129, 411–420.
- Murphy, B.R.; Kasel, J.A.; Chanock, R.M. Association of serum anti-neuraminidase antibody with resistance to influenza in man. N. Engl. J. Med. 1972, 286, 1329–1332.
- Memoli, M.J.; Shaw, P.A.; Han, A.; Czajkowski, L.; Reed, S.; Athota, R.; Bristol, T.; Fargis, S.; Risos, K.; Powers, J.H.; et al. Evaluation of Antihemagglutinin and Antineuraminidase Antibodies as Correlates of Protection in an Influenza A/H1N1 Virus Healthy Human Challenge Model. mBio 2016, 7, e00417-16.
- Maier, H.E.; Nachbagauer, R.; Kuan, G.; Ng, S.; Lopez, R.; Sanchez, N.; Stadlbauer, D.; Gresh, L.; Schiller, A.; Rajabhathor, A.; et al. Pre-existing anti-neuraminidase antibodies are associated with shortened duration of influenza A (H1N1)pdm virus shedding and illness in naturally infected adults. Clin. Infect. Dis. 2019, 70, 2290–2297.
- Couch, R.B.; Atmar, R.L.; Franco, L.M.; Quarles, J.M.; Wells, J.; Arden, N.; Nino, D.; Belmont, J.W. Antibody correlates and predictors of immunity to naturally occurring influenza in humans and the importance of antibody to the neuraminidase. J. Infect. Dis. 2013, 207, 974–981.
- Monto, A.S.; Petrie, J.G.; Cross, R.T.; Johnson, E.; Liu, M.; Zhong, W.; Levine, M.; Katz, J.M.; Ohmit, S.E. Antibody to Influenza Virus Neuraminidase: An Independent Correlate of Protection. J. Infect. Dis. 2015, 212, 1191–1199.
- Smith, W.; Andrews, C.H.; Laidlaw, P.P. A virus obtained from influenza patients. Lancet 1933, 222, 66–68.
- Johansson, B.E.; Moran, T.M.; Bona, C.A.; Popple, S.W.; Kilbourne, E.D. Immunologic response to influenza virus neuraminidase is influenced by prior experience with the associated viral hemagglutinin. II. Sequential infection of mice simulates human experience. J. Immunol. 1987, 139, 2010–2014.
- Chen, Y.Q.; Wohlbold, T.J.; Zheng, N.Y.; Huang, M.; Huang, Y.; Neu, K.E.; Lee, J.; Wan, H.; Rojas, K.T.; Kirkpatrick, E.; et al. Influenza Infection in Humans Induces Broadly Cross-Reactive and Protective Neuraminidase-Reactive Antibodies. Cell 2018, 173, 417–429.e10.
- Nachbagauer, R.; Choi, A.; Hirsh, A.; Margine, I.; Iida, S.; Barrera, A.; Ferres, M.; Albrecht, R.A.; Garcia-Sastre, A.; Bouvier, N.M.; et al. Defining the antibody cross-reactome directed against the influenza virus surface glycoproteins. Nat. Immunol. 2017, 18, 464–473.
- Changsom, D.; Jiang, L.; Lerdsamran, H.; Iamsirithaworn, S.; Kitphati, R.; Pooruk, P.; Auewarakul, P.; Puthavathana, P. Kinetics, Longevity, and Cross-Reactivity of Antineuraminidase Antibody after Natural Infection with Influenza A Viruses. Clin. Vaccine Immunol. 2017, 24, e00248-17.
- Smith, A.J.; Davies, J.R. Natural infection with influenza A (H3N2). The development, persistance and effect of antibodies to the surface antigens. Epidemiol. Infect. 1976, 77, 271–282.
- Schild, G.C. Antibody against influenza A2 virus neuraminidase in human sera. J. Hyg. 1969, 67, 353–365.
- Rajendran, M.; Nachbagauer, R.; Ermler, M.E.; Bunduc, P.; Amanat, F.; Izikson, R.; Cox, M.; Palese, P.; Eichelberger, M.; Krammer, F. Analysis of Anti-Influenza Virus Neuraminidase Antibodies in Children, Adults, and the Elderly by ELISA and Enzyme Inhibition: Evidence for Original Antigenic Sin. mBio 2017, 8.
- Gao, Z.; Robinson, K.; Skowronski, D.M.; De Serres, G.; Withers, S.G. Quantification of the total neuraminidase content of recent commercially-available influenza vaccines: Introducing a neuraminidase titration reagent. Vaccine 2020, 38, 715–718.
- Petrie, J.G.; Ohmit, S.E.; Johnson, E.; Truscon, R.; Monto, A.S. Persistence of Antibodies to Influenza Hemagglutinin and Neuraminidase Following One or Two Years of Influenza Vaccination. J. Infect. Dis. 2015, 212, 1914–1922.
- Ehrlich, H.J.; Muller, M.; Kollaritsch, H.; Pinl, F.; Schmitt, B.; Zeitlinger, M.; Loew-Baselli, A.; Kreil, T.R.; Kistner, O.; Portsmouth, D.; et al. Pre-vaccination immunity and immune responses to a cell culture-derived whole-virus H1N1 vaccine are similar to a seasonal influenza vaccine. Vaccine 2012, 30, 4543–4551.
- Couch, R.B.; Atmar, R.L.; Keitel, W.A.; Quarles, J.M.; Wells, J.; Arden, N.; Nino, D. Randomized comparative study of the serum antihemagglutinin and antineuraminidase antibody responses to six licensed trivalent influenza vaccines. Vaccine 2012, 31, 190–195.
- Laguio-Vila, M.R.; Thompson, M.G.; Reynolds, S.; Spencer, S.M.; Gaglani, M.; Naleway, A.; Ball, S.; Bozeman, S.; Baker, S.; Martinez-Sobrido, L.; et al. Comparison of serum hemagglutinin and neuraminidase inhibition antibodies after 2010-2011 trivalent inactivated influenza vaccination in healthcare personnel. Open Forum Infect. Dis. 2015, 2, ofu115.
- Gross, P.A.; Russo, C.; Dran, S.; Cataruozolo, P.; Munk, G.; Lancey, S.C. Time to earliest peak serum antibody response to influenza vaccine in the elderly. Clin. Diagn. Lab. Immunol. 1997, 4, 491–492.
- Gross, P.A.; Russo, C.; Teplitzky, M.; Dran, S.; Cataruozolo, P.; Munk, G. Time to peak serum antibody response to influenza vaccine in the elderly. Clin. Diagn. Lab. Immunol. 1996, 3, 361–362.
- Rastogi, S.; Gross, P.A.; Bonelli, J.; Dran, S.; Levandowski, R.A.; Russo, C.; Weksler, M.E.; Kaye, D.; Levison, M.; Abrutyn, E.; et al. Time to peak serum antibody response to influenza vaccine. Clin. Diagn. Lab. Immunol. 1995, 2, 120–121.
- Kositanont, U.; Assantachai, P.; Wasi, C.; Puthavathana, P.; Praditsuwan, R. Kinetics of the antibody response to seasonal influenza vaccination among the elderly. Viral Immunol. 2012, 25, 471–476.
- Mohn, K.G.; Smith, I.; Sjursen, H.; Cox, R.J. Immune responses after live attenuated influenza vaccination. Hum. Vaccin. Immunother. 2018, 14, 571–578.
- Ghendon, Y. The immune response to influenza vaccines. Acta Virol. 1990, 34, 295–304.
- Belongia, E.A.; Sundaram, M.E.; McClure, D.L.; Meece, J.K.; Ferdinands, J.; VanWormer, J.J. Waning vaccine protection against influenza A (H3N2) illness in children and older adults during a single season. Vaccine 2015, 33, 246–251.
- Puig-Barbera, J.; Mira-Iglesias, A.; Tortajada-Girbes, M.; Lopez-Labrador, F.X.; Librero-Lopez, J.; Diez-Domingo, J.; Carballido-Fernandez, M.; Carratala-Munuera, C.; Correcher-Medina, P.; Gil-Guillen, V.; et al. Waning protection of influenza vaccination during four influenza seasons, 2011/2012 to 2014/2015. Vaccine 2017, 35, 5799–5807.
- Kirkpatrick, E.; Qiu, X.; Wilson, P.C.; Bahl, J.; Krammer, F. The influenza virus hemagglutinin head evolves faster than the stalk domain. Sci. Rep. 2018, 8, 10432.
- Bangaru, S.; Lang, S.; Schotsaert, M.; Vanderven, H.A.; Zhu, X.; Kose, N.; Bombardi, R.; Finn, J.A.; Kent, S.J.; Gilchuk, P.; et al. A Site of Vulnerability on the Influenza Virus Hemagglutinin Head Domain Trimer Interface. Cell 2019, 177, 1136–1152.e18.
- Corti, D.; Cameroni, E.; Guarino, B.; Kallewaard, N.L.; Zhu, Q.; Lanzavecchia, A. Tackling influenza with broadly neutralizing antibodies. Curr. Opin. Virol. 2017, 24, 60–69.
- Tay, M.Z.; Wiehe, K.; Pollara, J. Antibody-Dependent Cellular Phagocytosis in Antiviral Immune Responses. Front. Immunol. 2019, 10, 332.
- Jegaskanda, S. The Potential Role of Fc-Receptor Functions in the Development of a Universal Influenza Vaccine. Vaccines 2018, 6, 27.
- Liu, Y.; Tan, H.X.; Koutsakos, M.; Jegaskanda, S.; Esterbauer, R.; Tilmanis, D.; Aban, M.; Kedzierska, K.; Hurt, A.C.; Kent, S.J.; et al. Cross-lineage protection by human antibodies binding the influenza B hemagglutinin. Nat. Commun. 2019, 10, 324.
- Whittle, J.R.; Zhang, R.; Khurana, S.; King, L.R.; Manischewitz, J.; Golding, H.; Dormitzer, P.R.; Haynes, B.F.; Walter, E.B.; Moody, M.A. Broadly neutralizing human antibody that recognizes the receptor-binding pocket of influenza virus hemagglutinin. Proc. Natl. Acad. Sci. USA 2011, 108, 14216–14221.
- Zost, S.J.; Lee, J.; Gumina, M.E.; Parkhouse, K.; Henry, C.; Wu, N.C.; Lee, C.D.; Wilson, I.A.; Wilson, P.C.; Bloom, J.D.; et al. Identification of Antibodies Targeting the H3N2 Hemagglutinin Receptor Binding Site following Vaccination of Humans. Cell 2019, 29, 4460–4470.e8.
- Shen, C.; Zhang, M.; Chen, Y.; Zhang, L.; Wang, G.; Chen, J.; Chen, S.; Li, Z.; Wei, F.; Chen, J.; et al. An IgM antibody targeting the receptor binding site of influenza B blocks viral infection with great breadth and potency. Theranostics 2019, 9, 210–231.
- Krammer, F.; Palese, P. Influenza virus hemagglutinin stalk-based antibodies and vaccines. Curr. Opin. Virol. 2013, 3, 521–530.
- Rajendran, M.; Sun, W.; Comella, P.; Nachbagauer, R.; Wohlbold, T.J.; Amanat, F.; Kirkpatrick, E.; Palese, P.; Krammer, F. An immuno-assay to quantify influenza virus hemagglutinin with correctly folded stalk domains in vaccine preparations. PLoS ONE 2018, 13, e0194830.
- Sui, J.; Hwang, W.C.; Perez, S.; Wei, G.; Aird, D.; Chen, L.M.; Santelli, E.; Stec, B.; Cadwell, G.; Ali, M.; et al. Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses. Nat. Struct. Mol. Biol. 2009, 16, 265–273.
- Ekiert, D.C.; Bhabha, G.; Elsliger, M.A.; Friesen, R.H.; Jongeneelen, M.; Throsby, M.; Goudsmit, J.; Wilson, I.A. Antibody recognition of a highly conserved influenza virus epitope. Science 2009, 324, 246–251.
- Ekiert, D.C.; Friesen, R.H.; Bhabha, G.; Kwaks, T.; Jongeneelen, M.; Yu, W.; Ophorst, C.; Cox, F.; Korse, H.J.; Brandenburg, B.; et al. A highly conserved neutralizing epitope on group 2 influenza A viruses. Science 2011, 333, 843–850.
- Gilchuk, I.M.; Bangaru, S.; Gilchuk, P.; Irving, R.P.; Kose, N.; Bombardi, R.G.; Thornburg, N.J.; Creech, C.B.; Edwards, K.M.; Li, S.; et al. Influenza H7N9 Virus Neuraminidase-Specific Human Monoclonal Antibodies Inhibit Viral Egress and Protect from Lethal Influenza Infection in Mice. Cell Host Microbe 2019, 26, 715–728.e8.
- Jiang, L.; Fantoni, G.; Couzens, L.; Gao, J.; Plant, E.; Ye, Z.; Eichelberger, M.C.; Wan, H. Comparative Efficacy of Monoclonal Antibodies That Bind to Different Epitopes of the 2009 Pandemic H1N1 Influenza Virus Neuraminidase. J. Virol. 2016, 90, 117–128.
- Wan, H.; Gao, J.; Xu, K.; Chen, H.; Couzens, L.K.; Rivers, K.H.; Easterbrook, J.D.; Yang, K.; Zhong, L.; Rajabi, M.; et al. Molecular basis for broad neuraminidase immunity: Conserved epitopes in seasonal and pandemic H1N1 as well as H5N1 influenza viruses. J. Virol. 2013, 87, 9290–9300.
- Wohlbold, T.J.; Chromikova, V.; Tan, G.S.; Meade, P.; Amanat, F.; Comella, P.; Hirsh, A.; Krammer, F. Hemagglutinin Stalk- and Neuraminidase-Specific Monoclonal Antibodies Protect against Lethal H10N8 Influenza Virus Infection in Mice. J. Virol. 2016, 90, 851–861.
- Shcherbik, S.; Carney, P.; Pearce, N.; Stevens, J.; Dugan, V.G.; Wentworth, D.E.; Bousse, T. Monoclonal antibody against N2 neuraminidase of cold adapted A/Leningrad/134/17/57 (H2N2) enables efficient generation of live attenuated influenza vaccines. Virology 2018, 522, 65–72.
- Job, E.R.; Ysenbaert, T.; Smet, A.; Van Hecke, A.; Meuris, L.; Kleanthous, H.; Saelens, X.; Vogel, T.U. Fcgamma Receptors Contribute to the Antiviral Properties of Influenza Virus Neuraminidase-Specific Antibodies. mBio 2019, 10, e01667-19.
- Von Holle, T.A.; Moody, M.A. Influenza and Antibody-Dependent Cellular Cytotoxicity. Front. Immunol. 2019, 10, 1457.
- Jegaskanda, S.; Reading, P.C.; Kent, S.J. Influenza-specific antibody-dependent cellular cytotoxicity: Toward a universal influenza vaccine. J. Immunol. 2014, 193, 469–475.
- Jegaskanda, S.; Weinfurter, J.T.; Friedrich, T.C.; Kent, S.J. Antibody-dependent cellular cytotoxicity is associated with control of pandemic H1N1 influenza virus infection of macaques. J. Virol. 2013, 87, 5512–5522.
- Valkenburg, S.A.; Fang, V.J.; Leung, N.H.; Chu, D.K.; Ip, D.K.; Perera, R.A.; Wang, Y.; Li, A.P.; Peiris, J.M.; Cowling, B.J.; et al. Cross-reactive antibody-dependent cellular cytotoxicity antibodies are increased by recent infection in a household study of influenza transmission. Clin. Transl. Immunol. 2019, 8, e1092.
- Rijal, P.; Wang, B.B.; Tan, T.K.; Schimanski, L.; Janesch, P.; Dong, T.; McCauley, J.W.; Daniels, R.S.; Townsend, A.R.; Huang, K.A. Broadly Inhibiting Antineuraminidase Monoclonal Antibodies Induced by Trivalent Influenza Vaccine and H7N9 Infection in Humans. J. Virol. 2020, 94.
- Zhu, X.; Turner, H.L.; Lang, S.; McBride, R.; Bangaru, S.; Gilchuk, I.M.; Yu, W.; Paulson, J.C.; Crowe, J.E., Jr.; Ward, A.B.; et al. Structural Basis of Protection against H7N9 Influenza Virus by Human Anti-N9 Neuraminidase Antibodies. Cell Host Microbe 2019, 26, 729–738.e4.
- Krammer, F.; Li, L.; Wilson, P.C. Emerging from the Shadow of Hemagglutinin: Neuraminidase Is an Important Target for Influenza Vaccination. Cell Host Microbe 2019, 26, 712–713.
- Madsen, A.; Dai, Y.N.; McMahon, M.; Schmitz, A.J.; Turner, J.S.; Tan, J.; Lei, T.; Alsoussi, W.B.; Strohmeier, S.; Amor, M.; et al. Human Antibodies Targeting Influenza B Virus Neuraminidase Active Site Are Broadly Protective. Immunity 2020, 53, 852–863.e7.
- Piepenbrink, M.S.; Nogales, A.; Basu, M.; Fucile, C.F.; Liesveld, J.L.; Keefer, M.C.; Rosenberg, A.F.; Martinez-Sobrido, L.; Kobie, J.J. Broad and Protective Influenza B Virus Neuraminidase Antibodies in Humans after Vaccination and their Clonal Persistence as Plasma Cells. mBio 2019, 10, e00066-19.
- Webster, R.G.; Laver, W.G.; Kilbourne, E.D. Reactions of Antibodies with Surface Antigens of Influenza Virus. J. Gen. Virol. 1968, 3, 315–326.
- Westgeest, K.B.; Russell, C.A.; Lin, X.; Spronken, M.I.; Bestebroer, T.M.; Bahl, J.; van Beek, R.; Skepner, E.; Halpin, R.A.; de Jong, J.C.; et al. Genomewide analysis of reassortment and evolution of human influenza A(H3N2) viruses circulating between 1968 and 2011. J. Virol. 2014, 88, 2844–2857.
- Hensley, S.E.; Das, S.R.; Gibbs, J.S.; Bailey, A.L.; Schmidt, L.M.; Bennink, J.R.; Yewdell, J.W. Influenza A virus hemagglutinin antibody escape promotes neuraminidase antigenic variation and drug resistance. PLoS ONE 2011, 6, e15190.
- Johansson, B.E.; Bucher, D.J.; Kilbourne, E.D. Purified influenza virus hemagglutinin and neuraminidase are equivalent in stimulation of antibody response but induce contrasting types of immunity to infection. J. Virol. 1989, 63, 1239–1246.
- Stadlbauer, D.; Zhu, X.; McMahon, M.; Turner, J.S.; Wohlbold, T.J.; Schmitz, A.J.; Strohmeier, S.; Yu, W.; Nachbagauer, R.; Mudd, P.A.; et al. Broadly protective human antibodies that target the active site of influenza virus neuraminidase. Science 2019, 366, 499–504.
- Matthew, J.; Syle, D.L.S. Influenza Neuraminidase as a Vaccine Antigen. In Vaccines for Pandemic Influenza; Richard, W., Compans, W.A.O., Eds.; Springer: Berlin, Germany, 2009; Volume 333, p. 512.
- Jagadesh, A.; Salam, A.A.; Mudgal, P.P.; Arunkumar, G. Influenza virus neuraminidase (NA): A target for antivirals and vaccines. Arch. Virol. 2016, 161, 2087–2094.
- Getie-Kebtie, M.; Sultana, I.; Eichelberger, M.; Alterman, M. Label-free mass spectrometry-based quantification of hemagglutinin and neuraminidase in influenza virus preparations and vaccines. Influenza Other Respir. Viruses 2013, 7, 521–530.
- Williams, T.L.; Pirkle, J.L.; Barr, J.R. Simultaneous quantification of hemagglutinin and neuraminidase of influenza virus using isotope dilution mass spectrometry. Vaccine 2012, 30, 2475–2482.
- Wan, H.; Sultana, I.; Couzens, L.K.; Mindaye, S.; Eichelberger, M.C. Assessment of influenza A neuraminidase (subtype N1) potency by ELISA. J. Virol. Methods 2017, 244, 23–28.
- Broecker, F.; Zheng, A.; Suntronwong, N.; Sun, W.; Bailey, M.J.; Krammer, F.; Palese, P. Extending the Stalk Enhances Immunogenicity of the Influenza Virus Neuraminidase. J. Virol. 2019, 93.
- Kilbourne, E.D.; Couch, R.B.; Kasel, J.A.; Keitel, W.A.; Cate, T.R.; Quarles, J.H.; Grajower, B.; Pokorny, B.A.; Johansson, B.E. Purified influenza A virus N2 neuraminidase vaccine is immunogenic and non-toxic in humans. Vaccine 1995, 13, 1799–1803.
- Liu, W.C.; Lin, C.Y.; Tsou, Y.T.; Jan, J.T.; Wu, S.C. Cross-Reactive Neuraminidase-Inhibiting Antibodies Elicited by Immunization with Recombinant Neuraminidase Proteins of H5N1 and Pandemic H1N1 Influenza A Viruses. J. Virol. 2015, 89, 7224–7234.
- Job, E.R.; Ysenbaert, T.; Smet, A.; Christopoulou, I.; Strugnell, T.; Oloo, E.O.; Oomen, R.P.; Kleanthous, H.; Vogel, T.U.; Saelens, X. Broadened immunity against influenza by vaccination with computationally designed influenza virus N1 neuraminidase constructs. NPJ Vaccines 2018, 3, 55.
- Brett, I.C.; Johansson, B.E. Immunization against influenza A virus: Comparison of conventional inactivated, live-attenuated and recombinant baculovirus produced purified hemagglutinin and neuraminidase vaccines in a murine model system. Virology 2005, 339, 273–280.
- Johansson, B.E.; Brett, I.C. Recombinant influenza B virus HA and NA antigens administered in equivalent amounts are immunogenically equivalent and induce equivalent homotypic and broader heterovariant protection in mice than conventional and live influenza vaccines. Hum. Vaccin. 2008, 4, 420–424.
- McMahon, M.; Kirkpatrick, E.; Stadlbauer, D.; Strohmeier, S.; Bouvier, N.M.; Krammer, F. Mucosal Immunity against Neuraminidase Prevents Influenza B Virus Transmission in Guinea Pigs. mBio 2019, 10.
- Kim, K.H.; Lee, Y.T.; Park, S.; Jung, Y.J.; Lee, Y.; Ko, E.J.; Kim, Y.J.; Li, X.; Kang, S.M. Neuraminidase expressing virus-like particle vaccine provides effective cross protection against influenza virus. Virology 2019, 535, 179–188.
- Easterbrook, J.D.; Schwartzman, L.M.; Gao, J.; Kash, J.C.; Morens, D.M.; Couzens, L.; Wan, H.; Eichelberger, M.C.; Taubenberger, J.K. Protection against a lethal H5N1 influenza challenge by intranasal immunization with virus-like particles containing 2009 pandemic H1N1 neuraminidase in mice. Virology 2012, 432, 39–44.
- Halbherr, S.J.; Ludersdorfer, T.H.; Ricklin, M.; Locher, S.; Berger Rentsch, M.; Summerfield, A.; Zimmer, G. Biological and protective properties of immune sera directed to the influenza virus neuraminidase. J. Virol. 2015, 89, 1550–1563.
- Mooney, A.J.; Gabbard, J.D.; Li, Z.; Dlugolenski, D.A.; Johnson, S.K.; Tripp, R.A.; He, B.; Tompkins, S.M. Vaccination with Recombinant Parainfluenza Virus 5 Expressing Neuraminidase Protects against Homologous and Heterologous Influenza Virus Challenge. J. Virol. 2017, 91.
- Meseda, C.A.; Atukorale, V.; Soto, J.; Eichelberger, M.C.; Gao, J.; Wang, W.; Weiss, C.D.; Weir, J.P. Immunogenicity and Protection Against Influenza H7N3 in Mice by Modified Vaccinia Virus Ankara Vectors Expressing Influenza Virus Hemagglutinin or Neuraminidase. Sci. Rep. 2018, 8, 5364.
- Sandbulte, M.R.; Jimenez, G.S.; Boon, A.C.; Smith, L.R.; Treanor, J.J.; Webby, R.J. Cross-reactive neuraminidase antibodies afford partial protection against H5N1 in mice and are present in unexposed humans. PLoS Med. 2007, 4, e59.
- Chen, Z.; Kadowaki, S.; Hagiwara, Y.; Yoshikawa, T.; Matsuo, K.; Kurata, T.; Tamura, S. Cross-protection against a lethal influenza virus infection by DNA vaccine to neuraminidase. Vaccine 2000, 18, 3214–3222.
- Freyn, A.W.; Ramos da Silva, J.; Rosado, V.C.; Bliss, C.M.; Pine, M.; Mui, B.L.; Tam, Y.K.; Madden, T.D.; de Souza Ferreira, L.C.; Weissman, D.; et al. A Multi-Targeting, Nucleoside-Modified mRNA Influenza Virus Vaccine Provides Broad Protection in Mice. Mol. Ther. 2020, 28, 1569–1584.
- Nicholson, K.G.; Webster, R.G.; Hay, A. Standardization of Inactivated Influenza Vaccines. In Textbook of Influenza; Woods, J.M., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 1998; pp. 333–345.
- Van Deusen, R.A.; Hinshaw, V.S.; Senne, D.A.; Pellacani, D. Micro neuraminidase-inhibition assay for classification of influenza A virus neuraminidases. Avian Dis. 1983, 27, 745–750.
- Aminoff, D. Methods for the quantitative estimation of N-acetylneuraminic acid and their application to hydrolysates of sialomucoids. Biochem. J. 1961, 81, 384–392.
- Eichelberger, M.C.; Monto, A.S. Neuraminidase, the Forgotten Surface Antigen, Emerges as an Influenza Vaccine Target for Broadened Protection. J. Infect. Dis. 2019, 219, S75–S80.
More
Information
Subjects:
Virology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
978
Revisions:
3 times
(View History)
Update Date:
25 Aug 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




