
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nobuo Fuke | + 3716 word(s) | 3716 | 2021-08-13 05:26:38 | | | |
| 2 | Peter Tang | + 19 word(s) | 3735 | 2021-08-25 03:00:36 | | |
Video Upload Options
Metabolic endotoxemia is a condition in which blood lipopolysaccharide (LPS) levels are elevated, regardless of the presence of obvious infection. It has been suggested to lead to chronic inflammation-related diseases such as obesity, type 2 diabetes mellitus, non-alcoholic fatty liver disease (NAFLD), pancreatitis, amyotrophic lateral sclerosis, and Alzheimer’s disease.
1. Introduction
2. Fat Intake and Metabolic Endotoxemia
2.1. Dysbiosis
2.2. Mechanisms of the Influx of LPS into the Bloodstream
2.3. Kinetics and Activity of LPS
3. Dietary Factors that Decrease Blood LPS Levels
|
Category |
Dietary Factor |
Dose |
Consumption Period |
Subject |
LPS |
LBP |
Gut Microbes with Significant Changes in Proportion ** |
|
|---|---|---|---|---|---|---|---|---|
|
Increase |
Decrease |
|||||||
|
Probiotics/ Prebiotics |
Yakult light (Lactobacillus casei Shirota 1 × 108 CFU/mL) [47] |
195 mL |
3 months |
Metabolic syndrome |
ND |
↑ |
— |
— |
|
Low-fat yogurt [48] |
339 g |
9 weeks |
Healthy subject or Obesity |
→ |
→ |
— |
— |
|
|
Low-fat yogurt [49] |
226 g |
Premeal |
Healthy subject or Obesity (postprandial endotoxemia was assessed) |
→ |
→ |
— |
— |
|
|
Oligofructose [50] |
21 g |
12 weeks |
Overweight/ Obesity |
↓ |
— |
— |
— |
|
|
Oligofructose- enriched inulin [51] |
10 g |
8 weeks |
Type 2 diabetes |
↓ |
— |
— |
— |
|
|
Inulin + Oligofructose [52] |
8 g 8 g |
3 months |
Obesity |
→ |
— |
Bifidobacterium, Faecalibacterium prausnitzii |
Bacteroides intestinalis, Bacteroides vulgatus, Propionibacterium |
|
|
Galacto- oligosaccharide [53] |
5.5 g |
12 weeks |
Type 2 diabetes |
→ |
→ |
none |
none |
|
|
Galacto- oligosaccharide [54] |
15 g |
12 weeks |
Overweight/ Obesity |
— |
→ |
Bifidobacterium spp. |
none |
|
|
α-Galacto- oligosaccharide [55] |
6–18 g |
14 days |
Overweight |
↓ |
— |
Bifidobacteria |
none |
|
|
Resistant dextrin [56] |
10 g |
8 weeks |
Type 2 diabetes |
↓ |
— |
— |
— |
|
|
Insoluble dietary fiber [from Fiber One Original cereal (General mills)] [57] |
30 g |
With high-fat, high-calorie meal |
Healthy subject (postprandial endotoxemia was assessed) |
↓* |
— |
— |
— |
|
|
Whole grains [58] |
3 servings |
6 weeks |
Overweight/ Obesity |
— |
↓ |
none |
none |
|
|
Probiotics/ Prebiotics |
Bifidobacterium longum + Oligofructose + Life style modification [59] |
— |
24 weeks |
Non-alcoholic steatohepatitis |
↓ |
— |
— |
— |
|
Polyphenol |
Resveratrol + Polyphenol [60] |
100 mg 75 mg |
10 minutes before intake of high-fat high-carbohydrate meal |
Healthy subjects (postprandial endotoxemia was assessed) |
— |
↓ * |
— |
— |
|
Red wine [61] |
272 mL |
With high-fat meal |
Healthy subjects (postprandial endotoxemia was assessed) |
→ |
→ |
— |
— |
|
|
Dietary habits |
Fruits + Vegetables [58] |
3 servings |
6 weeks |
Overweight/ Obesity |
— |
↓ |
α-diversity (No significant change in bacterial genera was found) |
none |
|
Caloric restriction [62] |
800 kcal |
4 weeks |
Obesity |
— |
↓ |
Anaerostipes hadrus, Blautia sp., Ruminococcus faecis, Bifidodbacterium sp. |
Agathobacter rectalis |
|
|
Others |
Glutamine [63] |
30 g |
14 weeks |
Overweight/ Obesity |
↓ |
— |
— |
— |
|
Subject |
Number of Subject |
Correlation of Dietary Factor and Gut Microbe * |
Correlation of Blood LPS and Gut Microbe |
Correlation of Blood LPS and Dietary Factor |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Over- weight pregnant women [64] |
88 |
P |
Dietary fiber vs. |
diversity, richness, Firmicutes in unidentified family of order Clostridiales, Barnciellaceae family belonging to the phylum Bacteroidetes |
P |
none |
P |
none |
||
|
Vitamin A, β-Carotene vs. |
Firmicutes |
|||||||||
|
N |
Fat vs. |
diversity, richness, Barnsiellaceae |
N |
none |
N |
none |
||||
|
Healthy subjects [65] |
150 |
N |
25-Hydroxy vitamin D vs. |
Coprococcus, Bifdobacterium |
N |
LPS vs. |
Faecalibacterium |
N |
LPS vs. |
25-Hydroxy vitamin D |
|
Type 1 diabetes [66] |
668 |
— |
— |
— |
— |
— |
— |
N |
LPS vs. |
Dietary pattern; “Fish”(frequently eat fish dishes), “Healthy snack” (frequently eat fruits, berries, fresh vegetable, yoghurt, low-fat cheese, and do not drink much soft drinks), “Modern”(frequently eat poultry, pasta, rice, meat dishes, fried and grilled foods, and fresh vegetables) |
|
Category |
Dietary Factor |
Dose |
Administration Period |
Model |
LPS |
LBP |
Significant Change in Gut Microbiota |
|---|---|---|---|---|---|---|---|
|
Probiotics/ Prebiotics |
Lactobacillus rhamnosus GG [67] |
1 × 108 CFU/day |
12 weeks |
HFD-fed ApoE KO mouse |
↓ |
— |
no |
|
Lactobacillus rhamnosus CNCM I-4036 [68] |
1 × 1010 CFU/day |
30 days |
Chow diet-fed Zucker-Lepfa/fa rat |
— |
↓ |
— |
|
|
Lactobacillus sakei OK67 +/− Lactobacillus sakei PK16 [69] |
1 × 109 CFU/day 1 × 109 CFU/day |
4 weeks |
HFD-fed C57BL/6 mouse |
↓ |
— |
yes |
|
|
Bifidobacterium longum BR-108 (sterilized) [70] |
200, 400 mg/kg/day |
4 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Bifidobacterium infantis + Lactobacillus acidophilus + Bacillus cereus [71] |
0.5 × 106 CFU/day 0.5 × 106 CFU/day 0.5 × 105 CFU/day |
12 weeks |
HFHSD-fed SD rat |
↓ |
— |
yes |
|
|
Lactobacillus plantarum LC27 +/− Bifidobacterium longum LC67 [43] |
1 × 109 CFU/day each (or 0.75 × 109 (LC27) + 0.25×109 (LC67) CFU/day in mix) |
4 weeks |
HFD-fed C57BL/6 mouse |
↓ |
— |
yes |
|
|
Oligofructose [72] |
10% (mixed in diet) |
12 weeks |
HFHSD-fed SD rat |
↓ |
— |
yes |
|
|
Galacto- oligosaccharide [73] |
800 mg/kg/day |
8 weeks |
HFD-fed SD rat |
↓ |
— |
yes |
|
|
Inulin [74] |
5% (intragastric administration, sample volume was not described) |
6 weeks |
standardized diet (kcal %: 10% fat, 20% protein, and 70% carbo- hydrate; 3.85 kcal g−1)-fed db/db mouse |
↓ |
— |
yes |
|
|
Wheat-derived arabinoxylan [75] |
7.5% (mixed in diet) |
8 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
— |
|
|
Polyphenols |
Grape seed proanthocyanidin [33] |
500 mg/kg/day |
10 days (prophylactic) or 17 weeks (with cafeteria diet) |
Cafeteria diet (high-fat/high carbohydrate diet)-fed Wistar rat |
↓ |
— |
— |
|
Grape-seed proanthocyanidin [29] |
100, 500 mg/kg/day |
2 weeks |
Cafeteria diet (high saturated-fat/high refined-carbohydrate diet)-fed Wistar rat |
↓ |
— |
— |
|
|
Resveratrol [76] |
50, 75, 100 mg/kg/day |
16 weeks |
HFD-fed C57BL/6 mouse |
↓ |
↓ |
yes |
|
|
Apple-derived polymeric procyanidins [77] |
0.5% (administration route was not described) |
20 weeks |
HFHSD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Genistein [78] |
0.2% (mixed in diet) |
6 months |
HFD-fed C57BL/6 mouse |
↓ |
— |
yes |
|
|
Isoflavone [79] |
0.1% (mixed in diet) |
5 weeks |
HFD-fed C57BL/6 mouse |
↓ |
↓ |
yes |
|
|
Syringaresinol [80] |
50 mg/kg/day |
10 weeks |
40-week-old C57BL/6 mouse |
— |
↓ |
yes |
|
|
Sulfated polysaccharide |
Sea cucumber-derived sulfated polysaccharide [81] |
300 mg/kg/day |
8 weeks |
HFD-fed BALB/c mouse |
— |
↓ |
yes |
|
Sea cucumber-derived sulfated polysaccharide [82] |
300 mg/kg/day |
42 days |
Chow-fed BALB/c mouse |
— |
↓ |
yes |
|
|
Acaudina molpadioides-derived fucosylated chondroitin sulfate [83] |
80 mg/kg/day |
10 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Chicken-derived chondroitin sulfate [84] |
150 mg/kg/day |
16 days |
Exhaustive exercise stress model BALB/c mouse |
↓ |
— |
yes |
|
|
Fucoidan [73] |
100 mg/kg/day |
8 weeks |
HFD-fed SD rat |
↓ |
— |
yes |
|
|
Other dietary components |
Tetrahydro iso-alpha acid (included in hops) [85] |
0.1% (mixed in diet) |
8 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
— |
|
Rhein (included in rhubarb) [86] |
120 mg/kg/day |
6 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Phlorizin (included in apple) [87] |
20 mg/kg/day |
10 weeks |
Chow-fed db/db mouse |
↓ |
— |
yes |
|
|
Capsaicin [88] |
0.01% (mixed in diet) |
12 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Rutin [89] |
0.64% (mixed in diet) |
20 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Lycopene [90] |
0.03% (mixed in diet) |
10 weeks |
HFD and fructose-fed C57BL/6 J mouse |
↓ |
— |
— |
|
|
Other extracts/dietary components |
Broccoli sprout extract [91] |
2.2% (mixed in diet) |
14 weeks |
HFD-fed C57BL/6JSlc mouse |
↓ |
↓ |
yes |
|
Camu camu extract [92] |
200 mg/kg/day |
8 weeks |
HFHSD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Other extracts/dietary components |
Cranberry extract [93] |
200 mg/kg/day |
8 weeks |
HFHSD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
Green tea extract [34] |
2% (mixed in diet) |
8 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
yes |
|
|
Tartary buckwheat protein [32] |
23.5% (mixed in diet) |
6 weeks |
HFD-fed C57BL/6 mouse |
↓ |
— |
yes |
|
|
Foods |
Cocoa [94] |
8% (mixed in diet) |
18 weeks |
HFD-fed C57BL/6J mouse |
↓ |
— |
— |
|
Nopal [95] |
5% of dietary fiber was replaced with those of nopal-derived (mixed in diet) |
1 month |
HFHSD-fed Wistar rat |
↓ |
— |
yes |
|
|
Steamed fish meat [96] |
Ad libitum (9:00–12:00 and 18:00–21:00) |
8 weeks |
Chow-fed C57BL/6 mouse |
— |
↓ |
yes |
|
|
Chinese medicines |
Geniposide + Chlorogenic acid [97] |
90 mg/kg/day 1.34 mg/kg/day |
4 weeks |
HFD-fed C57BL/6 mouse |
— |
↓ |
— |
|
Potentilla discolor Bunge water extract [98] |
400 mg/kg/day |
8 weeks |
HFD-fed, streptozotocin- injected C57BL/6J mouse |
↓ |
↓ |
yes |
|
|
Ganoderma lucidum mycelium water extract [99] |
2–8 mg/day |
8 weeks |
HFD-fed C57BL/6NCrlBltw mouse |
↓ |
— |
yes |
|
|
Semen hoveniae extract [100] |
300, 600 mg/kg/day |
8 weeks |
Alcohol-containing Lieber-DeCarli diet-fed SD rat (Alcoholic liver disorder model) |
↓ |
— |
yes |
|
|
Shenling Baizhu powder [101] |
30 g/kg/day |
16 weeks |
HFD-fed SD rat |
↓ |
— |
yes |
|
|
Caloric restriction |
30% caloric restriction [102] |
— |
62–141 weeks |
HFD, LFD-fed C57BL/6J mouse |
— |
↓ |
yes |
|
40% caloric restriction [103] |
— |
30 days |
Chow-fed C57BL/6J mouse |
↓ |
↓ |
yes |
—: No data, HFD: High-fat diet, HFHSD: High-fat high-sucrose diet, ↑: Significantly increased, →: Not significantly changed, ↓: Significantly decreased.
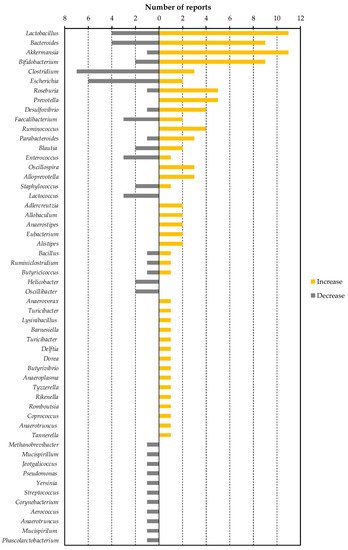
4. Association of Dietary Factor-Induced Reduction of Blood LPS and Modulation of Gut Microbiota
References
- Kiers, D.; Leijte, G.P.; Gerretsen, J.; Zwaag, J.; Kox, M.; Pickkers, P. Comparison of different lots of endotoxin and evaluation of in vivo potency over time in the experimental human endotoxemia model. Innate Immun. 2019, 25, 34–45.
- Benson, S.; Engler, H.; Wegner, A.; Rebernik, L.; Spreitzer, I.; Schedlowski, M.; Elsenbruch, S. What makes you feel sick after inflammation? Predictors of acute and persisting physical sickness symptoms induced by experimental endotoxemia. Clin. Pharmacol. Ther. 2017, 102, 141–151.
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772.
- Liang, H.; Hussey, S.E.; Sanchez-Avila, A.; Tantiwong, P.; Musi, N. Effect of lipopolysaccharide on inflammation and insulin action in human muscle. PLoS ONE 2013, 8, e63983.
- Pussinen, P.J.; Havulinna, A.S.; Lehto, M.; Sundvall, J.; Salomaa, V. Endotoxemia is associated with an increased risk of incident diabetes. Diabetes Care 2011, 34, 392–397.
- Jin, R.; Willment, A.; Patel, S.S.; Sun, X.; Song, M.; Mannery, Y.O.; Kosters, A.; McClain, C.J.; Vos, M.B. Fructose induced endotoxemia in pediatric nonalcoholic fatty liver disease. Int. J. Hepatol. 2014, 2014, 560620.
- Jandhyala, S.M.; Madhulika, A.; Deepika, G.; Rao, G.V.; Reddy, D.N.; Subramanyam, C.; Sasikala, M.; Talukdar, R. Altered intestinal microbiota in patients with chronic pancreatitis: Implications in diabetes and metabolic abnormalities. Sci. Rep. 2017, 7, 43640.
- Zhang, R.; Miller, R.G.; Gascon, R.; Champion, S.; Katz, J.; Lancero, M.; Narvaez, A.; Honrada, R.; Ruvalcaba, D.; McGrath, M.S. Circulating endotoxin and systemic immune activation in sporadic amyotrophic lateral sclerosis (sALS). J. Neuroimmunol. 2009, 206, 121–124.
- Kaliannan, K.; Hamarneh, S.R.; Economopoulos, K.P.; Nasrin Alam, S.; Moaven, O.; Patel, P.; Malo, N.S.; Ray, M.; Abtahi, S.M.; Muhammad, N.; et al. Intestinal alkaline phosphatase prevents metabolic syndrome in mice. Proc. Natl. Acad. Sci. USA 2013, 110, 7003–7008.
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481.
- Candido, T.L.N.; Bressan, J.; Alfenas, R.d.C.G. Dysbiosis and metabolic endotoxemia induced by high-fat diet. Nutr. Hosp. 2018, 35, 1432–1440.
- Devkota, S.; Wang, Y.; Musch, M.W.; Leone, V.; Fehlner-Peach, H.; Nadimpalli, A.; Antonopoulos, D.A.; Jabri, B.; Chang, E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 2012, 487, 104–108.
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563.
- Natividad, J.M.; Lamas, B.; Pham, H.P.; Michel, M.-L.; Rainteau, D.; Bridonneau, C.; da Costa, G.; van Hylckama Vlieg, J.; Sovran, B.; Chamignon, C.; et al. Bilophila wadsworthia aggravates high fat diet induced metabolic dysfunctions in mice. Nat. Commun. 2018, 9, 2802.
- Gehring, U.; Spithoven, J.; Schmid, S.; Bitter, S.; Braun-Fahrländer, C.; Dalphin, J.-C.; Hyvärinen, A.; Pekkanen, J.; Riedler, J.; Weiland, S.K.; et al. Endotoxin levels in cow’s milk samples from farming and non-farming families - the PASTURE study. Environ. Int. 2008, 34, 1132–1136.
- Lindenberg, F.C.B.; Ellekilde, M.; Thörn, A.C.; Kihl, P.; Larsen, C.S.; Hansen, C.H.F.; Metzdorff, S.B.; Aalbæk, B.; Hansen, A.K. Dietary LPS traces influences disease expression of the diet-induced obese mouse. Res. Vet. Sci. 2019, 123, 195–203.
- Gutsmann, T.; Howe, J.; Zähringer, U.; Garidel, P.; Schromm, A.B.; Koch, M.H.J.; Fujimoto, Y.; Fukase, K.; Moriyon, I.; Martínez-de-Tejada, G.; et al. Structural prerequisites for endotoxic activity in the limulus test as compared to cytokine production in mononuclear cells. Innate Immun. 2010, 16, 39–47.
- Bishop, R.E. Structural biology of membrane-intrinsic beta-barrel enzymes: Sentinels of the bacterial outer membrane. Biochim. Biophys. Acta 2008, 1778, 1881–1896.
- Van Den Berg, C.; de Neeling, A.J.; Schot, C.S.; Hustinx, W.N.M.; Wemer, J.; de Wildt, D.J. Delayed antibiotic-induced lysis of escherichia coli in vitro is correlated with enhancement of LPS release. Scand. J. Infect. Dis. 1992, 24, 619–627.
- Jin, Y.; Wu, Y.; Zeng, Z.; Jin, C.; Wu, S.; Wang, Y.; Fu, Z. From the cover: Exposure to oral antibiotics induces gut microbiota dysbiosis associated with lipid metabolism dysfunction and low-grade inflammation in mice. Toxicol. Sci. 2016, 154, 140–152.
- Radilla-Vázquez, R.B.; Parra-Rojas, I.; Martínez-Hernández, N.E.; Márquez-Sandoval, Y.F.; Illades-Aguiar, B.; Castro-Alarcón, N. Gut microbiota and metabolic endotoxemia in young obese mexican subjects. Obes. Facts 2016, 9, 1–11.
- Laugerette, F.; Vors, C.; Géloën, A.; Chauvin, M.; Soulage, C.; Lambert-Porcheron, S.; Peretti, N.; Alligier, M.; Burcelin, R.; Laville, M.; et al. Emulsified lipids increase endotoxemia: Possible role in early postprandial low-grade inflammation. J. Nutr. Biochem. 2011, 22, 53–59.
- Clemente-Postigo, M.; Queipo-Ortuño, M.I.; Murri, M.; Boto-Ordoñez, M.; Perez-Martinez, P.; Andres-Lacueva, C.; Cardona, F.; Tinahones, F.J. Endotoxin increase after fat overload is related to postprandial hypertriglyceridemia in morbidly obese patients. J. Lipid Res. 2012, 53, 973–978.
- Murakami, Y.; Tanabe, S.; Suzuki, T. High-fat diet-induced intestinal hyperpermeability is associated with increased bile acids in the large intestine of mice. J. Food Sci. 2016, 81, H216–H222.
- Ahmad, R.; Rah, B.; Bastola, D.; Dhawan, P.; Singh, A.B. Obesity-induces organ and tissue specific tight junction restructuring and barrier deregulation by claudin switching. Sci. Rep. 2017, 7, 5125.
- Guo, S.; Nighot, M.; Al-Sadi, R.; Alhmoud, T.; Nighot, P.; Ma, T.Y. Lipopolysaccharide regulation of intestinal tight junction permeability is mediated by TLR4 signal transduction pathway activation of FAK and MyD88. J. Immunol. 2015, 195, 4999–5010.
- Fang, W.; Xue, H.; Chen, X.; Chen, K.; Ling, W. Supplementation with sodium butyrate modulates the composition of the gut microbiota and ameliorates high-fat diet-induced obesity in mice. J. Nutr. 2019, 149, 747–754.
- Blanchard, C.; Moreau, F.; Chevalier, J.; Ayer, A.; Garcon, D.; Arnaud, L.; Pais de Barros, J.-P.; Gautier, T.; Neunlist, M.; Cariou, B.; et al. Sleeve gastrectomy alters intestinal permeability in diet-induced obese mice. Obes. Surg. 2017, 27, 2590–2598.
- González-Quilen, C.; Gil-Cardoso, K.; Ginés, I.; Beltrán-Debón, R.; Pinent, M.; Ardévol, A.; Terra, X.; Blay, M.T. Grape-Seed proanthocyanidins are able to reverse intestinal dysfunction and metabolic endotoxemia induced by a cafeteria diet in wistar rats. Nutrients 2019, 11, 979.
- Anitha, M.; Reichardt, F.; Tabatabavakili, S.; Nezami, B.G.; Chassaing, B.; Mwangi, S.; Vijay-Kumar, M.; Gewirtz, A.; Srinivasan, S. Intestinal dysbiosis contributes to the delayed gastrointestinal transit in high-fat diet fed mice. Cell. Mol. Gastroenterol. Hepatol. 2016, 2, 328–339.
- Reichardt, F.; Chassaing, B.; Nezami, B.G.; Li, G.; Tabatabavakili, S.; Mwangi, S.; Uppal, K.; Liang, B.; Vijay-Kumar, M.; Jones, D.; et al. Western diet induces colonic nitrergic myenteric neuropathy and dysmotility in mice via saturated fatty acid- and lipopolysaccharide-induced TLR4 signalling. J. Physiol. 2017, 595, 1831–1846.
- Zhou, X.-L.; Yan, B.; Xiao, Y.; Zhou, Y.; Liu, T. Tartary buckwheat protein prevented dyslipidemia in high-fat diet-fed mice associated with gut microbiota changes. Food Chem. Toxicol. 2018, 119, 296–301.
- Gil-Cardoso, K.; Ginés, I.; Pinent, M.; Ardévol, A.; Blay, M.; Terra, X. The co-administration of proanthocyanidins and an obesogenic diet prevents the increase in intestinal permeability and metabolic endotoxemia derived to the diet. J. Nutr. Biochem. 2018, 62, 35–42.
- Dey, P.; Sasaki, G.Y.; Wei, P.; Li, J.; Wang, L.; Zhu, J.; McTigue, D.; Yu, Z.; Bruno, R.S. Green tea extract prevents obesity in male mice by alleviating gut dysbiosis in association with improved intestinal barrier function that limits endotoxin translocation and adipose inflammation. J. Nutr. Biochem. 2019, 67, 78–89.
- Vreugdenhil, A.C.E.; Rousseau, C.H.; Hartung, T.; Greve, J.W.M.; van ’t Veer, C.; Buurman, W.A. Lipopolysaccharide (LPS)-binding protein mediates LPS detoxification by chylomicrons. J. Immunol. 2003, 170, 1399–1405.
- Vergès, B.; Duvillard, L.; Lagrost, L.; Vachoux, C.; Garret, C.; Bouyer, K.; Courtney, M.; Pomié, C.; Burcelin, R. Changes in lipoprotein kinetics associated with type 2 diabetes affect the distribution of lipopolysaccharides among lipoproteins. J. Clin. Endocrinol. Metab. 2014, 99, E1245–E1253.
- Levels, J.H.M.; Marquart, J.A.; Abraham, P.R.; van den Ende, A.E.; Molhuizen, H.O.F.; van Deventer, S.J.H.; Meijers, J.C.M. Lipopolysaccharide is transferred from high-density to low-density lipoproteins by lipopolysaccharide-binding protein and phospholipid transfer protein. Infect. Immun. 2005, 73, 2321–2326.
- Emancipator, K.; Csako, G.; Elin, R.J. In vitro inactivation of bacterial endotoxin by human lipoproteins and apolipoproteins. Infect. Immun. 1992, 60, 596–601.
- Correa, W.; Brandenburg, K.; Zähringer, U.; Ravuri, K.; Khan, T.; von Wintzingerode, F. Biophysical analysis of lipopolysaccharide formulations for an understanding of the low endotoxin recovery (LER) phenomenon. Int. J. Mol. Sci. 2017, 18, 2737.
- Yao, Z.; Mates, J.M.; Cheplowitz, A.M.; Hammer, L.P.; Maiseyeu, A.; Phillips, G.S.; Wewers, M.D.; Rajaram, M.V.S.; Robinson, J.M.; Anderson, C.L.; et al. Blood-Borne lipopolysaccharide is rapidly eliminated by liver sinusoidal endothelial cells via high-density lipoprotein. J. Immunol. 2016, 197, 2390–2399.
- van Bergenhenegouwen, J.; Kraneveld, A.D.; Rutten, L.; Garssen, J.; Vos, A.P.; Hartog, A. Lipoproteins attenuate TLR2 and TLR4 activation by bacteria and bacterial ligands with differences in affinity and kinetics. BMC Immunol. 2016, 17, 42.
- Wiesner, P.; Choi, S.; Almazan, F.; Benner, C.; Huang, W.; Diehl, C.J.; Gonen, A.; Butler, S.; Witztum, J.L.; Glass, C.K.; et al. Low doses of lipopolysaccharide and minimally oxidized low-density lipoprotein cooperatively activate macrophages via nuclear factor kappa B and activator protein-1: Possible mechanism for acceleration of atherosclerosis by subclinical endotoxemia. Circ. Res. 2010, 107, 56–65.
- Kim, H.I.; Kim, J.-K.; Kim, J.-Y.; Jang, S.E.; Han, M.J.; Kim, D.-H. Lactobacillus plantarum LC27 and Bifidobacterium longum LC67 simultaneously alleviate high-fat diet-induced colitis, endotoxemia, liver steatosis, and obesity in mice. Nutr. Res. 2019, 67, 78–89.
- Shao, B.; Munford, R.S.; Kitchens, R.; Varley, A.W. Hepatic uptake and deacylation of the LPS in bloodborne LPS-lipoprotein complexes. Innate Immun. 2012, 18, 825–833.
- Xie, G.; Wang, X.; Liu, P.; Wei, R.; Chen, W.; Rajani, C.; Hernandez, B.Y.; Alegado, R.; Dong, B.; Li, D.; et al. Distinctly altered gut microbiota in the progression of liver disease. Oncotarget 2016, 7, 19355–19366.
- Dandekar, A.; Qiu, Y.; Kim, H.; Wang, J.; Hou, X.; Zhang, X.; Zheng, Z.; Mendez, R.; Yu, F.-S.; Kumar, A.; et al. Toll-like receptor (TLR) signaling interacts with CREBH to modulate high-density lipoprotein (HDL) in response to bacterial endotoxin. J. Biol. Chem. 2016, 291, 23149–23158.
- Leber, B.; Tripolt, N.J.; Blattl, D.; Eder, M.; Wascher, T.C.; Pieber, T.R.; Stauber, R.; Sourij, H.; Oettl, K.; Stadlbauer, V. The influence of probiotic supplementation on gut permeability in patients with metabolic syndrome: An open label, randomized pilot study. Eur. J. Clin. Nutr. 2012, 66, 1110–1115.
- Pei, R.; DiMarco, D.M.; Putt, K.K.; Martin, D.A.; Gu, Q.; Chitchumroonchokchai, C.; White, H.M.; Scarlett, C.O.; Bruno, R.S.; Bolling, B.W. Low-fat yogurt consumption reduces biomarkers of chronic inflammation and inhibits markers of endotoxin exposure in healthy premenopausal women: A randomised controlled trial. Br. J. Nutr. 2017, 118, 1043–1051.
- Pei, R.; DiMarco, D.M.; Putt, K.K.; Martin, D.A.; Chitchumroonchokchai, C.; Bruno, R.S.; Bolling, B.W. Premeal low-fat yogurt consumption reduces postprandial inflammation and markers of endotoxin exposure in healthy premenopausal women in a randomized controlled trial. J. Nutr. 2018, 148, 910–916.
- Parnell, J.A.; Klancic, T.; Reimer, R.A. Oligofructose decreases serum lipopolysaccharide and plasminogen activator inhibitor-1 in adults with overweight/obesity. Obesity 2017, 25, 510–513.
- Dehghan, P.; Pourghassem Gargari, B.; Asghari Jafar-abadi, M. Oligofructose-enriched inulin improves some inflammatory markers and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized controlled clinical trial. Nutrition 2014, 30, 418–423.
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.B.; Neyrinck, A.M.; Bindels, L.B.; de Vos, W.M.; Gibson, G.R.; Thissen, J.; et al. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 2013, 62, 1112–1121.
- Pedersen, C.; Gallagher, E.; Horton, F.; Ellis, R.J.; Ijaz, U.Z.; Wu, H.; Jaiyeola, E.; Diribe, O.; Duparc, T.; Cani, P.D.; et al. Host-microbiome interactions in human type 2 diabetes following prebiotic fibre (galacto-oligosaccharide) intake. Br. J. Nutr. 2016, 116, 1869–1877.
- Canfora, E.E.; van der Beek, C.M.; Hermes, G.D.A.; Goossens, G.H.; Jocken, J.W.E.; Holst, J.J.; van Eijk, H.M.; Venema, K.; Smidt, H.; Zoetendal, E.G.; et al. Supplementation of diet with Galacto-oligosaccharides increases bifidobacteria, but not insulin sensitivity, in obese prediabetic individuals. Gastroenterology 2017, 153, 87–97.
- Morel, F.B.; Dai, Q.; Ni, J.; Thomas, D.; Parnet, P.; Fança-Berthon, P. α-Galacto-oligosaccharides dose-dependently reduce appetite and decrease inflammation in overweight adults. J. Nutr. 2015, 145, 2052–2059.
- Farhangi, M.A.; Javid, A.Z.; Sarmadi, B.; Karimi, P.; Dehghan, P. A randomized controlled trial on the efficacy of resistant dextrin, as functional food, in women with type 2 diabetes: Targeting the hypothalamic-pituitary-adrenal axis and immune system. Clin. Nutr. 2018, 37, 1216–1223.
- Ghanim, H.; Batra, M.; Abuaysheh, S.; Green, K.; Makdissi, A.; Kuhadiya, N.D.; Chaudhuri, A.; Dandona, P. Antiinflammatory and ROS suppressive effects of the addition of fiber to a high-fat high-calorie meal. J. Clin. Endocrinol. Metab. 2017, 102, 858–869.
- Kopf, J.C.; Suhr, M.J.; Clarke, J.; Eyun, S.; Riethoven, J.M.; Ramer-Tait, A.E.; Rose, D.J. Role of whole grains versus fruits and vegetables in reducing subclinical inflammation and promoting gastrointestinal health in individuals affected by overweight and obesity: A randomized controlled trial. Nutr. J. 2018, 17, 72.
- Malaguarnera, M.; Vacante, M.; Antic, T.; Giordano, M.; Chisari, G.; Acquaviva, R.; Mastrojeni, S.; Malaguarnera, G.; Mistretta, A.; Li Volti, G.; et al. Bifidobacterium longum with fructo-oligosaccharides in patients with non alcoholic steatohepatitis. Dig. Dis. Sci. 2012, 57, 545–553.
- Ghanim, H.; Sia, C.L.; Korzeniewski, K.; Lohano, T.; Abuaysheh, S.; Marumganti, A.; Chaudhuri, A.; Dandona, P. A resveratrol and polyphenol preparation suppresses oxidative and inflammatory stress response to a high-fat, high-carbohydrate meal. J. Clin. Endocrinol. Metab. 2011, 96, 1409–1414.
- Clemente-Postigo, M.; Queipo-Ortuño, M.I.; Boto-Ordoñez, M.; Coin-Aragüez, L.; Roca-Rodriguez, M.D.M.; Delgado-Lista, J.; Cardona, F.; Andres-Lacueva, C.; Tinahones, F.J. Effect of acute and chronic red wine consumption on lipopolysaccharide concentrations. Am. J. Clin. Nutr. 2013, 97, 1053–1061.
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Büttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 11955.
- Abboud, K.Y.; Reis, S.K.; Martelli, M.E.; Zordão, O.P.; Tannihão, F.; de Souza, A.Z.Z.; Assalin, H.B.; Guadagnini, D.; Rocha, G.Z.; Saad, M.J.A.; et al. Oral glutamine supplementation reduces obesity, pro-inflammatory markers, and improves insulin sensitivity in DIO wistar rats and reduces waist circumference in overweight and obese humans. Nutrients 2019, 11, 536.
- Röytiö, H.; Mokkala, K.; Vahlberg, T.; Laitinen, K. Dietary intake of fat and fibre according to reference values relates to higher gut microbiota richness in overweight pregnant women. Br. J. Nutr. 2017, 118, 343–352.
- Luthold, R.V.; Fernandes, G.R.; Franco-de-Moraes, A.C.; Folchetti, L.G.D.; Ferreira, S.R.G. Gut microbiota interactions with the immunomodulatory role of vitamin D in normal individuals. Metabolism. 2017, 69, 76–86.
- Ahola, A.J.; Lassenius, M.I.; Forsblom, C.; Harjutsalo, V.; Lehto, M.; Groop, P.-H. Dietary patterns reflecting healthy food choices are associated with lower serum LPS activity. Sci. Rep. 2017, 7, 6511.
- Chan, Y.K.; Brar, M.S.; Kirjavainen, P.V.; Chen, Y.; Peng, J.; Li, D.; Leung, F.C.; El-Nezami, H. High fat diet induced atherosclerosis is accompanied with low colonic bacterial diversity and altered abundances that correlates with plaque size, plasma A-FABP and cholesterol: A pilot study of high fat diet and its intervention with Lactobacillus rhamno. BMC Microbiol. 2016, 16, 264.
- Plaza-Díaz, J.; Robles-Sánchez, C.; Abadía-Molina, F.; Morón-Calvente, V.; Sáez-Lara, M.J.; Ruiz-Bravo, A.; Jiménez-Valera, M.; Gil, Á.; Gómez-Llorente, C.; Fontana, L. Adamdec1, Ednrb and Ptgs1/Cox1, inflammation genes upregulated in the intestinal mucosa of obese rats, are downregulated by three probiotic strains. Sci. Rep. 2017, 7, 1939.
- Jang, H.-M.; Han, S.-K.; Kim, J.-K.; Oh, S.-J.; Jang, H.-B.; Kim, D.-H. Lactobacillus sakei alleviates high-fat-diet-induced obesity and anxiety in mice by inducing AMPK activation and SIRT1 expression and inhibiting gut microbiota-mediated NF-κB activation. Mol. Nutr. Food Res. 2019, 63, e1800978.
- Kikuchi, K.; Ben Othman, M.; Sakamoto, K. Sterilized bifidobacteria suppressed fat accumulation and blood glucose level. Biochem. Biophys. Res. Commun. 2018, 501, 1041–1047.
- Xue, L.; He, J.; Gao, N.; Lu, X.; Li, M.; Wu, X.; Liu, Z.; Jin, Y.; Liu, J.; Xu, J.; et al. Probiotics may delay the progression of nonalcoholic fatty liver disease by restoring the gut microbiota structure and improving intestinal endotoxemia. Sci. Rep. 2017, 7, 45176.
- Rios, J.L.; Bomhof, M.R.; Reimer, R.A.; Hart, D.A.; Collins, K.H.; Herzog, W. Protective effect of prebiotic and exercise intervention on knee health in a rat model of diet-induced obesity. Sci. Rep. 2019, 9, 3893.
- Chen, Q.; Liu, M.; Zhang, P.; Fan, S.; Huang, J.; Yu, S.; Zhang, C.; Li, H. Fucoidan and galactooligosaccharides ameliorate high-fat diet-induced dyslipidemia in rats by modulating the gut microbiota and bile acid metabolism. Nutrition 2019, 65, 50–59.
- Li, K.; Zhang, L.; Xue, J.; Yang, X.; Dong, X.; Sha, L.; Lei, H.; Zhang, X.; Zhu, L.; Wang, Z.; et al. Dietary inulin alleviates diverse stages of type 2 diabetes mellitus via anti-inflammation and modulating gut microbiota in db/db mice. Food Funct. 2019, 10, 1915–1927.
- Neyrinck, A.M.; Van Hée, V.F.; Piront, N.; De Backer, F.; Toussaint, O.; Cani, P.D.; Delzenne, N.M. Wheat-derived arabinoxylan oligosaccharides with prebiotic effect increase satietogenic gut peptides and reduce metabolic endotoxemia in diet-induced obese mice. Nutr. Diabetes 2012, 2, e28.
- Campbell, C.L.; Yu, R.; Li, F.; Zhou, Q.; Chen, D.; Qi, C.; Yin, Y.; Sun, J. Modulation of fat metabolism and gut microbiota by resveratrol on high-fat diet-induced obese mice. Diabetes. Metab. Syndr. Obes. 2019, 12, 97–107.
- Masumoto, S.; Terao, A.; Yamamoto, Y.; Mukai, T.; Miura, T.; Shoji, T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016, 6, 31208.
- López, P.; Sánchez, M.; Perez-Cruz, C.; Velázquez-Villegas, L.A.; Syeda, T.; Aguilar-López, M.; Rocha-Viggiano, A.K.; Del Carmen Silva-Lucero, M.; Torre-Villalvazo, I.; Noriega, L.G.; et al. Long-term genistein consumption modifies gut microbiota, improving glucose metabolism, metabolic endotoxemia, and cognitive function in mice fed a high-fat diet. Mol. Nutr. Food Res. 2018, 62, e1800313.
- Kaliannan, K.; Robertson, R.C.; Murphy, K.; Stanton, C.; Kang, C.; Wang, B.; Hao, L.; Bhan, A.K.; Kang, J.X. Estrogen-mediated gut microbiome alterations influence sexual dimorphism in metabolic syndrome in mice. Microbiome 2018, 6, 205.
- Cho, S.-Y.; Kim, J.; Lee, J.H.; Sim, J.H.; Cho, D.; Bae, I.; Lee, H.; Seol, M.A.; Shin, H.M.; Kim, T.-J.; et al. Modulation of gut microbiota and delayed immunosenescence as a result of syringaresinol consumption in middle-aged mice. Sci. Rep. 2016, 6, 39026.
- Zhu, Z.; Zhu, B.; Sun, Y.; Ai, C.; Wang, L.; Wen, C.; Yang, J.; Song, S.; Liu, X. Sulfated polysaccharide from sea cucumber and its depolymerized derivative prevent obesity in association with modification of gut microbiota in high-fat diet-fed mice. Mol. Nutr. Food Res. 2018, 62, e1800446.
- Zhu, Z.; Zhu, B.; Sun, Y.; Ai, C.; Wu, S.; Wang, L.; Song, S.; Liu, X. Sulfated polysaccharide from sea cucumber modulates the gut microbiota and its metabolites in normal mice. Int. J. Biol. Macromol. 2018, 120, 502–512.
- Hu, S.; Wang, J.; Xu, Y.; Yang, H.; Wang, J.; Xue, C.; Yan, X.; Su, L. Anti-inflammation effects of fucosylated chondroitin sulphate from acaudina molpadioides by altering gut microbiota in obese mice. Food Funct. 2019, 10, 1736–1746.
- Liu, F.; Zhang, N.; Li, Z.; Wang, X.; Shi, H.; Xue, C.; Li, R.W.; Tang, Q. Chondroitin sulfate disaccharides modified the structure and function of the murine gut microbiome under healthy and stressed conditions. Sci. Rep. 2017, 7, 6783.
- Everard, A.; Geurts, L.; Van Roye, M.; Delzenne, N.M.; Cani, P.D. Tetrahydro iso-alpha acids from hops improve glucose homeostasis and reduce body weight gain and metabolic endotoxemia in high-fat diet-fed mice. PLoS ONE 2012, 7, e33858.
- Wang, S.; Huang, X.; Zhang, P.; Wang, H.; Zhang, Q.; Yu, S.; Yu, Y. Chronic rhein treatment improves recognition memory in high-fat diet-induced obese male mice. J. Nutr. Biochem. 2016, 36, 42–50.
- Mei, X.; Zhang, X.; Wang, Z.; Gao, Z.; Liu, G.; Hu, H.; Zou, L.; Li, X. Insulin sensitivity-enhancing activity of phlorizin Is associated with lipopolysaccharide decrease and gut microbiota changes in obese and type 2 diabetes (db/db) mice. J. Agric. Food Chem. 2016, 64, 7502–7511.
- Kang, C.; Wang, B.; Kaliannan, K.; Wang, X.; Lang, H.; Hui, S.; Huang, L.; Zhang, Y.; Zhou, M.; Chen, M.; et al. Gut microbiota mediates the protective effects of dietary capsaicin against chronic low-grade Inflammation and associated obesity induced by high-fat diet. MBio 2017, 8, 1–14.
- Guo, X.; Tang, R.; Yang, S.; Lu, Y.; Luo, J.; Liu, Z. Rutin and its combination with Inulin attenuate gut dysbiosis, the inflammatory status and endoplasmic reticulum stress in paneth cells of obese mice induced by high-fat diet. Front. Microbiol. 2018, 9, 2651.
- Wang, J.; Wang, Z.; Li, B.; Qiang, Y.; Yuan, T.; Tan, X.; Wang, Z.; Liu, Z.; Liu, X. Lycopene attenuates western-diet-induced cognitive deficits via improving glycolipid metabolism dysfunction and inflammatory responses in gut-liver-brain axis. Int. J. Obes. 2018.
- Nagata, N.; Xu, L.; Kohno, S.; Ushida, Y.; Aoki, Y.; Umeda, R.; Fuke, N.; Zhuge, F.; Ni, Y.; Nagashimada, M.; et al. Glucoraphanin ameliorates obesity and insulin resistance through adipose tissue browning and reduction of metabolic endotoxemia in mice. Diabetes 2017, 66, 1222–1236.
- Anhê, F.F.; Nachbar, R.T.; Varin, T.V.; Trottier, J.; Dudonné, S.; Le Barz, M.; Feutry, P.; Pilon, G.; Barbier, O.; Desjardins, Y.; et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut 2018, 1–12.
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883.
- Gu, Y.; Yu, S.; Park, J.Y.; Harvatine, K.; Lambert, J.D. Dietary cocoa reduces metabolic endotoxemia and adipose tissue inflammation in high-fat fed mice. J. Nutr. Biochem. 2014, 25, 439–445.
- Sánchez-Tapia, M.; Aguilar-López, M.; Pérez-Cruz, C.; Pichardo-Ontiveros, E.; Wang, M.; Donovan, S.M.; Tovar, A.R.; Torres, N. Nopal (Opuntia ficus indica) protects from metabolic endotoxemia by modifying gut microbiota in obese rats fed high fat/sucrose diet. Sci. Rep. 2017, 7, 4716.
- Zhang, Z.; Li, D.; Tang, R. Changes in mouse gut microbial community in response to the different types of commonly consumed meat. Microorganisms 2019, 7, 76.
- Peng, J.; Leng, J.; Tian, H.; Yang, T.; Fang, Y.; Feng, Q.; Zhao, Y.; Hu, Y.-Y. Geniposide and chlorogenic acid combination ameliorates non-alcoholic steatohepatitis involving the protection on the gut barrier function in mouse induced by high-fat diet. Front. Pharmacol. 2018, 9, 1399.
- Han, L.; Li, T.; Du, M.; Chang, R.; Zhan, B.; Mao, X. Beneficial effects of potentilla discolor bunge water extract on inflammatory cytokines release and gut microbiota in high-fat diet and streptozotocin-induced type 2 diabetic mice. Nutrients 2019, 11, 670.
- Chang, C.; Lin, C.; Lu, C.; Martel, J.; Ko, Y.; Ojcius, D.M.; Tseng, S.; Wu, T.; Chen, Y.M.; Young, J.D.; et al. Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat. Commun. 2015, 6, 7489.
- Qiu, P.; Dong, Y.; Zhu, T.; Luo, Y.-Y.; Kang, X.-J.; Pang, M.-X.; Li, H.-Z.; Xu, H.; Gu, C.; Pan, S.-H.; et al. Semen hoveniae extract ameliorates alcohol-induced chronic liver damage in rats via modulation of the abnormalities of gut-liver axis. Phytomedicine 2019, 52, 40–50.
- Zhang, Y.; Tang, K.; Deng, Y.; Chen, R.; Liang, S.; Xie, H.; He, Y.; Chen, Y.; Yang, Q. Effects of shenling baizhu powder herbal formula on intestinal microbiota in high-fat diet-induced NAFLD rats. Biomed. Pharmacother. 2018, 102, 1025–1036.
- Zhang, C.; Li, S.; Yang, L.; Huang, P.; Li, W.; Wang, S.; Zhao, G.; Zhang, M.; Pang, X.; Yan, Z.; et al. Structural modulation of gut microbiota in life-long calorie-restricted mice. Nat. Commun. 2013, 4, 2163.
- Fabbiano, S.; Suárez-Zamorano, N.; Chevalier, C.; Lazarević, V.; Kieser, S.; Rigo, D.; Leo, S.; Veyrat-Durebex, C.; Gaïa, N.; Maresca, M.; et al. Functional gut microbiota remodeling contributes to the caloric restriction-induced metabolic improvements. Cell Metab. 2018, 28, 907–921.
- Bergmann, K.R.; Liu, S.X.L.; Tian, R.; Kushnir, A.; Turner, J.R.; Li, H.-L.; Chou, P.M.; Weber, C.R.; De Plaen, I.G. Bifidobacteria stabilize claudins at tight junctions and prevent intestinal barrier dysfunction in mouse necrotizing enterocolitis. Am. J. Pathol. 2013, 182, 1595–1606.
- Ewaschuk, J.B.; Diaz, H.; Meddings, L.; Diederichs, B.; Dmytrash, A.; Backer, J.; Looijer-van Langen, M.; Madsen, K.L. Secreted bioactive factors from Bifidobacterium infantis enhance epithelial cell barrier function. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G1025–G1034.
- Wang, H.; Zhang, W.; Zuo, L.; Zhu, W.; Wang, B.; Li, Q.; Li, J. Bifidobacteria may be beneficial to intestinal microbiota and reduction of bacterial translocation in mice following ischaemia and reperfusion injury. Br. J. Nutr. 2013, 109, 1990–1998.
- Feng, Y.; Wang, Y.; Wang, P.; Huang, Y.; Wang, F. Short-Chain fatty acids manifest stimulative and protective effects on intestinal barrier function through the inhibition of NLRP3 inflammasome and autophagy. Cell. Physiol. Biochem. 2018, 49, 190–205.




