
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Qianling Zhang | + 1466 word(s) | 1466 | 2021-07-28 09:56:18 | | | |
| 2 | Enzi Gong | Meta information modification | 1466 | 2021-08-03 10:57:39 | | |
Video Upload Options
Lung cancer can be classified into central and peripheral lung cancer depending on the anatomical part affected, as well as into two main pathological entities: non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC). NSCLC can be further subdivided into three histological subtypes: lung squamous cell carcinoma, lung adenocarcinoma, and large cell carcinoma. NSCLC, which accounts for about approximately 85% of lung cancer cases, has shown an increased mortality rate in recent years. Radical surgery is the most common treatment applied to early-stage NSCLC patients, while chemotherapy is mainly used for NSCLC patients in advanced or recurrent stages.
1. Overview
Lung cancer is one of the most common malignancies with the highest mortality rate and the second-highest incidence rate after breast cancer, posing a serious threat to human health. The accidental discovery of the antitumor properties of cisplatin in the early 1960s aroused a growing interest in metal-based compounds for cancer treatment. However, the clinical application of cisplatin is limited by serious side effects and drug resistance. Therefore, other transition metal complexes have been developed for the treatment of different malignant cancers. Among them, Ru(II/III)-based complexes have emerged as promising anticancer drug candidates due to their potential anticancer properties and selective cytotoxic activity. In this review, we summarized the latest developments of Ru(II/III) complexes against lung cancer, focusing mainly on the mechanisms of their biological activities, including induction of apoptosis, necroptosis, autophagy, cell cycle arrest, inhibition of cell proliferation, and invasion and metastasis of lung cancer cells.
2. Primary Bronchogenic Carcinoma
3. Conclusions
References
- Bade, B.C.; Dela Cruz, C.S. Lung Cancer 2020: Epidemiology, Etiology, and Prevention. Clin. Chest Med. 2020, 41, 1–24.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Zappa, C.; Mousa, S.A. Non-small cell lung cancer: Current treatment and future advances. Transl. Lung. Cancer Res. 2016, 5, 288–300.
- Rossi, A.; Di Maio, M. Platinum-based chemotherapy in advanced non-small-cell lung cancer: Optimal number of treatment cycles. Expert Rev. Anticancer Ther. 2016, 16, 653–660.
- Giaccone, G.; Splinter, T.A.; Debruyne, C.; Kho, G.S.; Lianes, P.; van Zandwijk, N.; Pennucci, M.C.; Scagliotti, G.; van Meerbeeck, J.; van Hoesel, Q.; et al. Randomized study of paclitaxel-cisplatin versus cisplatin-teniposide in patients with advanced non-small-cell lung cancer. The European Organization for Research and Treatment of Cancer Lung Cancer Cooperative Group. J. Clin. Oncol. 1998, 16, 2133–2141.
- Jamieson, E.R.; Lippard, S.J. Structure, Recognition, and Processing of Cisplatin-DNA Adducts. Chem. Rev. 1999, 99, 2467–2498.
- Ghosh, S. Cisplatin: The first metal based anticancer drug. Bioorg. Chem. 2019, 88, 102925.
- Boulikas, T.; Vougiouka, M. Cisplatin and platinum drugs at the molecular level (Review). Oncol. Rep. 2003, 10, 1663–1682.
- Oun, R.; Moussa, Y.E.; Wheate, N.J. The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018, 47, 6645–6653.
- Ma, D.L.; He, H.Z.; Leung, K.H.; Chan, D.S.; Leung, C.H. Bioactive luminescent transition-metal complexes for biomedical applications. Angew. Chem. Int. Ed. Engl. 2013, 52, 7666–7682.
- Ma, D.L.; Wu, C.; Wu, K.J.; Leung, C.H. Iridium(III) Complexes Targeting Apoptotic Cell Death in Cancer Cells. Molecules 2019, 24, 2739.
- Ohata, J.; Ball, Z.T. Rhodium at the chemistry-biology interface. Dalton Trans. 2018, 47, 14855–14860.
- Omondi, R.O.; Ojwach, S.O.; Jaganyi, D. Review of comparative studies of cytotoxic activities of Pt(II), Pd(II), Ru(II)/(III) and Au(III) complexes, their kinetics of ligand substitution reactions and DNA/BSA interactions. Inorg. Chim. Acta 2020, 512, 119883.
- Koiri, R.K.; Mehrotra, A.; Trigun, S.K. Targetting cancer with Ru(III/II)-phosphodiesterase inhibitor adducts: A novel approach in the treatment of cancer. Med. Hypotheses 2013, 80, 841–846.
- Guo, W.; Zheng, W.; Luo, Q.; Li, X.; Zhao, Y.; Xiong, S.; Wang, F. Transferrin serves as a mediator to deliver organometallic ruthenium(II) anticancer complexes into cells. Inorg. Chem. 2013, 52, 5328–5338.
- Lee, S.Y.; Kim, C.Y.; Nam, T.G. Ruthenium Complexes as Anticancer Agents: A Brief History and Perspectives. Drug Des. Dev. Ther. 2020, 14, 5375–5392.
- Jablonska-Wawrzycka, A.; Rogala, P.; Michalkiewicz, S.; Hodorowicz, M.; Barszcz, B. Ruthenium complexes in different oxidation states: Synthesis, crystal structure, spectra and redox properties. Dalton Trans. 2013, 42, 6092–6101.
- Suss-Fink, G. Arene ruthenium complexes as anticancer agents. Dalton Trans. 2010, 39, 1673–1688.
- Hairat, S.; Zaki, M. Half sandwiched Ruthenium(II) complexes: En Route towards the targeted delivery by Human Serum Albumin (HSA). J. Organomet. Chem. 2021, 937, 121732, Corrigendum in 2021, 943, 121734.
- Lu, Y.; Zhu, D.; Gui, L.; Li, Y.; Wang, W.; Liu, J.; Wang, Y. A dual-targeting ruthenium nanodrug that inhibits primary tumor growth and lung metastasis via the PARP/ATM pathway. J. Nanobiotechnol. 2021, 19, 115.
- Lin, K.; Zhao, Z.Z.; Bo, H.B.; Hao, X.J.; Wang, J.Q. Applications of Ruthenium Complex in Tumor Diagnosis and Therapy. Front. Pharmacol. 2018, 9, 1323.
- Liu, J.; Lai, H.; Xiong, Z.; Chen, B.; Chen, T. Functionalization and cancer-targeting design of ruthenium complexes for precise cancer therapy. Chem. Commun. 2019, 55, 9904–9914.
- Jakubaszek, M.; Goud, B.; Ferrari, S.; Gasser, G. Mechanisms of action of Ru(ii) polypyridyl complexes in living cells upon light irradiation. Chem. Commun. 2018, 54, 13040–13059.
- Mede, T.; Jager, M.; Schubert, U.S. “Chemistry-on-the-complex”: Functional Ru(II) polypyridyl-type sensitizers as divergent building blocks. Chem. Soc. Rev. 2018, 47, 7577–7627.
- Oliveira, K.M.; Honorato, J.; Goncalves, G.R.; Cominetti, M.R.; Batista, A.A.; Correa, R.S. Ru(II)/diclofenac-based complexes: DNA, BSA interaction and their anticancer evaluation against lung and breast tumor cells. Dalton Trans. 2020, 49, 12643–12652.
- Rilak Simović, A.; Masnikosa, R.; Bratsos, I.; Alessio, E. Chemistry and reactivity of ruthenium(II) complexes: DNA/protein binding mode and anticancer activity are related to the complex structure. Coord. Chem. Rev. 2019, 398, 113011.
- Mari, C.; Gasser, G. Lightening up Ruthenium Complexes to Fight Cancer? Chimia 2015, 69, 176–181.
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic therapy. J. Nat. Inst. 1998, 90, 889–905.
- Carruth, J.A. Clinical applications of photodynamic therapy. Int. J. Clin. Pract. 1998, 52, 39–42.
- Thota, S.; Rodrigues, D.A.; Crans, D.C.; Barreiro, E.J. Ru(II) Compounds: Next-Generation Anticancer Metallotherapeutics? J. Med. Chem. 2018, 61, 5805–5821.
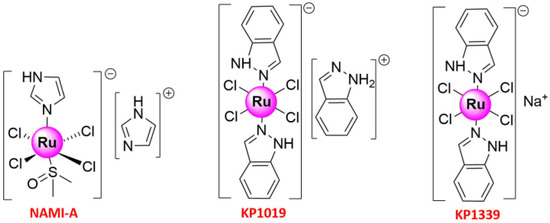
- Alessio, E.; Messori, L. NAMI-A and KP1019/1339, Two Iconic Ruthenium Anticancer Drug Candidates Face-to-Face: A Case Story in Medicinal Inorganic Chemistry. Molecules 2019, 24, 1995.
- Alessio, E. Thirty Years of the Drug Candidate NAMI-A and the Myths in the Field of Ruthenium Anticancer Compounds: A Personal Perspective. Eur. J. Inorg. Chem. 2017, 1549–1560.
- Coverdale, J.; Laroiya-McCarron, T.; Romero-Canelón, I. Designing Ruthenium Anticancer Drugs: What Have We Learnt from the Key Drug Candidates? Inorganics 2019, 7, 31.
- Leijen, S.; Burgers, S.A.; Baas, P.; Pluim, D.; Tibben, M.; van Werkhoven, E.; Alessio, E.; Sava, G.; Beijnen, J.H.; Schellens, J.H. Phase I/II study with ruthenium compound NAMI-A and gemcitabine in patients with non-small cell lung cancer after first line therapy. Investig. New Drugs 2015, 33, 201–214.
- Pluim, D.; van Waardenburg, R.C.; Beijnen, J.H.; Schellens, J.H. Cytotoxicity of the organic ruthenium anticancer drug Nami-A is correlated with DNA binding in four different human tumor cell lines. Cancer Chemother. Pharmacol. 2004, 54, 71–78.
- Hartinger, C.G.; Jakupec, M.A.; Zorbas-Seifried, S.; Groessl, M.; Egger, A.; Berger, W.; Zorbas, H.; Dyson, P.J.; Keppler, B.K. KP1019, a new redox-active anticancer agent--preclinical development and results of a clinical phase I study in tumor patients. Chem. Biodivers. 2008, 5, 2140–2155.
- Hartinger, C.G.; Zorbas-Seifried, S.; Jakupec, M.A.; Kynast, B.; Zorbas, H.; Keppler, B.K. From bench to bedside--preclinical and early clinical development of the anticancer agent indazolium trans-[tetrachlorobis(1H-indazole)ruthenate(III)] (KP1019 or FFC14A). J. Inorg. Biochem. 2006, 100, 891–904.
- Jakupec, M.A.; Arion, V.B.; Kapitza, S.; Reisner, E.; Eichinger, A.; Pongratz, M.; Marian, B.; Graf von Keyserlingk, N.; Keppler, B.K. KP1019 (FFC14A) from bench to bedside: Preclinical and early clinical development—An overview. Int. J. Clin. Pharmacol. Ther. 2005, 43, 595–596.
- Lentz, F.; Drescher, A.; Lindauer, A.; Henke, M.; Hilger, R.A.; Hartinger, C.G.; Scheulen, M.E.; Dittrich, C.; Keppler, B.K.; Jaehde, U.; et al. Pharmacokinetics of a novel anticancer ruthenium complex (KP1019, FFC14A) in a phase I dose-escalation study. Anticancer Drugs 2009, 20, 97–103.
- Kapitza, S.; Pongratz, M.; Jakupec, M.A.; Heffeter, P.; Berger, W.; Lackinger, L.; Keppler, B.K.; Marian, B. Heterocyclic complexes of ruthenium(III) induce apoptosis in colorectal carcinoma cells. J. Cancer Res. Clin. Oncol. 2005, 131, 101–110.
- Biersack, B.; Schobert, R. Current State of Metal-Based Drugs for the Efficient Therapy of Lung Cancers and Lung Metastases. Adv. Exp. Med. Biol. 2016, 893, 211–224.
- Alessio, E.; Mestroni, G.; Bergamo, A.; Sava, G. Ruthenium antimetastatic agents. Curr. Top. Med. Chem. 2004, 4, 1525–1535.
- Zheng, K.; Wu, Q.; Wang, C.; Tan, W.; Mei, W. Ruthenium(II) Complexes as Potential Apoptosis Inducers in Chemotherapy. Anticancer Agents Med. Chem. 2017, 17, 29–39.
- Sun, R.W.; Ng, M.F.; Wong, E.L.; Zhang, J.; Chui, S.S.; Shek, L.; Lau, T.C.; Che, C.M. Dual anti-angiogenic and cytotoxic properties of ruthenium(III) complexes containing pyrazolato and/or pyrazole ligands. Dalton Trans. 2009, 48, 10712–10716.
- Riccardi, C.; Musumeci, D.; Irace, C.; Paduano, L.; Montesarchio, D. RuIIIComplexes for Anticancer Therapy: The Importance of Being Nucleolipidic. Eur. J. Org. Chem. 2017, 2017, 1100–1119.
- Riccardi, C.; Musumeci, D.; Trifuoggi, M.; Irace, C.; Paduano, L.; Montesarchio, D. Anticancer Ruthenium(III) Complexes and Ru(III)-Containing Nanoformulations: An Update on the Mechanism of Action and Biological Activity. Pharmaceuticals 2019, 12, 146.
- Zeng, L.; Gupta, P.; Chen, Y.; Wang, E.; Ji, L.; Chao, H.; Chen, Z.S. The development of anticancer ruthenium(ii) complexes: From single molecule compounds to nanomaterials. Chem. Soc. Rev. 2017, 46, 5771–5804.




