
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Stanislav Kotlyarov | + 3342 word(s) | 3342 | 2021-07-02 08:50:51 | | | |
| 2 | Amina Yu | + 345 word(s) | 3687 | 2021-07-12 03:27:07 | | |
Video Upload Options
ABC transporters are a large family of membrane proteins that transport chemically diverse substrates through the lipid bilayer of cell plasma membranes while accompanied by ATP hydrolysis [47,48]. Currently, 49 different genes encoding ABC transporters are known in humans, which, based on the structural features, are divided into seven subfamilies, designated ABCA–ABCG .
1. Introduction
Chronic obstructive pulmonary disease (COPD) is an important medical problem, which is due to the high prevalence of the disease, its impact on the quality of life, and the high incidence of disability and mortality [1][2][3]. An important characteristic of COPD is the variety of its clinical manifestations, which are based on a variety of not fully studied pathophysiological mechanisms [4][5][6]. This clinical heterogeneity has different pulmonary and extrapulmonary characteristics, such as the development of emphysema and other comorbidities [7][8]. Comorbid diseases make a significant contribution to the clinical picture of COPD [9][10][11]. It is considered that atherosclerosis and COPD have a number of common mechanisms of development, which allows us to consider them comorbid diseases [11][12].
The close relationship between COPD and atherosclerosis is well known to clinicians [13]. The effect of the rate of bronchial obstruction increase and the increase in the number of COPD exacerbations on the progression of atherosclerosis is also well known [14][15][16].In turn, the significance of lipid metabolism disorders on lung function in atherosclerosis is of great clinical and research interest.It is known that blood plasma lipids can be associated with pulmonary function [17][18][19].
Interestingly, atherosclerosis is not often equally represented in different groups of patients with COPD but is more characteristic of the bronchitis phenotype, which suggests the presence of common disturbed mechanisms [20]. To date, there is no clear understanding of all the processes that connect the development and progression of COPD and atherosclerosis, but it is considered that the connecting link is systemic inflammation [20][21]. Many cells are involved in the maintenance of systemic inflammation, including macrophages, which are known for the diversity of their functions [21]. In both COPD and atherosclerosis, macrophages excessively infiltrate the bronchial or vascular wall, respectively [22][23][24].
They are also key participants in the initiation and progression of atherosclerosis, which is determined by their role in the uptake of modified lipoproteins in the arterial walls, and the production of inflammatory mediators and matrix metalloproteinases that contribute to the instability of atherosclerotic plaques [25]. A violation of the normal homeostasis of cholesterol in macrophages and a massive accumulation of its esters in lipid droplets lead to the acquisition of the so-called “foam cell” phenotype by cells [26]. Removing excess cholesterol from cells is called reverse cholesterol transport. Currently, the role of lipid metabolism disorder in the development of atherosclerosis is sufficiently understood and supported by the results of numerous studies.
Recent data have shown that lipid metabolism disorders are involved in many cross-immunometabolic pathways and participate in the development of COPD [27][28]. For example, the association of low body weight in COPD with high mortality is known, which has been called the “obesity paradox” [29][30][31]. Despite the fact that the prognostically significant cause of weight loss in this category of patients is the loss of muscle mass rather than fat, the importance of lipid metabolism disorders in COPD patients is not in doubt. As it is well known, excess body weight correlates well with an unfavorable prognosis in cardiovascular diseases of an atherosclerotic nature, but it has an inverse relationship with the prognosis of COPD [32], which is an important difference that characterizes the features of lipid metabolism in these patients.
The results of studies published in recent years suggest that cellular lipid metabolism has numerous cross-links with the immune response [33]. In this regard, disorders of lipid metabolism mediated by ATP-binding cassette (ABC) transporters are of clinical interest (Figure 1).
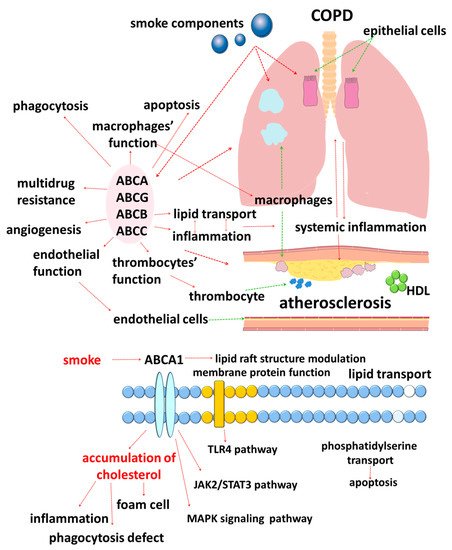
ABC transporters are a large family of membrane proteins that transport chemically diverse substrates through the lipid bilayer of cell plasma membranes while accompanied by ATP hydrolysis [34][35]. Currently, 49 different genes encoding ABC transporters are known in humans, which, based on the structural features, are divided into seven subfamilies, designated ABCA–ABCG [36][37][38][39][40].
Moreover, lipid carriers are present in all subfamilies of ABC transporters, which emphasizes the importance of lipid transport [41][42][43]. The need for special transport mechanisms for lipids is due to their insolubility in water [41]. In addition, cell membranes are heterogeneous in their lipid composition [42]. Unlike flippases, ABC transporters are responsible for the movement of lipid substrates from the inner leaflet of the plasma membrane to the outer leaflet, where lipids must be desorbed or diffuse to extracellular lipid acceptors [41].
The accumulated data in recent years indicate an important role of cholesterol in the functioning of ABC transporters. Cholesterol is a key molecule of the plasma membrane that provides stabilization of the spatial structure of the lipid bilayer [44][45]. Such a spatial arrangement of the molecules is considered to contribute to the participation of cholesterol in the regulation of the function of transmembrane proteins through two main mechanisms: direct interaction of the sterol with specific protein binding sites and indirect influence on the biophysical properties of the membrane [44][45][46][47]. This information is of great clinical and research interest since it allows us to assess the significance of separate processes in the development and progression of comorbid COPD and atherosclerosis in a different way.
The objective of this review was the analysis of the role of lipid metabolism disorders in the pathogenesis of the comorbid course of COPD and atherosclerosis and the participation of representatives of ABC transporters in these processes.
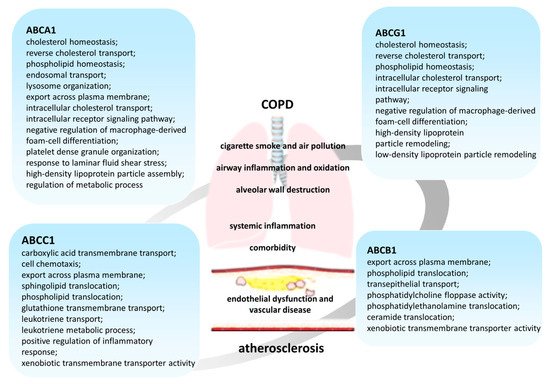
2. Subfamily of ABCB Transporters
Members of the ABCB subfamily are well known to clinicians and researchers for their role in the development of multiple drug resistance. At the same time, a number of members of the ABCB subfamily are also characterized by their ability to transport lipids, such as ABCB1 [42][43][48][49], or ABCB4, which participates in the translocation of phosphatidylcholine lipids into bile [43][50][51].
(P-glycoprotein, MDR1 (multidrug resistance 1 gene)), cloned by J.R. Riordan in 1985, was originally described as a major participant in the mechanism of multiple drug resistance to chemotherapeutic agents of colchicine-resistant Chinese hamster ovarian cells [52][53][54]. To date, ABCB1 is one of the most well-studied ABC transporters (Figure 2) [42][43][55][56]. Its broad substrate specificity allows it to carry out the transport of chemically diverse molecules, including medicinal substances [43][48][57]. However, the mechanism by which ABCB1 can recognize different chemical structure substrates remains largely unknown.
It is known that ABCB1 participates in lipid transport [42][43][58], moving lipids from the inner to the outer leaflet of the plasma membrane of cells [43][59]. It was shown that cholesterol is recognized and transported as an endogenous ABCB1 substrate [60][61][62]. Moreover, ABCB1 not only transports cholesterol across the membrane but also its functional activity is modulated by cholesterol in the membrane [61][63]. A direct interaction between lipid molecules and ABCB1 was revealed [43][59][64].
It was shown that the predominant localization of ABCB1 in lipid rafts is necessary for its functioning [62][65][66]. Cholesterol depletion, which leads to the destruction of lipid rafts, disrupts the membrane localization of ABCB1 and reduces its transport activity [67][68], which leads to the intracellular accumulation of drugs in cells [61]. There is a supposition that sterols may interact with ABCB1 and modulate its structure and function by occupying part of the drug-binding pocket or by binding to assumed consensus cholesterol-binding motifs (CRAC/CARC) that are located in the transmembrane domains [42][45][47][69][70]. Thus, the effect of cholesterol on the transport activity of ABCB1 is of great clinical interest.
Although the lungs are not among the organs with a high expression of ABCB1, the protein is found in the bronchi, where it is mainly localized on the apical surface of the ciliated epithelial cells, the apical and lateral surfaces of the serous cells of the bronchial glands, and the lumen surface of the endothelial cells of the bronchial capillaries, but it is not found in the mucus-secreting goblet cells [71][72]. In the alveoli, ABCB1 is expressed in type I alveolar epithelial cells [73][74].
ABCB1 is activated during differentiation from monocytes to macrophages and is sensitive to activation by LXR (liver X receptor) agonists [75][76]. In addition, it is also present in macrophages, with higher expression in the M2 subtype compared to M1 macrophages [65][77][78][79]. It has been reported that tobacco smoke components affect the expression and functional activity of ABCB1 [74][80]. A decrease in the expression ofABCB1in the cells of smokers compared to non-smokers was shown.
It should be noted that the inhaled administration of glucocorticoids in patients with severe COPD may cause existing differences in the expression ofABCB1in the lungs [81].
It has been shown that polymorphism of theABCB1gene can affect the effectiveness of COPD therapy [82] and mediate extrapulmonary complications of the disease [83]. In addition, elevated levels of mRNA ABCB1 were found in the tissues in atherosclerosis, which allows us to suggest a role of ABCB1 in the development of atherosclerotic lesions in vivo [76][84].
The expression of mRNA ABCB4 in monocytes and macrophages is also shown [85][86], and the ABCB4 of macrophages prevents the formation of “foam cells,” reducing the accumulation of lipids and providing an atheroprotective function [86]. A lower serum HDL cholesterol level was observed in Abcb4 knockout mice fed with normal food, which confirmed the effect of ABCB4 on cholesterol metabolism [86][87]. Since ABCB4 is a carrier of phosphatidylcholine from the inner to the outer sheet of the plasma membrane, violations of the functional activity of the protein can probably lead to asymmetry of the phospholipid membrane of macrophages [86][88]. Thus, ABCB4 can indirectly contribute to atherogenesis by affecting the accumulation of lipids in macrophages as a result of modulation of the lipid membrane asymmetry [86].
The results of bioinformatics analysis indicate that exposure to cigarette smoke is associated with increased levels ofABCB6expression in bronchial epithelial cells, while smoking cessation is associated with lowerABCB6expression [89].
Literature data suggest that ABCB6 protects cells from oxidative stress by modulating cytosolic reactive oxygen species [90][91]. Despite this, an increase in the expression ofABCB6may also have negative effects on the form of increased resistance to chemotherapeutic agents [89].
It is known that platelets participate in the pathogenesis of atherosclerosis [92][93]. ligand 4), on the surface of endothelial cells, facilitating the recruitment of leukocytes to inflammatory foci, and also form aggregates with neutrophils and monocytes, which plays a key role in the production of inflammatory cytokines, the biosynthesis of leukotrienes, and the production of reactive oxygen species [93][94][95][96]. A high level of ABCB6 is observed in megakaryocyte progenitor cells and its deficiency leads to an increase in the number of circulating platelets, the interaction of platelets with inflammatory leucocytes, and the accelerated development of atherosclerosis [97].
Thus, representatives of the ABCB family are of great clinical importance, not only due to their involvement in the efflux activity for xenobiotics but also due to the movement of endogenous substrates.
3. Subfamily of ABCC Transporters
ABCC1 (MRP1 (multidrug resistance-associated protein 1)) was initially identified as a glutathione-conjugate transporter [42][98][99], but later its participation in lipid transport [42][100] and inflammatory responses was described (Figure 2).
In the lungs, ABCC1 is found in alveolar macrophages, as well as in bronchial epithelial cells [101], and its expression levels differ in different parts of the respiratory tract [89]. Localization of the protein in the cells of the distal parts of bronchi was already associated with participation in the development of COPD [102]. Interestingly, in contrast to ABCB1, ABCC1 in basal cells was distributed along the entire circumference of the plasma membrane, and in ciliated cells, it is localized on the basolateral surface [103], which predetermines the functional differences of the transporters [104].
ABCC1 is localized in cholesterol-rich actin-dependent lipid rafts, which determines the functional dependence of the efflux activity of the transporter on cortical actin However, there is little information in the literature about the participation of cholesterol in the modulation of the ABCC1 transport function. In one study, it was found that the functional activity of the transporter decreased with a decrease in the level of cellular cholesterol [105], but in other studies, it was shown that cholesterol was not a necessary factor for the function of ABCC1 and, apparently, did not participate in the mechanisms of functional connection of ABCC1 with lipid rafts [106].
It was shown that ABCC1 can protect the lungs from developing COPD by reducing the oxidative stress caused by smoking, preventing the accumulation of toxic metabolites [107][108]. It is assumed that the expression of theABCC1gene is regulated by a feedback mechanism since it is associated with oxidative stress and exposure to toxins caused by exposure to cigarette smoke and contributes to the enhancement of antioxidant activity in the epithelial cells of the respiratory tract [89].
A lowerABCC1expression in the bronchial epithelium of COPD patients compared to healthy former smokers was shown [109]. Expression was also lower in patients with severe COPD than in those with mild or moderate COPD. In contrast, data from a recent bioinformatics analysis show an increase in the expression of theABCC1gene in smoking patients with COPD compared to people who do not have COPD [89]. The available contradictory information on the expression of the transporter in patients with COPD may be associated with the use of glucocorticoids in the severe course of disease [110].
Leukotrienes, which are a group of highly effective lipid mediators, are important participants in the antibacterial protection of the lungs. Leukotriene LTC4occupies an important place among the physiological substrates of ABCC1 [99][111][112]. In this connection, the transport activity of ABCC1 may participate in pulmonary inflammation. They showed reduced pneumococcal growth in the lungs and strongly reduced mortality, which was associated with an increase in circulating LTB4, which is a powerful chemoattractant for neutrophils and increases the activity of phagocytic cells [113][114][115].
In addition to these data, it was shown that mice with triple knockout of theMrp1andMdr1a/1bgenes were more susceptible to the development of COPD. These mice had lower levels of IL-8 production and showed an almost complete absence of inflammatory cells in response to cigarette smoke [116][111]. The impaired inflammatory response was likely associated with lower LTC4excretion [117].
Data about the participation of ABCC1 in the export of sphingosine-1-phosphate (S1P), which is a lipid mediator that is involved in many processes, including inflammation, angiogenesis, apoptosis, and macrophage function, are of great importance [118][119][120][121][122][123][124][125][126][127]. S1P also regulates the integrity of the endothelial barrier by modulating the endothelial cytoskeleton [128].
It was shown that S1P levels were elevated in the induced sputum of COPD patients compared to non-smokers [129]. It was suggested that S1P may be a participant in defective phagocytosis by macrophages in COPD [129][130].
Interestingly, the impairment of S1P metabolism is an important factor determining the emphysematous phenotype in COPD [131].
In addition to ABCC1, ABCA1 and ABCG2 also participate in S1P transport [120][132][133].
It has been determined that ABCC1 plays a definite role in the regulation of vascular endothelial homeostasis and arterial blood pressure by inducing the release of glutathione from vascular endothelial cells [134][135][136]. ABCC1 is found in large quantities in vascular smooth muscle cells, which make up the majority of vascular wall cells and are involved in the process of atherosclerosis. ABCC1 acts as a transporter for substances such as glutathione, oxidized glutathione, and leukotriene C4(LTC4) [134][136][137], which are potentially essential for regulating the production of reactive oxygen species in vascular cells. In addition, modulation of ABCC1 expression in human aortic endothelial cells affects vascular function [134][137].
Thus, ABCC1 demonstrates involvement in the development of COPD and atherosclerosis. However, the mechanisms of this participation require further study.
4. Subfamily of ABCG Transporters
It is known that many proteins of the ABCG subfamily also participate in lipid homeostasis [138]. An important role in lipid metabolism is assigned to ABCG1 and ABCG4 transporters, which are half-type ABC proteins since they consist of only one transmembrane domain and one nucleotide-binding domain. For activation to occur, the protein must form a dimer (homodimer or heterodimer) or even an oligomer depending on the function [139][140][141][142][143].
It is expressed in many cell types, including myeloid cells, lymphocytes, epithelial, and endothelial cells of various organs [144][145][146][147]. It is known that, like ABCA1, ABCG1 removes excess cholesterol from peripheral cells, saturating HDL with it and protecting cells from sterol overload [143][148][149]. In the lungs,ABCG1is expressed in various cell types, including alveolar macrophages, epithelial cells, and type II pneumocytes [144][150][151]. The absence of ABCG1 leads to progressive chronic lung inflammation, which is associated with impaired regulation of intracellular cholesterol levels [152][151][153].
It was shown that Abcg1−/−macrophages were characterized by increased production of proinflammatory cytokines IL-6, IL-1β, IL-1α, and IL-12, and a decrease in anti-inflammatory cytokine IL-10 [153][154], which is associated with the lipid load of macrophages. Elevated levels of matrix metalloproteinases MMP-8 and MMP-12 were also found out in the lungs ofAbcg1−/−mice [153], they destroy the extracellular matrix, are overexpressed in COPD patients, and are associated with airway inflammation and remodeling [155][156].
The information about the participation of ABCG1 in the polarization of macrophages is important. Experimental data indicate that ABCG1 deficiency contributes to the proinflammatory M1 polarization of human macrophages, and the molecular mechanism is probably mediated via the Akt signaling pathway [157]. Moreover, this phenotypic shift is more pronounced when the diet of mice was similar to the Western one [158]. According to the existing concepts, most of the cells in the center of the atherosclerotic plaque are M1 macrophages.
It is believed that ABCG1 participates in the apoptosis of cells, including macrophages [159][143][160][161]. It was determined that macrophages of miceAbcg1−/−have an increased ability to absorb apoptotic cells, accumulate lipids, and become apoptotic [154][162][163].
It is known that apoptosis plays an important role not only in the development of COPD but also in the pathogenesis of atherosclerosis: it is antiatherogenic in the early foci of lesions, and the apoptosis of macrophages contributes to atherogenesis in progressive lesions [159]. The mechanism of ABCG1 participation in apoptosis is probably determined by its cholesterol transport activity and its functioning as an inducer or inhibitor of apoptosis depends on the localization of the transporter on the plasma membrane or intracellular membranes of organelles [159].
Taking into account the presence of the ABCG1-transporter in macrophages (“foam cells”) in human atherosclerotic plaque, it was assumed that macrophagic ABCG1 plays an important role in the development of atherosclerotic lesions This assumption was confirmed in numerous studies [76][85][157][164][165][166]. The participation of ABCG1 in lipid homeostasis was analyzed by a number of authors who demonstrated that mRNA expression and ABCG1 synthesis in macrophages can be induced by cholesterol loading [159][85][166][167]. Thus, ABCG1 is also important for intracellular cholesterol transport [168][169].
Another member of the subfamily, ABCG4 has 69% identity and 84% similarity in amino acid composition to ABCG1 and mediates the outflow of cholesterol to However, unlike ABCG1, the expression of ABCG4 is limited. The transporter is found out in the brain and hematopoietic organs [146][170][171][172][173]. Another important difference is that the activation of LXR induces the expression of Abcg1, but does not affect the expression of Abcg4 [172].
The obtained results allowed for suggesting the role of ABCG1 and ABCG4 transporters in cell proliferation, apoptosis, and immune responses, and that these different processes may be related to the regulation of lipid metabolism [174][175][176].
Although ABCG4 is not expressed in macrophage foam cells, its potential role in atherogenesis is described [177]. It is known that ABCG4 inhibits the proliferation of megakaryocyte progenitor cells via reducing the transmission of thrombopoietin receptor signals in lipid rafts [143][178]. In addition to increased atherogenesis, arterial thrombosis was found in mice withAbcg4gene knockout, which correlated with an increase in the number of reticular platelets, platelet complexes, leucocytes, and microparticles of platelet origin, which have proven pro-atherosclerotic and prothrombotic properties. Researchers associated increased platelet production caused by impaired cholesterol metabolism in progenitor cells with accelerated atherogenesis and arterial thrombosis [179][178].
ABCG2 (BCRP, breast cancer resistance protein) is another representative of the ABCG subfamily and is most commonly associated with drug excretion [42][180]. BCRP has been found in many organs and tissues, including the endothelium of venous vessels and capillaries, where it performs a protective and barrier function [181].
The expression of ABCG2 in the lungs is low and decreases in the trachea– large bronchi–small bronchi series, but the protein is found in the epithelial layer and seromucinous glands, as well as in the endothelial cells of capillaries [182]. ABCG2 is also expressed in alveolar pneumocytes and is mainly localized on the apical membrane and, to a lesser extent, in the cytosol and in the cell nuclei, where, according to some assumptions, it acts as a transcription factor, regulating gene expression. During differentiation from alveolar pneumocytes of type II to alveolar pneumocytes of type-I-like phenotype, the expression of
The role of BCRP/ABCG2 in the lungs is completely unknown. Furthermore, lung pathologies in Abcg2−/−mice have not been reported yet [78][183][184][185].
It has been shown that ABCG2 in lung tissues is responsible for the formation of the SP-phenotype (side population) of lung cancer cells, which have high efflux activity [186].
It was found in numerous studies that BCRP also participates in the transport of sterols, and cholesterol can stimulate the ATPase activity of the transporter [42][177][187][188]. This was confirmed by the fact that a change in the structure of lipid rafts directly leads to the redistribution of the BCRP protein in areas with a higher cholesterol density. With a decrease in the cholesterol content in the lipid rafts, the content of BCRP substrates in the cells increases, which indicates the inhibition of the transporter protein. The restoration of cholesterol and its saturation of lipid rafts leads to the normalization of the functional activity of the transporter [189][190].
ABCG5 and ABCG8 play a role in lipid metabolism and mediate the outflow of cholesterol and sitosterol from the intestinal walls and hepatocytes to the bile duct and intestinal lumen [61][191]. There is no information in the literature about the participation of these transporters in lung function. However, it is known that mutations of theABCG5andABCG8genes cause sitosterolemia, which is characterized by an increase in the absorption of plant and fish sterols, and their reduced biliary excretion leads to an increase in the level of toxic sitosterols in the blood and the early development of atherosclerosis and myocardial infarction. The participation of ABCG5 and ABCG8 in atherogenesis may also consist in the fact that they provide trans-intestinal excretion of cholesterol, i.e., an alternative non-biliary route of its excretion [192].
Таким образом, представители подсемейства участвуют в транспорте липидов и множества важных функций. В патогенезе ХОБЛ и атеросклероза ABCG1 является одним из наиболее интересных, поскольку нарушения его транспортной активности нарушают ряд процессов, связанных с возбуждением.
References
- May, S.M.; Li, J.T. Burden of chronic obstructive pulmonary disease: Healthcare costs and beyond. Allergy Asthma Proc. 2015, 36, 4–10.
- Quaderi, S.A.; Hurst, J.R. The unmet global burden of COPD. Glob. Health Epidemiol. Genom. 2018, 3, e4.
- Soriano, J.B.; Kendrick, P.J.; Paulson, K.R.; Gupta, V.; Abrams, E.M.; Adedoyin, R.A.; Adhikari, T.B.; Advani, S.M.; Agrawal, A.; Ahmadian, E.; et al. Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir. Med. 2020, 8, 585–596.
- Barnes, P.J. COPD 2020: New directions needed. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 319, L884–L886.
- Castaldi, P.J.; Benet, M.; Petersen, H.; Rafaels, N.; Finigan, J.; Paoletti, M.; Marike Boezen, H.; Vonk, J.M.; Bowler, R.; Pistolesi, M.; et al. Do COPD subtypes really exist? COPD heterogeneity and clustering in 10 independent cohorts. Thorax 2017, 72, 998–1006.
- Ragland, M.F.; Benway, C.J.; Lutz, S.M.; Bowler, R.P.; Hecker, J.; Hokanson, J.E.; Crapo, J.D.; Castaldi, P.J.; DeMeo, D.L.; Hersh, C.P.; et al. Genetic Advances in Chronic Obstructive Pulmonary Disease. Insights from COPDGene. Am. J. Respir. Crit. Care Med. 2019, 200, 677–690.
- Sillen, M.J.; Franssen, F.M.; Delbressine, J.M.; Uszko-Lencer, N.H.; Vanfleteren, L.E.; Rutten, E.P.; Wouters, E.F.; Spruit, M.A. Heterogeneity in clinical characteristics and co-morbidities in dyspneic individuals with COPD GOLD D: Findings of the DICES trial. Respir. Med. 2013, 107, 1186–1194.
- Bai, S.; Ye, R.; Wang, C.; Sun, P.; Zhao, L. Comparative analysis of pathophysiological parameters between emphysematous smokers and emphysematous patients with COPD. Sci. Rep. 2020, 10, 420.
- Cavailles, A.; Brinchault-Rabin, G.; Dixmier, A.; Goupil, F.; Gut-Gobert, C.; Marchand-Adam, S.; Meurice, J.C.; Morel, H.; Person-Tacnet, C.; Leroyer, C.; et al. Comorbidities of COPD. Eur. Respir. Rev. 2013, 22, 454–475.
- Raherison, C.; Ouaalaya, E.-H.; Bernady, A.; Casteigt, J.; Nocent-Eijnani, C.; Falque, L.; Le Guillou, F.; Nguyen, L.; Ozier, A.; Molimard, M. Comorbidities and COPD severity in a clinic-based cohort. BMC Pulm. Med. 2018, 18, 117.
- Chandra, D.; Gupta, A.; Strollo, P.J., Jr.; Fuhrman, C.R.; Leader, J.K.; Bon, J.; Slivka, W.A.; Shoushtari, A.H.; Avolio, J.; Kip, K.E.; et al. Airflow Limitation and Endothelial Dysfunction. Unrelated and Independent Predictors of Atherosclerosis. Am. J. Respir. Crit. Care Med. 2016, 194, 38–47.
- Tuleta, I.; Farrag, T.; Busse, L.; Pizarro, C.; Schaefer, C.; Pingel, S.; Nickenig, G.; Skowasch, D.; Schahab, N. High prevalence of COPD in atherosclerosis patients. Int. J. Chron. Obstruct. Pulm. Dis. 2017, 12, 3047–3053.
- Carter, P.; Lagan, J.; Fortune, C.; Bhatt, D.L.; Vestbo, J.; Niven, R.; Chaudhuri, N.; Schelbert, E.B.; Potluri, R.; Miller, C.A. Association of Cardiovascular Disease With Respiratory Disease. J. Am. Coll. Cardiol. 2019, 73, 2166–2177.
- Malo de Molina, R.; Aguado, S.; Arellano, C.; Valle, M.; Ussetti, P. Ischemic Heart Disease during Acute Exacerbations of COPD. Med. Sci. 2018, 6, 83.
- Rothnie, K.J.; Connell, O.; Mullerova, H.; Smeeth, L.; Pearce, N.; Douglas, I.; Quint, J.K. Myocardial Infarction and Ischemic Stroke after Exacerbations of Chronic Obstructive Pulmonary Disease. Ann. Am. Thorac. Soc. 2018, 15, 935–946.
- Milne, K.; Sin, D.D. Acute Exacerbations of Chronic Lung Disease: Cardiac Considerations. In Cardiac Considerations in Chronic Lung Disease; Bhatt, S.P., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 229–245.
- Rafie, S.; Moitra, S.; Brashier, B.B. Association between the Serum Metabolic Profile and Lung Function in Chronic Obstructive Pulmonary Disease. Turk. Thorac. J. 2018, 19, 13–18.
- Can, U.; Yerlikaya, F.H.; Yosunkaya, S. Role of oxidative stress and serum lipid levels in stable chronic obstructive pulmonary disease. J. Chin. Med. Assoc. 2015, 78, 702–708.
- Jimenez-Cepeda, A.; Davila-Said, G.; Orea-Tejeda, A.; Gonzalez-Islas, D.; Elizondo-Montes, M.; Perez-Cortes, G.; Keirns-Davies, C.; Castillo-Aguilar, L.F.; Verdeja-Vendrell, L.; Pelaez-Hernandez, V.; et al. Dietary intake of fatty acids and its relationship with FEV1/FVC in patients with chronic obstructive pulmonary disease. Clin. Nutr. ESPEN 2019, 29, 92–96.
- Morgan, A.D.; Zakeri, R.; Quint, J.K. Defining the relationship between COPD and CVD: What are the implications for clinical practice? Ther. Adv. Respir. Dis. 2018, 12, 1753465817750524.
- Hughes, M.J.; McGettrick, H.M.; Sapey, E. Shared mechanisms of multimorbidity in COPD, atherosclerosis and type-2 diabetes: The neutrophil as a potential inflammatory target. Eur. Respir. Rev. 2020, 29.
- Finkelstein, R.; Fraser, R.S.; Ghezzo, H.; Cosio, M.G. Alveolar inflammation and its relation to emphysema in smokers. Am. J. Respir. Crit. Care Med. 1995, 152, 1666–1672.
- Finkelstein, R.; Ma, H.D.; Ghezzo, H.; Whittaker, K.; Fraser, R.S.; Cosio, M.G. Morphometry of small airways in smokers and its relationship to emphysema type and hyperresponsiveness. Am. J. Respir. Crit. Care Med. 1995, 152, 267–276.
- Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005, 352, 1685–1695.
- Moore, K.J.; Sheedy, F.J.; Fisher, E.A. Macrophages in atherosclerosis: A dynamic balance. Nat. Rev. Immunol. 2013, 13, 709–721.
- Yu, X.H.; Fu, Y.C.; Zhang, D.W.; Yin, K.; Tang, C.K. Foam cells in atherosclerosis. Clin. Chim. Acta 2013, 424, 245–252.
- Agarwal, A.R.; Kadam, S.; Brahme, A.; Agrawal, M.; Apte, K.; Narke, G.; Kekan, K.; Madas, S.; Salvi, S. Systemic Immuno-metabolic alterations in chronic obstructive pulmonary disease (COPD). Respir. Res. 2019, 20, 171.
- Song, D.; Liu, F.; Zhu, B.; Yin, J.; Kuang, Z.; Dong, Z.; Yan, L.; Ye, L.; Zhang, Y.; Song, Z.; et al. Global Immunometabolic Profiling of AECOPD. Small Methods 2020, 4, 2000483.
- Iyer, A.S.; Dransfield, M.T. The “Obesity Paradox” in Chronic Obstructive Pulmonary Disease: Can It Be Resolved? Ann. Am. Thorac. Soc. 2018, 15, 158–159.
- Blum, A.; Simsolo, C.; Sirchan, R.; Haiek, S. “Obesity paradox” in chronic obstructive pulmonary disease. Isr. Med. Assoc. J. 2011, 13, 672–675.
- Yamauchi, Y.; Hasegawa, W.; Yasunaga, H.; Sunohara, M.; Jo, T.; Takami, K.; Matsui, H.; Fushimi, K.; Nagase, T. Paradoxical association between body mass index and in-hospital mortality in elderly patients with chronic obstructive pulmonary disease in Japan. Int. J. Chron. Obstr. Pulm. Dis. 2014, 9, 1337–1346.
- Wada, H.; Ikeda, A.; Maruyama, K.; Yamagishi, K.; Barnes, P.J.; Tanigawa, T.; Tamakoshi, A.; Iso, H. Low BMI and weight loss aggravate COPD mortality in men, findings from a large prospective cohort: The JACC study. Sci. Rep. 2021, 11, 1531.
- Bernardi, S.; Marcuzzi, A.; Piscianz, E.; Tommasini, A.; Fabris, B. The Complex Interplay between Lipids, Immune System and Interleukins in Cardio-Metabolic Diseases. Int. J. Mol. Sci. 2018, 19, 4058.
- ter Beek, J.; Guskov, A.; Slotboom, D.J. Structural diversity of ABC transporters. J. Gen. Physiol. 2014, 143, 419–435.
- Ye, Z.; Lu, Y.; Wu, T. The impact of ATP-binding cassette transporters on metabolic diseases. Nutr. Metab. 2020, 17, 61.
- Dean, M.; Hamon, Y.; Chimini, G. The human ATP-binding cassette (ABC) transporter superfamily. J. Lipid Res. 2001, 42, 1007–1017.
- Phillips, M.C. Is ABCA1 a lipid transfer protein? J. Lipid Res. 2018, 59, 749–763.
- Jones, P.M.; George, A.M. The ABC transporter structure and mechanism: Perspectives on recent research. Cell. Mol. Life Sci. 2004, 61, 682–699.
- Theodoulou, F.L.; Kerr, I.D. ABC transporter research: Going strong 40 years on. Biochem. Soc. Trans. 2015, 43, 1033–1040.
- Tachikawa, M.; Watanabe, M.; Hori, S.; Fukaya, M.; Ohtsuki, S.; Asashima, T.; Terasaki, T. Distinct spatio-temporal expression of ABCA and ABCG transporters in the developing and adult mouse brain. J. Neurochem. 2005, 95, 294–304.
- Tarling, E.J.; de Aguiar Vallim, T.Q.; Edwards, P.A. Role of ABC transporters in lipid transport and human disease. Trends Endocrinol. Metab. 2013, 24, 342–350.
- Neumann, J.; Rose-Sperling, D.; Hellmich, U.A. Diverse relations between ABC transporters and lipids: An overview. Biochim. Biophys. Acta Biomembr. 2017, 1859, 605–618.
- Barreto-Ojeda, E.; Corradi, V.; Gu, R.X.; Tieleman, D.P. Coarse-grained molecular dynamics simulations reveal lipid access pathways in P-glycoprotein. J. Gen. Physiol. 2018, 150, 417–429.
- Song, Y.; Kenworthy, A.K.; Sanders, C.R. Cholesterol as a co-solvent and a ligand for membrane proteins. Protein Sci. 2014, 23, 1–22.
- Fantini, J.; Barrantes, F.J. How cholesterol interacts with membrane proteins: An exploration of cholesterol-binding sites including CRAC, CARC, and tilted domains. Front. Physiol. 2013, 4, 31.
- Fantini, J.; Barrantes, F.J. Sphingolipid/cholesterol regulation of neurotransmitter receptor conformation and function. Biochim. Biophys. Acta 2009, 1788, 2345–2361.
- Fantini, J.; Epand, R.M.; Barrantes, F.J. Cholesterol-Recognition Motifs in Membrane Proteins. Adv. Exp. Med. Biol 2019, 1135, 3–25.
- Sharom, F.J. The P-glycoprotein efflux pump: How does it transport drugs? J. Membr. Biol. 1997, 160, 161–175.
- Sharom, F.J. Complex Interplay between the P-Glycoprotein Multidrug Efflux Pump and the Membrane: Its Role in Modulating Protein Function. Front. Oncol. 2014, 4, 41.
- Linton, K.J. Lipid flopping in the liver. Biochem. Soc. Trans. 2015, 43, 1003–1010.
- Zhao, Y.; Ishigami, M.; Nagao, K.; Hanada, K.; Kono, N.; Arai, H.; Matsuo, M.; Kioka, N.; Ueda, K. ABCB4 exports phosphatidylcholine in a sphingomyelin-dependent manner. J. Lipid Res. 2015, 56, 644–652.
- Riordan, J.R.; Deuchars, K.; Kartner, N.; Alon, N.; Trent, J.; Ling, V. Amplification of P-glycoprotein genes in multidrug-resistant mammalian cell lines. Nature 1985, 316, 817–819.
- Cario, E. P-glycoprotein multidrug transporter in inflammatory bowel diseases: More questions than answers. World J. Gastroenterol. 2017, 23, 1513–1520.
- Juliano, R.L.; Ling, V. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim. Biophys. Acta 1976, 455, 152–162.
- Ambudkar, S.V.; Kimchi-Sarfaty, C.; Sauna, Z.E.; Gottesman, M.M. P-glycoprotein: From genomics to mechanism. Oncogene 2003, 22, 7468–7485.
- Chufan, E.E.; Sim, H.M.; Ambudkar, S.V. Molecular basis of the polyspecificity of P-glycoprotein (ABCB1): Recent biochemical and structural studies. Adv. Cancer Res. 2015, 125, 71–96.
- Subramanian, N.; Condic-Jurkic, K.; O’Mara, M.L. Structural and dynamic perspectives on the promiscuous transport activity of P-glycoprotein. Neurochem. Int. 2016, 98, 146–152.
- Higgins, C.F.; Gottesman, M.M. Is the multidrug transporter a flippase? Trends Biochem. Sci. 1992, 17, 18–21.
- Ruysschaert, J.M.; Lonez, C. Role of lipid microdomains in TLR-mediated signalling. Biochim. Biophys. Acta 2015, 1848, 1860–1867.
- van Helvoort, A.; Smith, A.J.; Sprong, H.; Fritzsche, I.; Schinkel, A.H.; Borst, P.; van Meer, G. MDR1 P-glycoprotein is a lipid translocase of broad specificity, while MDR3 P-glycoprotein specifically translocates phosphatidylcholine. Cell 1996, 87, 507–517.
- Kimura, Y.; Kioka, N.; Kato, H.; Matsuo, M.; Ueda, K. Modulation of drug-stimulated ATPase activity of human MDR1/P-glycoprotein by cholesterol. Biochem. J. 2007, 401, 597–605.
- Garrigues, A.; Escargueil, A.E.; Orlowski, S. The multidrug transporter, P-glycoprotein, actively mediates cholesterol redistribution in the cell membrane. Proc. Natl. Acad. Sci. USA 2002, 99, 10347–10352.
- Clay, A.T.; Lu, P.; Sharom, F.J. Interaction of the P-Glycoprotein Multidrug Transporter with Sterols. Biochemistry 2015, 54, 6586–6597.
- Bosch, I.; Dunussi-Joannopoulos, K.; Wu, R.L.; Furlong, S.T.; Croop, J. Phosphatidylcholine and phosphatidylethanolamine behave as substrates of the human MDR1 P-glycoprotein. Biochemistry 1997, 36, 5685–5694.
- Bossennec, M.; Di Roio, A.; Caux, C.; Ménétrier-Caux, C. MDR1 in immunity: Friend or foe? OncoImmunology 2018, 7, e1499388.
- Rothnie, A.; Theron, D.; Soceneantu, L.; Martin, C.; Traikia, M.; Berridge, G.; Higgins, C.F.; Devaux, P.F.; Callaghan, R. The importance of cholesterol in maintenance of P-glycoprotein activity and its membrane perturbing influence. Eur. Biophys. J. 2001, 30, 430–442.
- Troost, J.; Lindenmaier, H.; Haefeli, W.E.; Weiss, J. Modulation of cellular cholesterol alters P-glycoprotein activity in multidrug-resistant cells. Mol. Pharmacol. 2004, 66, 1332–1339.
- Luker, G.D.; Pica, C.M.; Kumar, A.S.; Covey, D.F.; Piwnica-Worms, D. Effects of cholesterol and enantiomeric cholesterol on P-glycoprotein localization and function in low-density membrane domains. Biochemistry 2000, 39, 7651–7661.
- Li, H.; Papadopoulos, V. Peripheral-type benzodiazepine receptor function in cholesterol transport. Identification of a putative cholesterol recognition/interaction amino acid sequence and consensus pattern. Endocrinology 1998, 139, 4991–4997.
- Baier, C.J.; Fantini, J.; Barrantes, F.J. Disclosure of cholesterol recognition motifs in transmembrane domains of the human nicotinic acetylcholine receptor. Sci. Rep. 2011, 1, 69.
- Lechapt-Zalcman, E.; Hurbain, I.; Lacave, R.; Commo, F.; Urban, T.; Antoine, M.; Milleron, B.; Bernaudin, J.F. MDR1-Pgp 170 expression in human bronchus. Eur. Respir. J. 1997, 10, 1837–1843.
- Berg, T.; Hegelund-Myrbäck, T.; Öckinger, J.; Zhou, X.-H.; Brännström, M.; Hagemann-Jensen, M.; Werkström, V.; Seidegård, J.; Grunewald, J.; Nord, M.; et al. Expression of MATE1, P-gp, OCTN1 and OCTN2, in epithelial and immune cells in the lung of COPD and healthy individuals. Respir. Res. 2018, 19, 68.
- Campbell, L.; Abulrob, A.-N.G.; Kandalaft, L.E.; Plummer, S.; Hollins, A.J.; Gibbs, A.; Gumbleton, M. Constitutive expression of p-glycoprotein in normal lung alveolar epithelium and functionality in primary alveolar epithelial cultures. J. Pharmacol. Exp. Ther. 2003, 304, 441–452.
- Takano, M.; Naka, R.; Sasaki, Y.; Nishimoto, S.; Yumoto, R. Effect of cigarette smoke extract on P-glycoprotein function in primary cultured and newly developed alveolar epithelial cells. Drug Metab. Pharmacokinet. 2016, 31, 417–424.
- Langmann, T.; Mauerer, R.; Schmitz, G. Human ATP-binding cassette transporter TaqMan low-density array: Analysis of macrophage differentiation and foam cell formation. Clin. Chem. 2006, 52, 310–313.
- Pennings, M.; Meurs, I.; Ye, D.; Out, R.; Hoekstra, M.; Van Berkel, T.J.; Van Eck, M. Regulation of cholesterol homeostasis in macrophages and consequences for atherosclerotic lesion development. FEBS Lett. 2006, 580, 5588–5596.
- Cory, T.J.; He, H.; Winchester, L.C.; Kumar, S.; Fletcher, C.V. Alterations in P-Glycoprotein Expression and Function Between Macrophage Subsets. Pharm. Res. 2016, 33, 2713–2721.
- van der Deen, M.; de Vries, E.G.E.; Timens, W.; Scheper, R.J.; Timmer-Bosscha, H.; Postma, D.S. ATP-binding cassette (ABC) transporters in normal and pathological lung. Respir. Res. 2005, 6, 59.
- Langmann, T.; Mauerer, R.; Zahn, A.; Moehle, C.; Probst, M.; Stremmel, W.; Schmitz, G. Real-time reverse transcription-PCR expression profiling of the complete human ATP-binding cassette transporter superfamily in various tissues. Clin. Chem. 2003, 49, 230–238.
- Pan, W.C.; Chen, R.M.; Shen, Y.C.; Chen, C.C.; Ueng, Y.F. Suppressive effect of tobacco smoke extracts on oral P-glycoprotein function and its impact in smoke-induced insult to oral epidermal cells. Toxicol. Lett. 2009, 185, 116–123.
- Berg, T.; Hegelund Myrbäck, T.; Olsson, M.; Seidegård, J.; Werkström, V.; Zhou, X.-H.; Grunewald, J.; Gustavsson, L.; Nord, M. Gene expression analysis of membrane transporters and drug-metabolizing enzymes in the lung of healthy and COPD subjects. Pharmacol. Res. Perspect. 2014, 2, e00054.
- Wang, X.W.; Zhang, Y.L.; Xiong, Y. Impact of ABCB1 single-nucleotide polymorphisms on treatment outcomes with salmeterol/fluticasone combination therapy for stable chronic obstructive pulmonary disease. Genet. Test. Mol. Biomark. 2015, 19, 566–572.
- Yucel, O.; Gunes, H.; Yucel, H.; Zorlu, A. Association between multidrug resistance-1 C3435T gene polymorphism and right ventricular dysfunction in patients with chronic obstructive pulmonary disease: Cross-sectional study. Sao Paulo Med. J. 2018, 136, 140–143.
- Batetta, B.; Mulas, M.F.; Petruzzo, P.; Putzolu, M.; Bonatesta, R.R.; Sanna, F.; Cappai, A.; Brotzu, G.; Dessi, S. Opposite pattern of MDR1 and caveolin-1 gene expression in human atherosclerotic lesions and proliferating human smooth muscle cells. Cell. Mol. Life Sci. 2001, 58, 1113–1120.
- Klucken, J.; Buchler, C.; Orso, E.; Kaminski, W.E.; Porsch-Ozcurumez, M.; Liebisch, G.; Kapinsky, M.; Diederich, W.; Drobnik, W.; Dean, M.; et al. ABCG1 (ABC8), the human homolog of the Drosophila white gene, is a regulator of macrophage cholesterol and phospholipid transport. Proc. Natl. Acad. Sci. USA 2000, 97, 817–822.
- Pennings, M.; Hildebrand, R.B.; Ye, D.; Kunne, C.; Van Berkel, T.J.; Groen, A.K.; Van Eck, M. Bone marrow-derived multidrug resistance protein ABCB4 protects against atherosclerotic lesion development in LDL receptor knockout mice. Cardiovasc. Res. 2007, 76, 175–183.
- Voshol, P.J.; Havinga, R.; Wolters, H.; Ottenhoff, R.; Princen, H.M.; Oude Elferink, R.P.; Groen, A.K.; Kuipers, F. Reduced plasma cholesterol and increased fecal sterol loss in multidrug resistance gene 2 P-glycoprotein-deficient mice. Gastroenterology 1998, 114, 1024–1034.
- Daleke, D.L. Regulation of transbilayer plasma membrane phospholipid asymmetry. J. Lipid Res. 2003, 44, 233–242.
- Aguiar, J.A.; Tamminga, A.; Lobb, B.; Huff, R.D.; Nguyen, J.P.; Kim, Y.; Dvorkin-Gheva, A.; Stampfli, M.R.; Doxey, A.C.; Hirota, J.A. The impact of cigarette smoke exposure, COPD, or asthma status on ABC transporter gene expression in human airway epithelial cells. Sci. Rep. 2019, 9.
- Chavan, H.; Oruganti, M.; Krishnamurthy, P. The ATP-binding cassette transporter ABCB6 is induced by arsenic and protects against arsenic cytotoxicity. Toxicol. Sci. 2011, 120, 519–528.
- Boswell-Casteel, R.C.; Fukuda, Y.; Schuetz, J.D. ABCB6, an ABC Transporter Impacting Drug Response and Disease. AAPS J. 2017, 20, 8.
- Huo, Y.; Schober, A.; Forlow, S.B.; Smith, D.F.; Hyman, M.C.; Jung, S.; Littman, D.R.; Weber, C.; Ley, K. Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nat. Med. 2003, 9, 61–67.
- Wang, N.; Tall, A.R. Cholesterol in platelet biogenesis and activation. Blood 2016, 127, 1949–1953.
- Koenen, R.R.; von Hundelshausen, P.; Nesmelova, I.V.; Zernecke, A.; Liehn, E.A.; Sarabi, A.; Kramp, B.K.; Piccinini, A.M.; Paludan, S.R.; Kowalska, M.A.; et al. Disrupting functional interactions between platelet chemokines inhibits atherosclerosis in hyperlipidemic mice. Nat. Med. 2009, 15, 97–103.
- Sreeramkumar, V.; Adrover, J.M.; Ballesteros, I.; Cuartero, M.I.; Rossaint, J.; Bilbao, I.; Nacher, M.; Pitaval, C.; Radovanovic, I.; Fukui, Y.; et al. Neutrophils scan for activated platelets to initiate inflammation. Science 2014, 346, 1234–1238.
- Schrottmaier, W.C.; Kral, J.B.; Badrnya, S.; Assinger, A. Aspirin and P2Y12 Inhibitors in platelet-mediated activation of neutrophils and monocytes. Thromb. Haemost. 2015, 114, 478–489.
- Soehnlein, O. The ABC of Thrombopoiesis. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 700–701.
- Cole, S.P.; Deeley, R.G. Transport of glutathione and glutathione conjugates by MRP1. Trends Pharmacol. Sci. 2006, 27, 438–446.
- Leier, I.; Jedlitschky, G.; Buchholz, U.; Cole, S.; Deeley, R.G.; Keppler, D. The MRP gene encodes an ATP-dependent export pump for leukotriene C4 and structurally related conjugates. J. Biol. Chem. 1994, 269, 27807–27810.
- Raggers, R.J.; van Helvoort, A.; Evers, R.; van Meer, G. The human multidrug resistance protein MRP1 translocates sphingolipid analogs across the plasma membrane. J. Cell Sci. 1999, 112, 415–422.
- Nooter, K.; Bosman, F.T.; Burger, H.; van Wingerden, K.E.; Flens, M.J.; Scheper, R.J.; Oostrum, R.G.; Boersma, A.W.; van der Gaast, A.; Stoter, G. Expression of the multidrug resistance-associated protein (MRP) gene in primary non-small-cell lung cancer. Ann. Oncol. 1996, 7, 75–81.
- Selo, M.A.; Delmas, A.-S.; Springer, L.; Zoufal, V.; Sake, J.A.; Clerkin, C.G.; Huwer, H.; Schneider-Daum, N.; Lehr, C.-M.; Nickel, S.; et al. Tobacco Smoke and Inhaled Drugs Alter Expression and Activity of Multidrug Resistance-Associated Protein-1 (MRP1) in Human Distal Lung Epithelial Cells in vitro. Front. Bioeng. Biotechnol. 2020, 8, 1030.
- Bréchot, J.M.; Hurbain, I.; Fajac, A.; Daty, N.; Bernaudin, J.F. Different pattern of MRP localization in ciliated and basal cells from human bronchial epithelium. J. Histochem. Cytochem. 1998, 46, 513–517.
- Scheffer, G.L.; Pijnenborg, A.C.; Smit, E.F.; Müller, M.; Postma, D.S.; Timens, W.; van der Valk, P.; de Vries, E.G.; Scheper, R.J. Multidrug resistance related molecules in human and murine lung. J. Clin. Pathol. 2002, 55, 332–339.
- Marbeuf-Gueye, C.; Stierle, V.; Sudwan, P.; Salerno, M.; Garnier-Suillerot, A. Perturbation of membrane microdomains in GLC4 multidrug-resistant lung cancer cells—Modification of ABCC1 (MRP1) localization and functionality. FEBS J. 2007, 274, 1470–1480.
- Meszaros, P.; Klappe, K.; Hummel, I.; Hoekstra, D.; Kok, J.W. Function of MRP1/ABCC1 is not dependent on cholesterol or cholesterol-stabilized lipid rafts. Biochem. J. 2011, 437, 483–491.
- Leslie, E.M.; Deeley, R.G.; Cole, S.P. Multidrug resistance proteins: Role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicol. Appl. Pharmacol. 2005, 204, 216–237.
- Zeng, H.; Liu, G.; Rea, P.A.; Kruh, G.D. Transport of amphipathic anions by human multidrug resistance protein 3. Cancer Res. 2000, 60, 4779–4784.
- van der Deen, M.; Marks, H.; Willemse, B.W.; Postma, D.S.; Müller, M.; Smit, E.F.; Scheffer, G.L.; Scheper, R.J.; de Vries, E.G.; Timens, W. Diminished expression of multidrug resistance-associated protein 1 (MRP1) in bronchial epithelium of COPD patients. Virchows Arch. 2006, 449, 682–688.
- Bandi, N.; Kompella, U.B. Budesonide reduces multidrug resistance-associated protein 1 expression in an airway epithelial cell line (Calu-1). Eur. J. Pharmacol. 2002, 437, 9–17.
- Wijnholds, J.; Evers, R.; van Leusden, M.R.; Mol, C.A.; Zaman, G.J.; Mayer, U.; Beijnen, J.H.; van der Valk, M.; Krimpenfort, P.; Borst, P. Increased sensitivity to anticancer drugs and decreased inflammatory response in mice lacking the multidrug resistance-associated protein. Nat. Med. 1997, 3, 1275–1279.
- Ishikawa, T. The ATP-dependent glutathione S-conjugate export pump. Trends Biochem. Sci. 1992, 17, 463–468.
- Bailie, M.B.; Standiford, T.J.; Laichalk, L.L.; Coffey, M.J.; Strieter, R.; Peters-Golden, M. Leukotriene-deficient mice manifest enhanced lethality from Klebsiella pneumonia in association with decreased alveolar macrophage phagocytic and bactericidal activities. J. Immunol. 1996, 157, 5221–5224.
- Demitsu, T.; Katayama, H.; Saito-Taki, T.; Yaoita, H.; Nakano, M. Phagocytosis and bactericidal action of mouse peritoneal macrophages treated with leukotriene B4. Int. J. Immunopharmacol. 1989, 11, 801–808.
- Funk, C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science 2001, 294, 1871–1875.
- van der Deen, M.; Timens, W.; Timmer-Bosscha, H.; van der Strate, B.W.; Scheper, R.J.; Postma, D.S.; de Vries, E.G.; Kerstjens, H.A. Reduced inflammatory response in cigarette smoke exposed Mrp1/Mdr1a/1b deficient mice. Respir. Res. 2007, 8, 49.
- Leier, I.; Jedlitschky, G.; Buchholz, U.; Keppler, D. Characterization of the ATP-dependent leukotriene C4 export carrier in mastocytoma cells. Eur. J. Biochem. 1994, 220, 599–606.
- Low, F.G.; Shabir, K.; Brown, J.E.; Bill, R.M.; Rothnie, A.J. Roles of ABCC1 and ABCC4 in Proliferation and Migration of Breast Cancer Cell Lines. Int. J. Mol. Sci. 2020, 21, 7664.
- Weigert, A.; Weis, N.; Brüne, B. Regulation of macrophage function by sphingosine-1-phosphate. Immunobiology 2009, 214, 748–760.
- Takabe, K.; Kim, R.H.; Allegood, J.C.; Mitra, P.; Ramachandran, S.; Nagahashi, M.; Harikumar, K.B.; Hait, N.C.; Milstien, S.; Spiegel, S. Estradiol induces export of sphingosine 1-phosphate from breast cancer cells via ABCC1 and ABCG2. J. Biol. Chem. 2010, 285, 10477–10486.
- Yamada, A.; Nagahashi, M.; Aoyagi, T.; Huang, W.-C.; Lima, S.; Hait, N.C.; Maiti, A.; Kida, K.; Terracina, K.P.; Miyazaki, H.; et al. ABCC1-Exported Sphingosine-1-phosphate, Produced by Sphingosine Kinase 1, Shortens Survival of Mice and Patients with Breast Cancer. Mol. Cancer Res. MCR 2018, 16, 1059–1070.
- Nagahashi, M.; Takabe, K.; Terracina, K.P.; Soma, D.; Hirose, Y.; Kobayashi, T.; Matsuda, Y.; Wakai, T. Sphingosine-1-Phosphate Transporters as Targets for Cancer Therapy. BioMed Res. Int. 2014, 2014, 651727.
- Garcia, J.G.; Liu, F.; Verin, A.D.; Birukova, A.; Dechert, M.A.; Gerthoffer, W.T.; Bamberg, J.R.; English, D. Sphingosine 1-phosphate promotes endothelial cell barrier integrity by Edg-dependent cytoskeletal rearrangement. J. Clin. Investig. 2001, 108, 689–701.
- Olivera, A.; Spiegel, S. Sphingosine kinase: A mediator of vital cellular functions. Prostaglandins Other Lipid Mediat. 2001, 64, 123–134.
- Pyne, S.; Pyne, N.J. Sphingosine 1-phosphate signalling and termination at lipid phosphate receptors. Biochim. Biophys. Acta 2002, 1582, 121–131.
- Spiegel, S.; Milstien, S. Exogenous and intracellularly generated sphingosine 1-phosphate can regulate cellular processes by divergent pathways. Biochem. Soc. Trans. 2003, 31, 1216–1219.
- Spiegel, S.; Milstien, S. Sphingosine-1-phosphate: An enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 2003, 4, 397–407.
- Usatyuk, P.V.; He, D.; Bindokas, V.; Gorshkova, I.A.; Berdyshev, E.V.; Garcia, J.G.; Natarajan, V. Photolysis of caged sphingosine-1-phosphate induces barrier enhancement and intracellular activation of lung endothelial cell signaling pathways. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 300, L840–L850.
- Guney, T.G.; Holloway, R.; Taylor, A.; Thomas, C.; Barnes, P.; Donnelly, L. Increased expression of sphingosine-1-phosphate and S1PR2 are associated with attenuated macrophage phagocytosis in COPD. Eur. Respir. J. 2013, 42, P626.
- Barnawi, J.; Tran, H.; Jersmann, H.; Pitson, S.; Roscioli, E.; Hodge, G.; Meech, R.; Haberberger, R.; Hodge, S. Potential Link between the Sphingosine-1-Phosphate (S1P) System and Defective Alveolar Macrophage Phagocytic Function in Chronic Obstructive Pulmonary Disease (COPD). PLoS ONE 2015, 10, e0122771.
- Berdyshev, E.V.; Serban, K.A.; Schweitzer, K.S.; Bronova, I.A.; Mikosz, A.; Petrache, I. Ceramide and sphingosine-1 phosphate in COPD lungs. Thorax 2021.
- Sato, K.; Malchinkhuu, E.; Horiuchi, Y.; Mogi, C.; Tomura, H.; Tosaka, M.; Yoshimoto, Y.; Kuwabara, A.; Okajima, F. Critical role of ABCA1 transporter in sphingosine 1-phosphate release from astrocytes. J. Neurochem. 2007, 103, 2610–2619.
- Liu, X.; Ren, K.; Suo, R.; Xiong, S.-L.; Zhang, Q.-H.; Mo, Z.-C.; Tang, Z.-L.; Jiang, Y.; Peng, X.-S.; Yi, G.-H. ApoA-I induces S1P release from endothelial cells through ABCA1 and SR-BI in a positive feedback manner. J. Physiol. Biochem. 2016, 72, 657–667.
- Mueller, C.F.; Widder, J.D.; McNally, J.S.; McCann, L.; Jones, D.P.; Harrison, D.G. The role of the multidrug resistance protein-1 in modulation of endothelial cell oxidative stress. Circ. Res. 2005, 97, 637–644.
- Widder, J.D.; Guzik, T.J.; Mueller, C.F.; Clempus, R.E.; Schmidt, H.H.; Dikalov, S.I.; Griendling, K.K.; Jones, D.P.; Harrison, D.G. Role of the multidrug resistance protein-1 in hypertension and vascular dysfunction caused by angiotensin II. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 762–768.
- Hirrlinger, J.; Konig, J.; Keppler, D.; Lindenau, J.; Schulz, J.B.; Dringen, R. The multidrug resistance protein MRP1 mediates the release of glutathione disulfide from rat astrocytes during oxidative stress. J. Neurochem. 2001, 76, 627–636.
- Mueller, C.F.; Wassmann, K.; Widder, J.D.; Wassmann, S.; Chen, C.H.; Keuler, B.; Kudin, A.; Kunz, W.S.; Nickenig, G. Multidrug resistance protein-1 affects oxidative stress, endothelial dysfunction, and atherogenesis via leukotriene C4 export. Circulation 2008, 117, 2912–2918.
- Khunweeraphong, N.; Mitchell-White, J.; Szöllősi, D.; Hussein, T.; Kuchler, K.; Kerr, I.D.; Stockner, T.; Lee, J.-Y. Picky ABCG5/G8 and promiscuous ABCG2—A tale of fatty diets and drug toxicity. FEBS Lett. 2020, 594, 4035–4058.
- Robey, R.W.; To, K.K.; Polgar, O.; Dohse, M.; Fetsch, P.; Dean, M.; Bates, S.E. ABCG2: A perspective. Adv. Drug Deliv. Rev. 2009, 61, 3–13.
- Kobayashi, A.; Takanezawa, Y.; Hirata, T.; Shimizu, Y.; Misasa, K.; Kioka, N.; Arai, H.; Ueda, K.; Matsuo, M. Efflux of sphingomyelin, cholesterol, and phosphatidylcholine by ABCG1. J. Lipid Res. 2006, 47, 1791–1802.
- Cserepes, J.; Szentpetery, Z.; Seres, L.; Ozvegy-Laczka, C.; Langmann, T.; Schmitz, G.; Glavinas, H.; Klein, I.; Homolya, L.; Varadi, A.; et al. Functional expression and characterization of the human ABCG1 and ABCG4 proteins: Indications for heterodimerization. Biochem. Biophys. Res. Commun. 2004, 320, 860–867.
- Sano, O.; Ito, S.; Kato, R.; Shimizu, Y.; Kobayashi, A.; Kimura, Y.; Kioka, N.; Hanada, K.; Ueda, K.; Matsuo, M. ABCA1, ABCG1, and ABCG4 are distributed to distinct membrane meso-domains and disturb detergent-resistant domains on the plasma membrane. PLoS ONE 2014, 9, e109886.
- Wang, N.; Lan, D.; Chen, W.; Matsuura, F.; Tall, A.R. ATP-binding cassette transporters G1 and G4 mediate cellular cholesterol efflux to high-density lipoproteins. Proc. Natl. Acad. Sci. USA 2004, 101, 9774–9779.
- Kennedy, M.A.; Barrera, G.C.; Nakamura, K.; Baldan, A.; Tarr, P.; Fishbein, M.C.; Frank, J.; Francone, O.L.; Edwards, P.A. ABCG1 has a critical role in mediating cholesterol efflux to HDL and preventing cellular lipid accumulation. Cell Metab. 2005, 1, 121–131.
- Nakamura, K.; Kennedy, M.A.; Baldán, A.; Bojanic, D.D.; Lyons, K.; Edwards, P.A. Expression and regulation of multiple murine ATP-binding cassette transporter G1 mRNAs/isoforms that stimulate cellular cholesterol efflux to high density lipoprotein. J. Biol. Chem. 2004, 279, 45980–45989.
- Wang, N.; Yvan-Charvet, L.; Lütjohann, D.; Mulder, M.; Vanmierlo, T.; Kim, T.W.; Tall, A.R. ATP-binding cassette transporters G1 and G4 mediate cholesterol and desmosterol efflux to HDL and regulate sterol accumulation in the brain. FASEB J. 2008, 22, 1073–1082.
- Tarr, P.T.; Tarling, E.J.; Bojanic, D.D.; Edwards, P.A.; Baldán, Á. Emerging new paradigms for ABCG transporters. Biochim. Biophys. Acta BBA Mol. Cell Biol. Lipids 2009, 1791, 584–593.
- Bensinger, S.J.; Bradley, M.N.; Joseph, S.B.; Zelcer, N.; Janssen, E.M.; Hausner, M.A.; Shih, R.; Parks, J.S.; Edwards, P.A.; Jamieson, B.D.; et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 2008, 134, 97–111.
- Out, R.; Hoekstra, M.; Habets, K.; Meurs, I.; de Waard, V.; Hildebrand, R.B.; Wang, Y.; Chimini, G.; Kuiper, J.; Van Berkel, T.J.; et al. Combined deletion of macrophage ABCA1 and ABCG1 leads to massive lipid accumulation in tissue macrophages and distinct atherosclerosis at relatively low plasma cholesterol levels. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 258–264.
- Draper, D.W.; Gowdy, K.M.; Madenspacher, J.H.; Wilson, R.H.; Whitehead, G.S.; Nakano, H.; Pandiri, A.R.; Foley, J.F.; Remaley, A.T.; Cook, D.N.; et al. ATP binding cassette transporter G1 deletion induces IL-17-dependent dysregulation of pulmonary adaptive immunity. J. Immunol. 2012, 188, 5327–5336.
- Baldan, A.; Gonen, A.; Choung, C.; Que, X.; Marquart, T.J.; Hernandez, I.; Bjorkhem, I.; Ford, D.A.; Witztum, J.L.; Tarling, E.J. ABCG1 is required for pulmonary B-1 B cell and natural antibody homeostasis. J. Immunol. 2014, 193, 5637–5648.
- Chai, A.B.; Ammit, A.J.; Gelissen, I.C. Examining the role of ABC lipid transporters in pulmonary lipid homeostasis and inflammation. Respir. Res. 2017, 18, 41.
- Baldán, Á.; Gomes, A.V.; Ping, P.; Edwards, P.A. Loss of ABCG1 Results in Chronic Pulmonary Inflammation. J. Immunol. 2008, 180, 3560–3568.
- Wojcik, A.J.; Skaflen, M.D.; Srinivasan, S.; Hedrick, C.C. A Critical Role for ABCG1 in Macrophage Inflammation and Lung Homeostasis. J. Immunol. 2008, 180, 4273–4282.
- Karakioulaki, M.; Papakonstantinou, E.; Stolz, D. Extracellular matrix remodelling in COPD. Eur. Respir. Rev. 2020, 29, 190124.
- Ilumets, H.; Rytilä, P.; Demedts, I.; Brusselle, G.G.; Sovijärvi, A.; Myllärniemi, M.; Sorsa, T.; Kinnula, V.L. Matrix metalloproteinases -8, -9 and -12 in smokers and patients with stage 0 COPD. Int. J. Chronic Obstr. Pulm. Dis. 2007, 2, 369–379.
- Sag, D.; Purcu, D.U.; Altunay, M. The cholesterol transporter ABCG1 modulates macrophage polarization in human monocyte-derived macrophages. J. Immunol. 2019, 202, 187.
- Sag, D.; Cekic, C.; Wu, R.; Linden, J.; Hedrick, C.C. The cholesterol transporter ABCG1 links cholesterol homeostasis and tumour immunity. Nat. Commun. 2015, 6, 6354.
- Seres, L.; Cserepes, J.; Elkind, N.B.; Torocsik, D.; Nagy, L.; Sarkadi, B.; Homolya, L. Functional ABCG1 expression induces apoptosis in macrophages and other cell types. Biochim. Biophys. Acta 2008, 1778, 2378–2387.
- Yvan-Charvet, L.; Pagler, T.A.; Seimon, T.A.; Thorp, E.; Welch, C.L.; Witztum, J.L.; Tabas, I.; Tall, A.R. ABCA1 and ABCG1 protect against oxidative stress-induced macrophage apoptosis during efferocytosis. Circ. Res. 2010, 106, 1861–1869.
- Pommier, A.J.; Alves, G.; Viennois, E.; Bernard, S.; Communal, Y.; Sion, B.; Marceau, G.; Damon, C.; Mouzat, K.; Caira, F.; et al. Liver X Receptor activation downregulates AKT survival signaling in lipid rafts and induces apoptosis of prostate cancer cells. Oncogene 2010, 29, 2712–2723.
- Baldán, Á.; Pei, L.; Lee, R.; Tarr, P.; Tangirala, R.K.; Weinstein, M.M.; Frank, J.; Li, A.C.; Tontonoz, P.; Edwards, P.A. Impaired Development of Atherosclerosis in Hyperlipidemic Ldlr−/− and ApoE−/− Mice Transplanted With Abcg1−/− Bone Marrow. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2301–2307.
- Tarling, E.J.; Bojanic, D.D.; Tangirala, R.K.; Wang, X.; Lovgren-Sandblom, A.; Lusis, A.J.; Bjorkhem, I.; Edwards, P.A. Impaired development of atherosclerosis in Abcg1−/− Apoe−/− mice: Identification of specific oxysterols that both accumulate in Abcg1−/− Apoe−/− tissues and induce apoptosis. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1174–1180.
- Lorkowski, S.; Kratz, M.; Wenner, C.; Schmidt, R.; Weitkamp, B.; Fobker, M.; Reinhardt, J.; Rauterberg, J.; Galinski, E.A.; Cullen, P. Expression of the ATP-binding cassette transporter gene ABCG1 (ABC8) in Tangier disease. Biochem. Biophys. Res. Commun. 2001, 283, 821–830.
- Venkateswaran, A.; Laffitte, B.A.; Joseph, S.B.; Mak, P.A.; Wilpitz, D.C.; Edwards, P.A.; Tontonoz, P. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXR alpha. Proc. Natl. Acad. Sci. USA 2000, 97, 12097–12102.
- Baldan, A.; Tarr, P.; Lee, R.; Edwards, P.A. ATP-binding cassette transporter G1 and lipid homeostasis. Curr. Opin. Lipidol. 2006, 17, 227–232.
- Venkateswaran, A.; Repa, J.J.; Lobaccaro, J.M.; Bronson, A.; Mangelsdorf, D.J.; Edwards, P.A. Human white/murine ABC8 mRNA levels are highly induced in lipid-loaded macrophages. A transcriptional role for specific oxysterols. J. Biol. Chem. 2000, 275, 14700–14707.
- Sturek, J.M.; Castle, J.D.; Trace, A.P.; Page, L.C.; Castle, A.M.; Evans-Molina, C.; Parks, J.S.; Mirmira, R.G.; Hedrick, C.C. An intracellular role for ABCG1-mediated cholesterol transport in the regulated secretory pathway of mouse pancreatic β cells. J. Clin. Investig. 2010, 120, 2575–2589.
- Tarling, E.J.; Edwards, P.A. ATP binding cassette transporter G1 (ABCG1) is an intracellular sterol transporter. Proc. Natl. Acad. Sci. USA 2011, 108, 19719–19724.
- Annilo, T.; Tammur, J.; Hutchinson, A.; Rzhetsky, A.; Dean, M.; Allikmets, R. Human and mouse orthologs of a new ATP-binding cassette gene, ABCG4. Cytogenet. Cell Genet. 2001, 94, 196–201.
- Oldfield, S.; Lowry, C.; Ruddick, J.; Lightman, S. ABCG4: A novel human white family ABC-transporter expressed in the brain and eye. Biochim. Biophys. Acta 2002, 1591, 175–179.
- Tarr, P.T.; Edwards, P.A. ABCG1 and ABCG4 are coexpressed in neurons and astrocytes of the CNS and regulate cholesterol homeostasis through SREBP-2. J. Lipid Res. 2008, 49, 169–182.
- Bojanic, D.D.; Tarr, P.T.; Gale, G.D.; Smith, D.J.; Bok, D.; Chen, B.; Nusinowitz, S.; Lövgren-Sandblom, A.; Björkhem, I.; Edwards, P.A. Differential expression and function of ABCG1 and ABCG4 during development and aging. J. Lipid Res. 2010, 51, 169–181.
- Terasaka, N.; Wang, N.; Yvan-Charvet, L.; Tall, A.R. High-density lipoprotein protects macrophages from oxidized low-density lipoprotein-induced apoptosis by promoting efflux of 7-ketocholesterol via ABCG1. Proc. Natl. Acad. Sci. USA 2007, 104, 15093–15098.
- Vaughan, A.M.; Oram, J.F. ABCA1 redistributes membrane cholesterol independent of apolipoprotein interactions. J. Lipid Res. 2003, 44, 1373–1380.
- Vaughan, A.M.; Oram, J.F. ABCG1 redistributes cell cholesterol to domains removable by high density lipoprotein but not by lipid-depleted apolipoproteins. J. Biol. Chem. 2005, 280, 30150–30157.
- Kerr, I.D.; Hutchison, E.; Gerard, L.; Aleidi, S.M.; Gelissen, I.C. Mammalian ABCG-transporters, sterols and lipids: To bind perchance to transport? Biochim. Biophys. Acta BBA Mol. Cell Biol. Lipids 2021, 1866, 158860.
- Murphy, A.J.; Bijl, N.; Yvan-Charvet, L.; Welch, C.B.; Bhagwat, N.; Reheman, A.; Wang, Y.; Shaw, J.A.; Levine, R.L.; Ni, H.; et al. Cholesterol efflux in megakaryocyte progenitors suppresses platelet production and thrombocytosis. Nat. Med. 2013, 19, 586–594.
- Westerterp, M.; Bochem, A.E.; Yvan-Charvet, L.; Murphy, A.J.; Wang, N.; Tall, A.R. ATP-binding cassette transporters, atherosclerosis, and inflammation. Circ. Res. 2014, 114, 157–170.
- Homolya, L. Medically Important Alterations in Transport Function and Trafficking of ABCG2. Int. J. Mol. Sci. 2021, 22, 2786.
- Maliepaard, M.; Scheffer, G.L.; Faneyte, I.F.; van Gastelen, M.A.; Pijnenborg, A.C.; Schinkel, A.H.; van De Vijver, M.J.; Scheper, R.J.; Schellens, J.H. Subcellular localization and distribution of the breast cancer resistance protein transporter in normal human tissues. Cancer Res. 2001, 61, 3458–3464.
- Liang, S.C.; Yang, C.Y.; Tseng, J.Y.; Wang, H.L.; Tung, C.Y.; Liu, H.W.; Chen, C.Y.; Yeh, Y.C.; Chou, T.Y.; Yang, M.H.; et al. ABCG2 localizes to the nucleus and modulates CDH1 expression in lung cancer cells. Neoplasia 2015, 17, 265–278.
- Sung, J.M.; Cho, H.J.; Yi, H.; Lee, C.H.; Kim, H.S.; Kim, D.K.; Abd El-Aty, A.M.; Kim, J.S.; Landowski, C.P.; Hediger, M.A.; et al. Characterization of a stem cell population in lung cancer A549 cells. Biochem. Biophys. Res. Commun. 2008, 371, 163–167.
- Scharenberg, C.W.; Harkey, M.A.; Torok-Storb, B. The ABCG2 transporter is an efficient Hoechst 33342 efflux pump and is preferentially expressed by immature human hematopoietic progenitors. Blood 2002, 99, 507–512.
- Singh, A.; Wu, H.; Zhang, P.; Happel, C.; Ma, J.; Biswal, S. Expression of ABCG2 (BCRP) is regulated by Nrf2 in cancer cells that confers side population and chemoresistance phenotype. Mol. Cancer Ther. 2010, 9, 2365–2376.
- Hirschmann-Jax, C.; Foster, A.E.; Wulf, G.G.; Nuchtern, J.G.; Jax, T.W.; Gobel, U.; Goodell, M.A.; Brenner, M.K. A distinct “side population” of cells with high drug efflux capacity in human tumor cells. Proc. Natl. Acad. Sci. USA 2004, 101, 14228–14233.
- Janvilisri, T.; Venter, H.; Shahi, S.; Reuter, G.; Balakrishnan, L.; van Veen, H.W. Sterol transport by the human breast cancer resistance protein (ABCG2) expressed in Lactococcus lactis. J. Biol. Chem. 2003, 278, 20645–20651.
- Pal, A.; Mehn, D.; Molnar, E.; Gedey, S.; Meszaros, P.; Nagy, T.; Glavinas, H.; Janaky, T.; von Richter, O.; Bathori, G.; et al. Cholesterol potentiates ABCG2 activity in a heterologous expression system: Improved in vitro model to study function of human ABCG2. J. Pharmacol. Exp. Ther. 2007, 321, 1085–1094.
- Szilagyi, J.T.; Vetrano, A.M.; Laskin, J.D.; Aleksunes, L.M. Localization of the placental BCRP/ABCG2 transporter to lipid rafts: Role for cholesterol in mediating efflux activity. Placenta 2017, 55, 29–36.
- Storch, C.H.; Ehehalt, R.; Haefeli, W.E.; Weiss, J. Localization of the human breast cancer resistance protein (BCRP/ABCG2) in lipid rafts/caveolae and modulation of its activity by cholesterol in vitro. J. Pharmacol. Exp. Ther. 2007, 323, 257–264.
- Yu, L.; Hammer, R.E.; Li-Hawkins, J.; Von Bergmann, K.; Lutjohann, D.; Cohen, J.C.; Hobbs, H.H. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc. Natl. Acad. Sci. USA 2002, 99, 16237–16242.
- Williams, K.; Segard, A.; Graf, G.A. Sitosterolemia: Twenty Years of Discovery of the Function of ABCG5ABCG8. Int. J. Mol. Sci. 2021, 22, 2641.




