
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sung Keun Jung | + 3142 word(s) | 3142 | 2021-08-12 05:25:40 | | | |
| 2 | Vivi Li | Meta information modification | 3142 | 2021-08-24 08:39:20 | | | | |
| 3 | Catherine Yang | Meta information modification | 3142 | 2021-09-30 04:06:24 | | |
Video Upload Options
Here, we compared the chemical properties and antioxidant effects of black pepper (Piper nigrum L.) and pink pepper (Schinus molle L.). Additionally, the antioxidant and anti-inflammatory capacities of pink pepper were measured to determine nutraceutical potential. Pink peppers from Brazil (PPB), India (PPI), and Sri Lanka (PPS) had higher Hunter a* (redness) values and lower L* (lightness) and b* (yellowness) values than black pepper from Vietnam (BPV). Fructose and glucose were detected in PPB, PPI, and PPS, but not in BPV. PPB, PPI, and PPS had greater 2,2-diphenyl-1-picrylhydrazyl and 3-ethylbenzothiazoline-6-sulphonic acid radical scavenging stabilities and higher total phenolic contents than BPV. BPV had higher levels of piperine than the pink peppers. Gallic acid, protocatechuic acid, epicatechin, and p-coumaric acid were detected only in the three pink peppers. PPB significantly suppressed lipopolysaccharide-induced reactive oxygen species production with increased Nrf2 translocation from cytosol to nucleus and heme oxygenase-1 expression. PPB and PPS significantly suppressed lipopolysaccharide-induced nitrite production and nitric oxide synthase expression by suppressing phosphorylation of p38 without affecting cell viability. Additionally, PPB and PPS significantly suppressed ultraviolet B-induced cyclooxygenase-2 expression by affecting the phosphorylation of ERK1/2 without cell cytotoxicity. These results suggest that pink pepper is a potential nutraceutical against oxidative and inflammatory stress.
1. Introduction
2. Results and Discussion
2.1. Color and Sugar Content Analysis

| Region | L | a | b | Fructose (mg/100 g) |
Glucose (mg/100 g) |
|
|---|---|---|---|---|---|---|
| Pink | Brazil | 28.87 ± 0.01 b | 11.94 ± 0.01 a | 11.41 ± 0.004 b | 11,507.21 ± 90.5 b | 9816.07 ± 36.51 a |
| India | 25.02 ± 0.09 d | 6.71 ± 0.05 b | 8.55 ± 0.04 d | 9528.74 ± 46.67 c | 6181.37 ± 315.61 b | |
| Sri Lanka | 27.36 ± 0.03 c | 11.92 ± 0.06 a | 10.89 ± 0.05 c | 11,829.82 ± 23.73 a | 9758.15 ± 330.28 a | |
| Black | Vietnam | 43.65 ± 0.01 a | 3.81 ± 0.02 c | 13.16 ± 0.01 a | N.D. | N.D. |
2.2. Total Flavonoid Contents and Total Phenolic Contents
| Region | Total Phenolic Content (mg GAE/100 g) |
Total Flavonoid Content (mg CE/100 g) |
|
|---|---|---|---|
| Pink | Brazil | 1607.80 ± 21.11 a | 266.67 ± 2.42 b |
| India | 1588.29 ± 17.88 a | 230.30 ± 2.10 d | |
| Sri Lanka | 1250.08 ± 10.75 b | 248.89 ± 5.60 c | |
| Black | Vietnam | 794.47 ± 17.19 c | 344.24 ± 3.78 |
2.3. Quantification of Major Compounds in Peppers by HPLC
| Region | DPPH (mg VCE/100 g) |
ABTS (mg VCE/100 g) |
|
|---|---|---|---|
| Pink | Brazil | 4015.32 ± 13.00 a | 2741.25 ± 19.69 a |
| India | 4081.92 ± 34.39 a | 2845.12 ± 3.91 a | |
| Sri Lanka | 2812.30 ± 10.81 b | 1956.96 ± 54.26 b | |
| Black | Vietnam | 271.45 ± 12.01 | 861.92 ± 83.23 |
| Region | Piperine (mg/100 g) |
Gallic Acid (mg/100 g) |
Protocatechuic Acid (mg/100 g) |
Epicatechin (mg/100 g) |
p-Coumaric Acid (mg/100 g) |
|
|---|---|---|---|---|---|---|
| Pink | Brazil | 134.60 ± 3.20 b | 526.72 ± 6.06 b | 144.85 ± 0.71 b | 85.91 ± 2.88 a | 115.92 ± 5.00 b |
| India | 101.10 ± 2.84 c | 657.59 ± 5.25 a | 237.52 ± 0.64 a | 89.24 ± 2.04 a | 151.33 ± 7.07 a | |
| Sri Lanka | 120.67 ± 1.91 bc | 168.15 ± 1.43 c | 29.47 ± 0.18 c | 38.26 ± 1.28 b | 48.24 ± 1.28 c | |
| Black | Vietnam | 4097.53 ± 46.87 a | N.D | N.D | N.D | N.D |
2.4. Radical Scavenging Effects
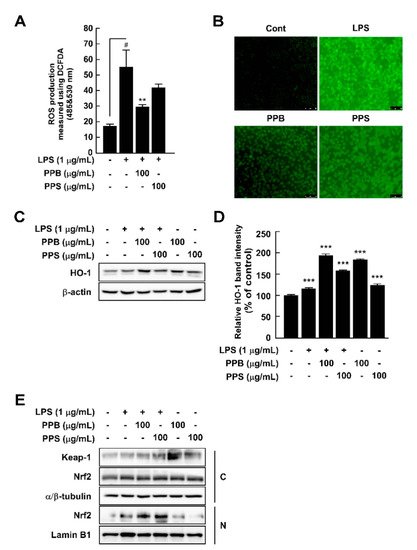
2.5. Pink Peppers Inhibited LPS-Induced ROS Production and Increased HO-1 Expression in RAW264.7 Cells
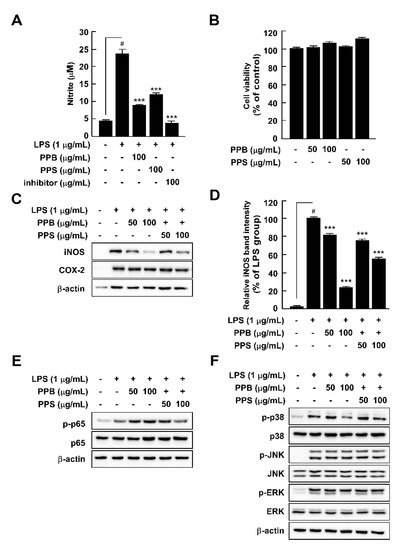
2.6. Effects of PPE on LPS-Induced Nitrite Production, iNOS and COX-2 Expression, and p65 and MAPK Phosphorylation in RAW264.7 Cells
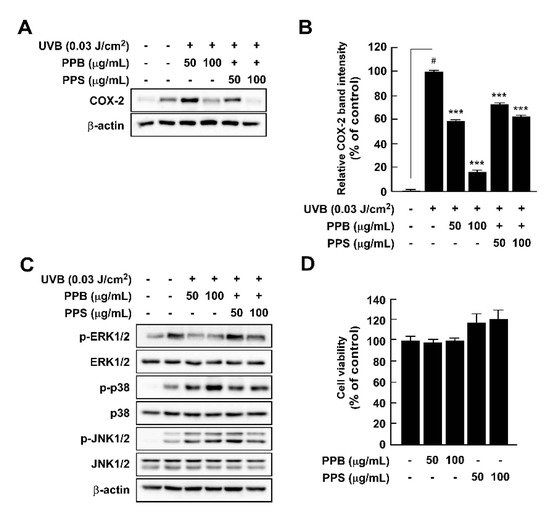
2.7. Effects of PPE on UVB-Induced COX-2 Expression and MAPK Phosphorylation in HaCaT Cells
References
- Wu, A.; Tymoszuk, P.; Haschka, D.; Heeke, S.; Dichtl, S.; Petzer, V.; Seifert, M.; Hilbe, R.; Sopper, S.; Talasz, H.; et al. Salmonella Utilizes Zinc To Subvert Antimicrobial Host Defense of Macrophages via Modulation of NF-kappaB Signaling. Infect. Immun. 2017, 85.
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 2014, 20, 1126–1167.
- Droge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95.
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84.
- Araujo, J.A.; Zhang, M.; Yin, F. Heme oxygenase-1, oxidation, inflammation, and atherosclerosis. Front. Pharmacol. 2012, 3, 119.
- Kim, E.N.; Kim, G.R.; Yu, J.S.; Kim, K.H.; Jeong, G.S. Inhibitory Effect of (2R)-4-(4-hydroxyphenyl)-2-butanol 2-O-beta-d-apiofuranosyl-(1-->6)-beta-d-glucopyranoside on RANKL-Induced Osteoclast Differentiation and ROS Generation in Macrophages. Int. J. Mol. Sci. 2020, 22, 222.
- Song, K.M.; Ha, S.J.; Lee, J.E.; Kim, S.H.; Kim, Y.H.; Kim, Y.; Hong, S.P.; Jung, S.K.; Lee, N.H. High yield ultrasonication extraction method for Undaria pinnatifida sporophyll and its anti-inflammatory properties associated with AP-1 pathway suppression. LWT-Food Sci. Technol. 2015, 64, 1315–1322.
- Jung, S.K.; Ha, S.J.; Jung, C.H.; Kim, Y.T.; Lee, H.K.; Kim, M.O.; Lee, M.H.; Mottamal, M.; Bode, A.M.; Lee, K.W.; et al. Naringenin targets ERK2 and suppresses UVB-induced photoaging. J. Cell Mol. Med. 2016, 20, 909–919.
- Ha, S.J.; Lee, J.; Park, J.; Kim, Y.H.; Lee, N.H.; Kim, Y.E.; Song, K.M.; Chang, P.S.; Jeong, C.H.; Jung, S.K. Syringic acid prevents skin carcinogenesis via regulation of NoX and EGFR signaling. Biochem. Pharmacol. 2018, 154, 435–445.
- Garzoli, S.; Laghezza Masci, V.; Turchetti, G.; Pesci, L.; Tiezzi, A.; Ovidi, E. Chemical investigations of male and female leaf extracts from Schinus molle L. Nat. Prod. Res. 2019, 33, 1980–1983.
- Too, J.J.Y.; Shek, L.P.; Rajakulendran, M. Cross-reactivity of pink peppercorn in cashew and pistachio allergic individuals. Asia Pac. Allergy 2019, 9, e25.
- Giuffrida, D.; Martínez, N.; Arrieta-Garay, Y.; Fariña, L.; Boido, E.; Dellacassa, E. Valorisation of Schinus molle fruit as a source of volatile compounds in foods as flavours and fragrances. Food Res. Int. 2020, 133, 109103.
- Do Prado, A.C.; Garces, H.G.; Bagagli, E.; Rall, V.L.M.; Furlanetto, A.; Fernandes, A.; Furtado, F.B. Schinus molle essential oil as a potential source of bioactive compounds: Antifungal and antibacterial properties. J. Appl. Microbiol. 2019, 126, 516–522.
- Guerra-Boone, L.; Alvarez-Roman, R.; Salazar-Aranda, R.; Torres-Cirio, A.; Rivas-Galindo, V.M.; de Torres, N.W.; Gonzalez, G.M.G.; Perez-Lopez, L.A. Chemical Compositions and Antimicrobial and Antioxidant Activities of the Essential Oils from Magnolia grandiflora, Chrysactinia mexicana, and Schinus molle Found in Northeast Mexico. Nat. Prod. Commun. 2013, 8, 135–138.
- Feriani, A.; Tir, M.; Hamed, M.; Sila, A.; Nahdi, S.; Alwasel, S.; Harrath, A.H.; Tlili, N. Multidirectional insights on polysaccharides from Schinus terebinthifolius and Schinus molle fruits: Physicochemical and functional profiles, in vitro antioxidant, anti-genotoxicity, antidiabetic, and antihemolytic capacities, and in vivo anti-inflammatory and anti-nociceptive properties. Int. J. Biol. Macromol. 2020, 165, 2576–2587.
- Zhang, C.; Zhao, J.; Famous, E.; Pan, S.; Peng, X.; Tian, J. Antioxidant, hepatoprotective and antifungal activities of black pepper (Piper nigrum L.) essential oil. Food Chem. 2021, 346, 128845.
- Hwang, Y.P.; Yun, H.J.; Kim, H.G.; Han, E.H.; Choi, J.H.; Chung, Y.C.; Jeong, H.G. Suppression of phorbol-12-myristate-13-acetate-induced tumor cell invasion by piperine via the inhibition of PKCalpha/ERK1/2-dependent matrix metalloproteinase-9 expression. Toxicol. Lett. 2011, 203, 9–19.
- Yang, H.; Kim, Y.J.; Shin, Y. Influence of Ripening Stage and Cultivar on Physicochemical Properties and Antioxidant Compositions of Aronia Grown in South Korea. Foods 2019, 8, 598.
- Solis, J.L.; Davila, R.; Sandoval, C.; Guzmán, D.; Guzmán, H.; Alejo, L.; Kiros, Y. Ethanol Production from Schinus molle Essential Oil Extraction Residues. Waste Biomass Valorization 2020, 11, 4053–4065.
- Kim, D.W.; Kim, M.J.; Shin, Y.; Jung, S.K.; Kim, Y.J. Green Pepper (Piper nigrum L.) Extract Suppresses Oxidative Stress and LPS-Induced Inflammation via Regulation of JNK Signaling Pathways. Appl. Sci. 2020, 10, 2519.
- Li, S.; Lei, Y.; Jia, Y.; Li, N.; Wink, M.; Ma, Y. Piperine, a piperidine alkaloid from Piper nigrum re-sensitizes P-gp, MRP1 and BCRP dependent multidrug resistant cancer cells. Phytomedicine 2011, 19, 83–87.
- Feuereisen, M.M.; Zimmermann, B.F.; Schulze-Kaysers, N.; Schieber, A. Differentiation of Brazilian Peppertree (Schinus terebinthifolius Raddi) and Peruvian Peppertree (Schinus molle L.) Fruits by UHPLC-UV-MS Analysis of Their Anthocyanin and Biflavonoid Profiles. J. Agric. Food Chem. 2017, 65, 5330–5338.
- Parra, C.; Muñoz, P.; Bustos, L.; Parra, F.; Simirgiotis, M.J.; Escobar, H. UHPLC-DAD Characterization of Origanum vulgare L. from Atacama Desert Andean Region and Antioxidant, Antibacterial and Enzyme Inhibition Activities. Molecules 2021, 269, 2100.
- Zhang, G.; Chen, S.; Zhou, W.; Meng, J.; Deng, K.; Zhou, H.; Hu, N.; Suo, Y. Rapid qualitative and quantitative analyses of eighteen phenolic compounds from Lycium ruthenicum Murray by UPLC-Q-Orbitrap MS and their antioxidant activity. Food Chem. 2018, 269, 150–156.
- Kefayati, Z.; Motamed, S.M.; Shojaii, A.; Noori, M.; Ghods, R. Antioxidant Activity and Phenolic and Flavonoid Contents of the Extract and Subfractions of Euphorbia splendida Mobayen. Pharmacogn. Res. 2017, 9, 362–365.
- Hwang, J.H.; Oh, Y.S.; Lim, S.B. Anti-inflammatory Activities of Some Brown Marine Algae in LPS-Stimulated RAW 264.7 Cells. Food Sci. Biotechnol. 2014, 23, 865–871.
- Nishikawa, S.; Inoue, Y.; Hori, Y.; Miyajima, C.; Morishita, D.; Ohoka, N.; Hida, S.; Makino, T.; Hayashi, H. Anti-Inflammatory Activity of Kurarinone Involves Induction of HO-1 via the KEAP1/Nrf2 Pathway. Antioxidants 2020, 9, 842.
- Ranilla, L.G.; Kwon, Y.I.; Apostolidis, E.; Shetty, K. Phenolic compounds, antioxidant activity and in vitro inhibitory potential against key enzymes relevant for hyperglycemia and hypertension of commonly used medicinal plants, herbs and spices in Latin America. Bioresour. Technol. 2010, 101, 4676–4689.
- Reis, J.; Guan, X.Q.; Kisselev, A.F.; Papasian, C.J.; Qureshi, A.A.; Morrison, D.C.; Van Way, C.W., 3rd; Vogel, S.N.; Qureshi, N. LPS-induced formation of immunoproteasomes: TNF-alpha and nitric oxide production are regulated by altered composition of proteasome-active sites. Cell Biochem. Biophys. 2011, 60, 77–88.
- Gu, I.; Brownmiller, C.; Stebbins, N.B.; Mauromoustakos, A.; Howard, L.; Lee, S.O. Berry Phenolic and Volatile Extracts Inhibit Pro-Inflammatory Cytokine Secretion in LPS-Stimulated RAW264.7 Cells through Suppression of NF-kappaB Signaling Pathway. Antioxidants 2020, 9, 871.
- Kim, M.J.; Kim, J.G.; Sydara, K.M.; Lee, S.W.; Jung, S.K. Croton hirtus L’Her Extract Prevents Inflammation in RAW264.7 Macrophages Via Inhibition of NF-kappaB Signaling Pathway. J. Microbiol. Biotechnol. 2020, 30, 490–496.
- Jung, S.K.; Lee, K.W.; Byun, S.; Kang, N.J.; Lim, S.H.; Heo, Y.S.; Bode, A.M.; Bowden, G.T.; Lee, H.J.; Dong, Z. Myricetin suppresses UVB-induced skin cancer by targeting Fyn. Cancer Res. 2008, 68, 6021–6029.
- Ha, S.J.; Park, J.; Lee, J.; Song, K.M.; Um, M.Y.; Cho, S.; Jung, S.K. Rice bran supplement prevents UVB-induced skin photoaging in vivo. Biosci. Biotechnol. Biochem. 2018, 82, 320–328.
- So, B.R.; Yeo, H.J.; Lee, J.J.; Jung, Y.H.; Jung, S.K. Cellulose nanocrystal preparation from Gelidium amansii and analysis of its anti-inflammatory effect on the skin in vitro and in vivo. Carbohydr. Polym. 2021, 254, 117315.
- Choi, B.Y.; Choi, H.S.; Ko, K.; Cho, Y.Y.; Zhu, F.; Kang, B.S.; Ermakova, S.P.; Ma, W.Y.; Bode, A.M.; Dong, Z.G. The tumor suppressor p16(INK4a) prevents cell transformation through inhibition of c-Jun phosphorylation and AP-1 activity. Nat. Struct. Mol. Biol. 2005, 12, 699–707.




