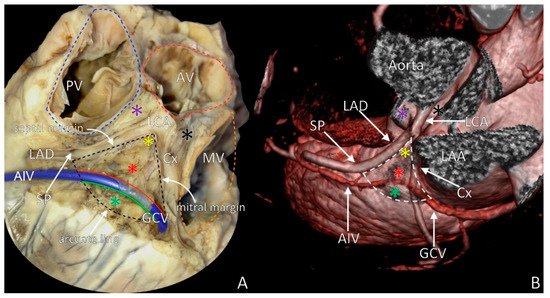
The left ventricular summit (LVS) is a triangular area located at the most superior portion of the left epicardial ventricular region, surrounded by the two branches of the left coronary artery: the left anterior interventricular artery and the left circumflex artery. The triangle is bounded by the apex, septal and mitral margins and base.
1. Introduction
Despite the intensive research in the anatomical sciences for the past couple of centuries, some structures within the body remain incompletely described
[1][2][3]. Moreover, recent developments in invasive cardiology and electrophysiology have renewed interest in detailed cardiac anatomy
[4][5]. One of the structures that are currently of special interest is the left ventricular summit (LVS). The LVS is defined as a triangular epicardial area located at the bifurcation of the left coronary artery. Recent studies are reemphasizing the importance of LVS as the source of ventricular arrhythmias with a high level of difficulty for treatment with radiofrequency ablation
[6]. There are various approaches for reaching LVS arrhythmias.
Nevertheless, the proximity of the surrounding major cardiac structures might pose a risk of complications, and therefore, ablation within the LVS region may be challenging
[7]. Important anatomical structures, such as surrounding coronary arteries, epicardial fat and fibrotic components, may complicate the approach
[8].
2. LVS Definition
The LVS is a triangular area located at the most superior portion of the left epicardial ventricular region, surrounded by the two branches of the left coronary artery: the left anterior interventricular artery and the left circumflex artery
[9]. The triangle is bounded by septal and mitral margins (
Figure 1). A delineation by an arched line, with the radius of this arc being the distance from the left coronary bifurcation artery to the first prominent left coronary artery septal perforator, represents the most inferior boundary of this arc region (triangle base). Inside this triangular area, the anterior interventricular cardiac vein (that travels through the anterior part of the atrioventricular groove) becomes the great cardiac vein that is further heading to the posterior part of the atrioventricular groove
[10][11][12][13][14]. Thus, the great cardiac vein/anterior interventricular cardiac vein bisects the LVS into two distinct areas: superior area (also named the inaccessible area for radiofrequency ablation because of the significant risk of coronary vasculature damage) and inferior area (the accessible area for radiofrequency ablation, where the interventions are relatively safe) (
Figure 1A)
[7]. Each part of the LVS has a specific relation to the adjacent anatomical structures, and each will be described below (
Figure 1).
Figure 1. The left ventricular summit (LVS) with marked boundaries. (A) The LVS in heart cadaveric specimen; (B) LVS in medical imaging visualization (angio-computed tomography rendered image). Red asterisk = inaccessible area, green asterisk = accessible area, violet asterisk = septal summit, black asterisk = aortic–mitral continuity, yellow asterisk = apex of LVS, AIV = anterior interventricular vein, AV = aortic valve, Cx = circumflex branch of left coronary artery, GCV = great cardiac vein, LAA = left atrial appendage, LAD = left anterior descending artery, LCA = left coronary artery, MV = mitral valve, PT = pulmonary trunk, PV = pulmonary valve, SP = septal perforator.
3. LVS Apex
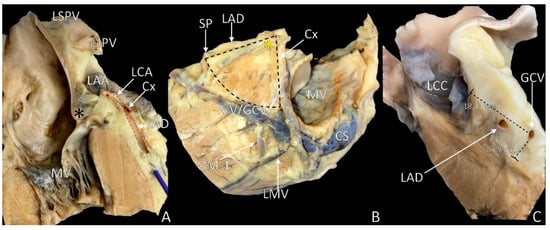
The apex of the LVS is located superiorly towards the left coronary arterial ostium. It begins at the left coronary artery’s bifurcation to the left circumflex coronary artery and left anterior descending artery. The nearest neighboring structure to the LVS apex is the aortic root, covered by a large fibrous structure—the aorto-ventricular membrane
[9] (
Figure 2A). The aortic root is the continuation of the left ventricle outflow tract. It occupies a central position within the heart, located to the right and posteriorly, relative to the subpulmonary infundibulum
[7]. The apex into the deep of the myocardium correlates with the left aortic sinus of Valsalva, septal summit to the right and aortic–mitral continuity to the left (
Figure 1). The epicardial adipose tissue and pulmonary trunk cover the LVS apex (
Figure 2A). The mean distance from the LVS apex to the aortic sinus origin of the left coronary artery is approximately 10 mm, but may even reach 21 mm
[15][16]. In approximately 10% of hearts, the left coronary artery trifurcates. Its third branch, the ramus intermedius, penetrates the LVS area, trespassing in the midsection over the accessible and inaccessible areas. In almost 2% of cases, the apex of the LVS cannot be defined because of the absence of the circumflex branch of the left coronary artery since it originates directly from the left sinus of Valsalva. In patients with bicuspid aortic valves, variants are even more commonly observed
[16].
Figure 2. Photographs of cadaveric heart specimens showing the left ventricular summit (LVS) area with overlying epicardial adipose tissue. (A) Section through the septal margin of the LVS; (B) view of the LVS after adipose tissue removal; (C) section through the LVS showing the abundance of the adipose tissue. Black asterisk = aortic–mitral continuity, yellow asterisk = apex of LVS, AIV = anterior interventricular vein, CS = coronary sinus, Cx = circumflex branch of left coronary artery, GCV = great cardiac vein, LAA = left atrial appendage, LAD = left anterior descending artery, LCC = left coronary cusp, LIPV = left inferior pulmonary vein, LMV = left marginal vein, LSPV = left superior pulmonary vein, OM = obtuse marginal branch, MV = mitral valve, SP = septal perforator.
4. The Septal Margin of LVS
The anterior interventricular groove with the adjacent structures—the left anterior descending artery and anterior interventricular vein—determines the septal (or superior) boundary of the LVS
[8][17][18]. The relationship between those two vessels in the majority (54.4%) of cases presents a profound intersection of the anterior interventricular vein to the left anterior descending artery
[19]. Along the artery and vein, the left coronary cardiac nerve forms the left coronary subplexus that contributes to the autonomic innervation of the left ventricle
[20]. The septal margin corresponds with the pulmonary trunk. Nevertheless, a few millimeters of the septal summit, or the septal aspect of LVS, are present between the anterior part of the interventricular groove and the pulmonary trunk. The distance from the left coronary artery bifurcation to the first dominant septal perforator denotes the length of the septal margin of the LVS
[8]. The annulus of the pulmonary valve and pulmonary trunk is present above the LVS septal margin. These structures are overlying the LVS in the most superior aspect. The right ventricular outflow tract correlates with the lower portion of the septal margin, and various amounts of epicardial adipose tissue are present between those structures (
Figure 2C). From the septal margin of LVS, the first or sometimes second diagonal branch enters the LVS region while septal perforators penetrate the ventricular septum. Rarely, the right-sided branches occur, creating preconal ring anastomosis—arterial ring of Vieussens
[21]. The anterior interventricular vein enters from the anterior aspect of the interventricular groove into the LVS.
In some cases, when the most dominant septal perforator is more proximal to the left coronary artery bifurcation, the anterior interventricular vein may not cross the septal margin of the LVS but is then located more distally, outside the LVS triangle. In an uncommon variation, the anterior interventricular vein may not be present within the LVS area as it may drain to the right, towards the anterior cardiac vein
[22]. The presence of the myocardial bridges over the left anterior descending artery should also be mentioned, as their presence is not insignificant within the left coronary artery tree. Nevertheless, within the anterior interventricular artery, the myocardial bridges are usually located outside the LVS area as they are much more likely to be found in the middle segment of the artery (75.3%), followed by distal (20.5%) and proximal (7.4%) segments of the left anterior interventricular artery
[23][24].
5. Mitral Margin of LVS
The left circumflex coronary artery, together with the accompanying left lateral cardiac nerve, forms the mitral (or inferior) boundary of the LVS. In the distal sector of the mitral margin, the small section of the great cardiac vein is also present (
Figure 1)
[25]. In some cases, the circumflex coronary artery is accompanied by the conus vein, which trespasses under the trunk of the left coronary artery (
Figure 2A,B). The length of the mitral margin is equal to the length of the septal margin. This margin neighbors close to the mitral annulus (thus the name of the margin), left fibrous trigon, left atrium and the left atrial appendage base
[19]. The left atrial appendage overlaps the mitral margin, most commonly covering around 80% of its length, but this may vary depending on the left atrial appendage body type (shape)
[26].
In some cases, the mitral margin’s superior aspect may be covered by the pulmonary trunk or left main pulmonary artery
[15]. The first (or sometimes the second) obtuse marginal branch originates from the left circumflex coronary artery entering into the LVS from the mitral margin aspect. The great cardiac vein enters over a mitral margin of the LVS. It crosses the left circumflex coronary artery and then runs into the posterior aspect of the atrioventricular groove. Several spatial relationships between the great cardiac vein and the left circumflex coronary artery may be found, with the vein located over the artery being the most common
[19]. The left phrenic nerve trespassing epicardially over the left atrial appendage and then directing to the left dome of the diaphragm may cross the mitral margin
[27][28][29][30].