An extraction technology works on the principle of two consecutive steps that involves mixture of solute with solvent and the movement of soluble compounds from the cell into the solvent and its consequent diffusion and extraction. The conventional extraction techniques are mostly based on the use of mild/high temperatures (50–90 °C) that can cause thermal degradation, are dependent on the mass transfer rate, being reflected on long extraction times, high costs, low extraction efficiency, with consequent low extraction yields. Due to these disadvantages, it is of interest to develop non-thermal extraction methods, such as microwave, ultrasounds, supercritical fluids (mostly using carbon dioxide, SC-CO2), and high hydrostatic pressure-assisted extractions which works on the phenomena of minimum heat exposure with reduced processing time, thereby minimizing the loss of bioactive compounds during extraction. Further, to improve the stability of these extracted compounds, nano-encapsulation is required. Nano-encapsulation is a process which forms a thin layer of protection against environmental degradation and retains the nutritional and functional qualities of bioactive compounds in nano-scale level capsules by employing fats, starches, dextrins, alginates, protein and lipid materials as encapsulation materials.
Bioactive compounds also known as secondary metabolites are widely present in plant matrix and over the past few decades, several in vitro and in vivo reports including epidemiological, and cohort studies provide evidence that consumption of plant-based food provides protection against several diseases. These bioactive extracts are also capable of treating chronic diseases including cancer, cardiovascular and diabetes mellitus (DM). Nutraceutical and pharmaceutical sectors use these extracts to develop functional food- and plant-based medicines, which have a potential to cure and deliver health benefits. According to the World Health Organization (WHO), about 80% of the global population depends on natural medicines. The initial steps followed to use these active compounds from plant matrix are extraction followed by pharmacological testing, isolation, characterization and clinical evaluation. Figure 1 represents a detailed flow chart of bioactive compound extraction from plant matrix.
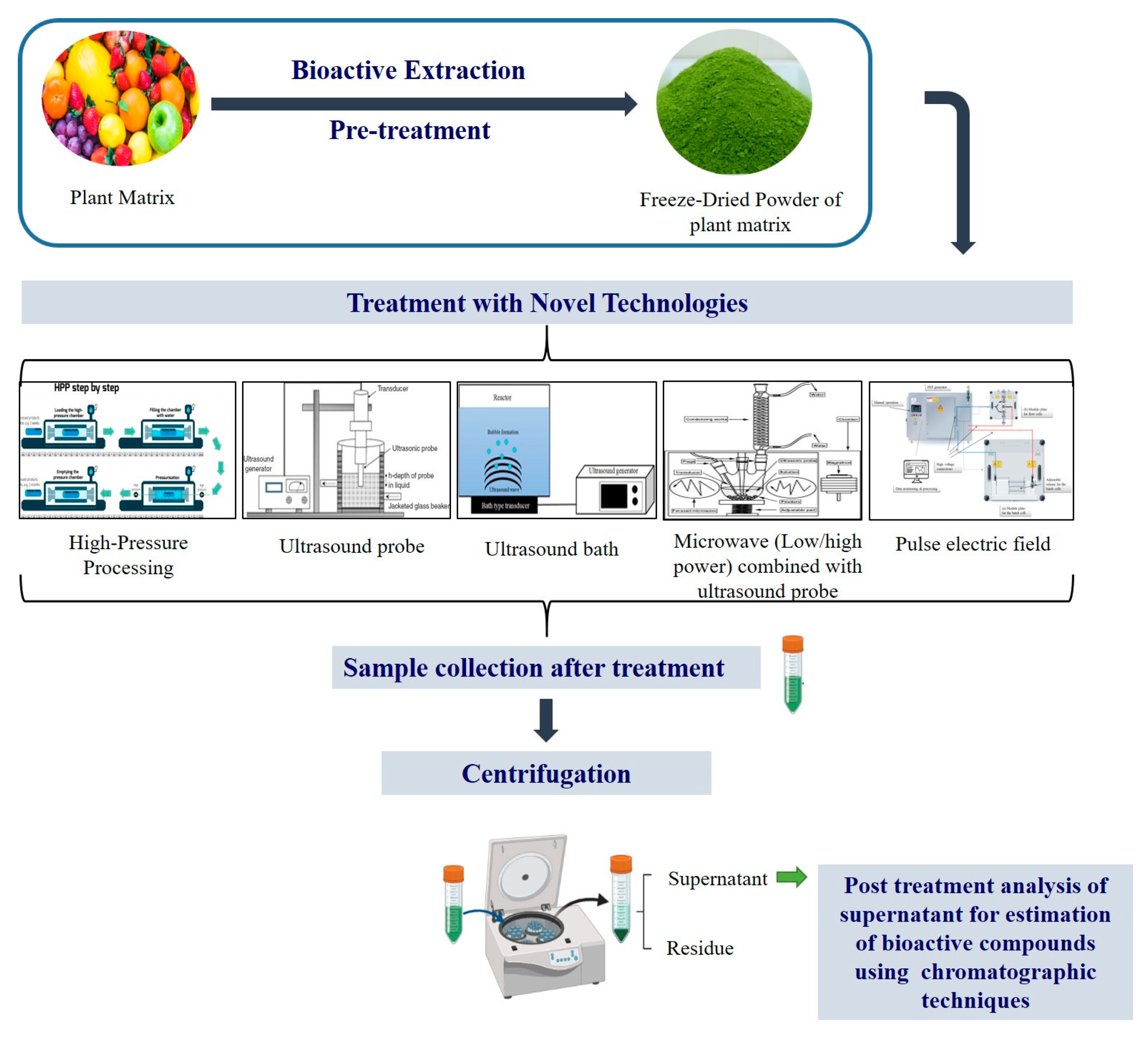 Figure 1.
Figure 1. Illustration for the extraction of bioactive compound using novel strategies.
The quality and yield of the bioactive compounds depend on two important factors: (a) the method opted for its extraction, (b) its extraction parameters including plant matrix type, solvent used, time and temperature. The most conventional method employed for bioactive extraction is Soxhlet extraction, maceration and hydro-distillation. Although, these techniques are commercially employed, but excessive use of solvents and longer processing times are the downsides of these technologies. Presently, demand for sustainable, chemical-free, advanced extraction processes with enhanced overall yield of bioactive compounds, also known as “green techniques”, which include ultrasound-assisted, enzyme-assisted, microwave-assisted, pulsed electric field-assisted, high-pressure processing, supercritical fluid and pressurized liquid extraction processes are gaining attention. Treating the plant matrix with these green technologies helps in breaking the cell structure, which allows the bioactive compound to leach or rinse out from the cell wall through solvents; as a result, it enhanced extraction yield. Further, purification of the extracted bioactive poses another technological challenge as each of these compounds has a unique molecular structure depending on their type, source and biological activity. The extracted compounds can be further purified, employing super critical CO2 isolation, by addition of a co-solvent including ethanol, water to isolate the respective bioactive compound efficiently at an optimized temperature and pressure
[1]. In addition, it is essential to protect the extracted bioactive compound post extraction and purification, as these compounds are highly sensitive to environment exposure including moisture and high temperature (sensitive under heat, light, oxygen). Therefore, protection techniques such as nanoencapsulation are used to ensure that biological activity of these compounds is preserved until they reach and perform their function at the targeted location in the human body.
Encapsulation plays a vital role in protecting the bioactive compounds from getting degraded. At present, there are two kinds of encapsulation including microencapsulation and nanoencapsulation. The reason why nanoencapsulation is preferred over microencapsulation is due to its nano-scale size, as the smaller the size of the capsules, the higher their bioavailability and their release can be modified and controlled in a better way comparatively. Nanoencapsulation provides a protective shield around bioactive compounds. It is a system where a suitable nano-carrier, resistant to enzymatic degradation especially in gastrointestinal tract including chitosan, zein, and alginate, are widely used to encapsulate bioactive compounds employing several delivery methods including association colloids, nano-particles, nano-emulsions, nano-fibers/nano-tubes, nano-laminates. The selection encapsulation method is based on two main factors: (a) nature of the core material; (b) nature of wall material including wall material size, thickness, solubility, permeability and its rate of delivery. Basically, these techniques are classified into three main genres including chemical (emulsion and interfacial polymerization), physical–chemical (emulsification and coacervation) and physical–mechanical methods (spray-drying/spray-cooling/spray-congealing/prilling, freeze-drying, electrodynamic methods and extrusion)
[2]. However, in certain cases, combinations of these techniques are practiced as in the case of emulsification; first using homogenization, the emulsions are prepared and later converted to dry powder state using spray-drying and/or freeze-drying techniques
[3]. Reports indicate that about 80–90% of flavor encapsulation is done using spray-drying, while 5–10% by spray-chilling, 2–3% by melt extrusion and ~2% by melt injection
[4]. Castro et al.
[5] reported electro-spinning encapsulation as a heat-free technique to encapsulate fragrance and flavor, which is extremely promising for heat-sensitive compounds.
To recapitulate, this chapter provides a comprehensive summary on several aspects of bioactive extraction using non-thermal technologies and its nanoencapsulation. A brief description on nano-carriers employed for encapsulation is also discussed along with the detailed description of their application in food systems. Various opportunities and future challenges are also outlined.

 Figure 1. Illustration for the extraction of bioactive compound using novel strategies.
Figure 1. Illustration for the extraction of bioactive compound using novel strategies.
