
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jordi Ribas-Maynou | + 2364 word(s) | 2364 | 2021-06-30 08:01:54 | | | |
| 2 | Vivi Li | Meta information modification | 2364 | 2021-07-09 04:59:58 | | |
Video Upload Options
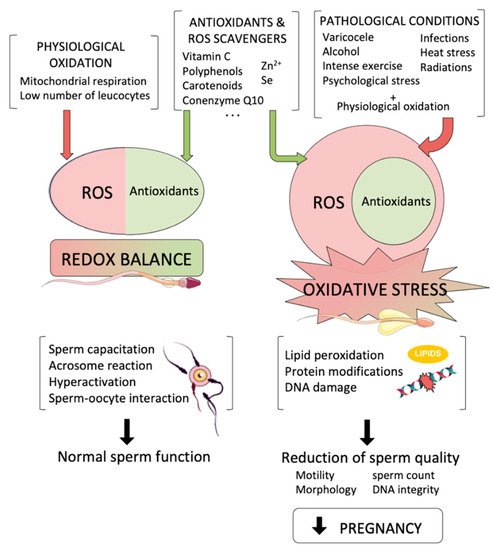
The spermatozoon is a highly specialized cell, whose main function is the transport of the intact male genetic material into the oocyte. During its formation and transit throughout male and female reproductive tracts, sperm cells are internally and externally surrounded by reactive oxygen species (ROS), which are produced from both endogenous and exogenous sources. While low amounts of ROS are known to be necessary for crucial physiological sperm processes, such as acrosome reaction and sperm–oocyte interaction, high levels of those species underlie misbalanced antioxidant-oxidant molecules, generating oxidative stress (OS), which is one of the most damaging factors that affect sperm function and lower male fertility potential.
1. Introduction
2. Sources of Oxidative Stress with Potential Effects to the Male Germline
2.1. Endogenous Sources of ROS
2.2. Exogenous Sources of ROS
2.2.1. Varicocele
2.2.2. Infections and Leucocytospermia
2.2.3. Alcohol and Tobacco
2.2.4. Physical Exercise and Heat Stress
2.2.5. Radiations and Pollution
References
- Louis, J.F.; Thoma, M.E.; Sørensen, D.N.; McLain, A.C.; King, R.B.; Sundaram, R.; Keiding, N.; Buck Louis, G.M. The prevalence of couple infertility in the United States from a male perspective: Evidence from a nationally representative sample. Andrology 2013, 1, 741–748.
- Datta, J.; Palmer, M.J.; Tanton, C.; Gibson, L.J.; Jones, K.G.; Macdowall, W.; Glasier, A.; Sonnenberg, P.; Field, N.; Mercer, C.H.; et al. Prevalence of infertility and help seeking among 15000 women and men. Hum. Reprod. 2016, 31, 2108–2118.
- Polis, C.B.; Cox, C.M.; Tunçalp, Ö.; McLain, A.C.; Thoma, M.E. Estimating infertility prevalence in low-to-middle-income countries: An application of a current duration approach to Demographic and Health Survey data. Hum. Reprod. 2017, 32, 1064–1074.
- Mascarenhas, M.N.; Flaxman, S.R.; Boerma, T.; Vanderpoel, S.; Stevens, G.A. National, Regional, and Global Trends in Infertility Prevalence Since 1990: A Systematic Analysis of 277 Health Surveys. PLoS Med. 2012, 9, e1001356.
- Bushnik, T.; Cook, J.L.; Yuzpe, A.A.; Tough, S.; Collins, J. Estimating the prevalence of infertility in Canada. Hum. Reprod. 2012, 27, 738–746.
- Agarwal, A.; Mulgund, A.; Hamada, A.; Chyatte, M.R. A unique view on male infertility around the globe. Reprod. Biol. Endocrinol. 2015, 13, 37.
- Mehra, B.L.; Skandhan, K.P.; Prasad, B.S.; Pawankumar, G.; Singh, G.; Jaya, V. Male infertility rate: A retrospective study. Urologia 2018, 85, 22–24.
- Vander Borght, M.; Wyns, C. Fertility and Infertility: Definition and Epidemiology. Clin. Biochem. 2018, 62, 2–10.
- Krausz, C.; Escamilla, A.R.; Chianese, C. Genetics of male infertility: From research to clinic. Reproduction 2015, 150, R159–R174.
- Sharma, P.; Ghanghas, P.; Kaushal, N.; Kaur, J.; Kaur, P. Epigenetics and oxidative stress: A twin-edged sword in spermatogenesis. Andrologia 2019, 51, e13432.
- Jodar, M.; Selvaraju, S.; Sendler, E.; Diamond, M.P.; Krawetz, S.A. Reproductive Medicine Network The presence, role and clinical use of spermatozoal RNAs. Hum. Reprod. Update 2013, 19, 604–624.
- Jensen, C.F.S.; Østergren, P.; Dupree, J.M.; Ohl, D.A.; Sønksen, J.; Fode, M. Varicocele and male infertility. Nat. Rev. Urol. 2017, 14, 523–533.
- Soraggi, S.; Riera, M.; Rajpert-De Meyts, E.; Schierup, M.H.; Almstrup, K. Evaluating genetic causes of azoospermia: What can we learn from a complex cellular structure and single-cell transcriptomics of the human testis? Hum. Genet. 2020, 1–9.
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012.
- Tosic, J.; Walton, A. Metabolism of spermatozoa. The formation and elimination of hydrogen peroxide by spermatozoa and effects on motility and survival. Biochem. J. 1950, 47, 199–212.
- Lamirande, E.; Harakat, A.; Gagnon, C. Human Sperm Capacitation Induced by Biological Fluids and Progesterone, but Not by NADH or NADPH, Is Associated with the Production of Superoxide Anion. J. Androl. 1998, 19, 215–225.
- Herrero, M.B.; de Lamirande, E.; Gagnon, C. Nitric Oxide Regulates Human Sperm Capacitation and Protein-Tyrosine Phosphorylation in Vitro. Biol. Reprod. 1999, 61, 575–581.
- Barati, E.; Nikzad, H.; Karimian, M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life Sci. 2020, 77, 93–113.
- Griveau, J.F.; Lannou, D.L. Reactive oxygen species and human spermatozoa: Physiology and pathology. Int. J. Androl. 1997, 20, 61–69.
- Camello-Almaraz, C.; Gomez-Pinilla, P.J.; Pozo, M.J.; Camello, P.J. Mitochondrial reactive oxygen species and Ca2+ signaling. Am. J. Physiol. Cell Physiol. 2006, 291, C1082–C10828.
- Aitken, R.J.; Buckingham, D.W.; Carreras, A.; Irvine, D.S. Superoxide dismutase in human sperm suspensions: Relationship with cellular composition, oxidative stress, and sperm function. Free Radic. Biol. Med. 1996, 21, 495–504.
- Said, T.M.; Agarwal, A.; Sharma, R.K.; Mascha, E.; Sikka, S.C.; Thomas, A.J. Human sperm superoxide anion generation and correlation with semen quality in patients with male infertility. Fertil. Steril. 2004, 82, 871–877.
- Aitken, R.J.; Gibb, Z.; Baker, M.A.; Drevet, J.; Gharagozloo, P. Causes and consequences of oxidative stress in spermatozoa. Reprod. Fertil. Dev. 2016, 28, 1–10.
- Guerriero, G.; Trocchia, S.; Abdel-Gawad, F.K.; Ciarcia, G. Roles of Reactive Oxygen Species in the Spermatogenesis Regulation. Front. Endocrinol. 2014, 5, 56.
- Krzyściak, W. Activity of selected aromatic amino acids in biological systems. Acta Biochim. Pol. 2011, 58, 461–466.
- Mehta, A.; Goldstein, M. Microsurgical varicocelectomy: A review. Asian J. Androl. 2013, 15, 56–60.
- Kroese, A.C.; de Lange, N.M.; Collins, J.; Evers, J.L. Surgery or embolization for varicoceles in subfertile men. Cochrane Database Syst. Rev. 2012.
- Plante, M.; de Lamirande, E.; Gagnon, C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil. Steril. 1994, 62, 387–393.
- WHO. WHO Laboratory Manual for the Examination and Precesssing of Human Semen, 5th ed.; World Heal Organ Press: Geneva, Switzerland, 2010.
- Henkel, R. The impact of oxidants on sperm function. Andrologia 2005, 37, 205–206.
- Lobascio, A.M.; De Felici, M.; Anibaldi, M.; Greco, P.; Minasi, M.G.; Greco, E. Involvement of seminal leukocytes, reactive oxygen species, and sperm mitochondrial membrane potential in the DNA damage of the human spermatozoa. Andrology 2015, 3, 265–270.
- Seshadri, S.; Flanagan, B.; Vince, G.; Lewis Jones, D.I. Leucocyte subpopulations in the seminal plasma and their effects on fertilisation rates in an IVF cycle. Andrologia 2012, 44, 396–400.
- Saleh, R.A.; Agarwal, A.; Kandirali, E.; Sharma, R.K.; Thomas, A.J.; Nada, E.A.; Evenson, D.P.; Alvarez, J.G. Leukocytospermia is associated with increased reactive oxygen species production by human spermatozoa. Fertil. Steril. 2002, 78, 1215–1224.
- Akang, E.N.; Oremosu, A.A.; Osinubi, A.A.; James, A.B.; Biose, I.J.; Dike, S.I.; Idoko, K.M. Alcohol-induced male infertility: Is sperm DNA fragmentation a causative? J. Exp. Clin. Anat. 2017, 16, 53.
- Manzo-Avalos, S.; Saavedra-Molina, A. Cellular and Mitochondrial Effects of Alcohol Consumption. Int. J. Environ. Res. Public Health 2010, 7, 4281–4304.
- Rompala, G.R.; Homanics, G.E. Intergenerational Effects of Alcohol: A Review of Paternal Preconception Ethanol Exposure Studies and Epigenetic Mechanisms in the Male Germline. Alcohol. Clin. Exp. Res. 2019, 43, 1032–1045.
- Zhu, Q.; Meisinger, J.; Emanuele, N.V.; Emanuele, M.A.; LaPaglia, N.; Van Thiel, D.H. Ethanol exposure enhances apoptosis within the testes. Alcohol. Clin. Exp. Res. 2000, 24, 1550–1556.
- Koh, P.O.; Kim, M.O. Ethanol Exposure Decreases Cell Proliferation and Increases Apoptosis in Rat Testes. J. Vet. Med. Sci. 2006, 68, 1013–1017.
- Sadeghzadeh, M.; Shirpoor, A.; Naderi, R.; Kheradmand, F.; Gharalari, F.H.; Samadi, M.; Khalaji, N.; Gharaaghaji, R. Long-term ethanol consumption promotes changes in β-defensin isoform gene expression and induces structural changes and oxidative DNA damage to the epididymis of rats. Mol. Reprod. Dev. 2019, 86, 624–631.
- Aitken, R.J.; Baker, M.A. Causes and consequences of apoptosis in spermatozoa; contributions to infertility and impacts on development. Int. J. Dev. Biol. 2013, 57, 265–272.
- Sakkas, D.; Alvarez, J.G. Sperm DNA fragmentation: Mechanisms of origin, impact on reproductive outcome, and analysis. Fertil. Steril. 2010, 93, 1027–1036.
- Aparicio, I.M.; Espino, J.; Bejarano, I.; Gallardo-Soler, A.; Campo, M.L.; Salido, G.M.; Pariente, J.A.; Peña, F.J.; Tapia, J.A. Autophagy-related proteins are functionally active in human spermatozoa and may be involved in the regulation of cell survival and motility. Sci. Rep. 2016, 6, 33647.
- Zhang, M.; Jiang, M.; Bi, Y.; Zhu, H.; Zhou, Z.; Sha, J. Autophagy and Apoptosis Act as Partners to Induce Germ Cell Death After Heat Stress in Mice. PLoS ONE 2012, 7, e41412.
- Rehman, R.; Zahid, N.; Amjad, S.; Baig, M.; Gazzaz, Z.J. Relationship Between Smoking Habit and Sperm Parameters Among Patients Attending an Infertility Clinic. Front. Physiol. 2019, 10, 1356.
- Dai, J.-B.; Wang, Z.-X.; Qiao, Z.-D. The hazardous effects of tobacco smoking on male fertility. Asian J. Androl. 2015, 17, 954–960.
- Santos, M.; Rodríguez-González, G.L.; Ibáñez, C.; Vega, C.C.; Nathanielsz, P.W.; Zambrano, E. Adult exercise effects on oxidative stress and reproductive programming in male offspring of obese rats. Am. J. Physiol. Integr. Comp. Physiol. 2015, 308, R219–R225.
- Vaamonde, D.; Da Silva-Grigoletto, M.E.; García-Manso, J.M.; Barrera, N.; Vaamonde-Lemos, R. Physically active men show better semen parameters and hormone values than sedentary men. Eur. J. Appl. Physiol. 2012, 112, 3267–3273.
- Gaskins, A.J.; Mendiola, J.; Afeiche, M.; Jørgensen, N.; Swan, S.H.; Chavarro, J.E. Physical activity and television watching in relation to semen quality in young men. Br. J. Sports Med. 2015, 49, 265–270.
- Eisenberg, M.L.; Kim, S.; Chen, Z.; Sundaram, R.; Schisterman, E.F.; Louis, G.M.B. The relationship between male BMI and waist circumference on semen quality: Data from the LIFE study. Hum. Reprod. 2015, 30, 493–494.
- Hajizadeh Maleki, B.; Tartibian, B.; Chehrazi, M. The effects of three different exercise modalities on markers of male reproduction in healthy subjects: A randomized controlled trial. Reproduction 2017, 153, 157–174.
- Mastaloudis, A.; Leonard, S.W.; Traber, M.G. Oxidative stress in athletes during extreme endurance exercise. Free Radic. Biol. Med. 2001, 31, 911–922.
- Durairajanayagam, D.; Agarwal, A.; Ong, C. Causes, effects and molecular mechanisms of testicular heat stress. Reprod. Biomed. Online 2015, 30, 14–27.
- Parazzini, F.; Marchini, M.; Luchini, L.; Tozzi, L.; Mezzopane, R.; Fedele, L. Tight underpants and trousers and risk of dyspermia. Int. J. Androl. 1995, 18, 137–140.
- Kesari, K.K.; Agarwal, A.; Henkel, R. Radiations and male fertility. Reprod. Biol. Endocrinol. 2018, 16, 118.
- Kumar, S.; Nirala, J.P.; Behari, J.; Paulraj, R. Effect of electromagnetic irradiation produced by 3G mobile phone on male rat reproductive system in a simulated scenario. Indian J. Exp. Biol. 2014, 52, 890–897.
- Agarwal, A.; Desai, N.R.; Makker, K.; Varghese, A.; Mouradi, R.; Sabanegh, E.; Sharma, R. Effects of radiofrequency electromagnetic waves (RF-EMW) from cellular phones on human ejaculated semen: An in vitro pilot study. Fertil. Steril. 2009, 92, 1318–1325.
- Mailankot, M.; Kunnath, A.P.; Jayalekshmi, H.; Koduru, B.; Valsalan, R. Radio frequency electromagnetic radiation (RF-EMR) from GSM (0.9/1.8GHz) mobile phones induces oxidative stress and reduces sperm motility in rats. Clinics 2009, 64, 561–565.
- Meena, R.; Kumari, K.; Kumar, J.; Rajamani, P.; Verma, H.N.; Kesari, K.K. Therapeutic approaches of melatonin in microwave radiations-induced oxidative stress-mediated toxicity on male fertility pattern of Wistar rats. Electromagn. Biol. Med. 2014, 33, 81–91.
- Avendano, C.; Mata, A.; Sarmiento, C.A.S.; Doncel, G.F. Use of Laptop Computers Connected to Internet Through Wi-Fi Decreases Human Sperm Motility and Increases Sperm DNA Fragmentation. Fertil. Steril. 2012, 97, 39–44.
- Kumar, S.; Behari, J.; Sisodia, R. Influence of electromagnetic fields on reproductive system of male rats. Int. J. Radiat. Biol. 2013, 89, 147–154.
- Loseva, O.; Shubbar, E.; Haghdoost, S.; Evers, B.; Helleday, T.; Harms-Ringdahl, M. Chronic Low Dose Rate Ionizing Radiation Exposure Induces Premature Senescence in Human Fibroblasts that Correlates with up Regulation of Proteins Involved in Protection against Oxidative Stress. Proteomes 2014, 2, 341–362.
- Einor, D.; Bonisoli-Alquati, A.; Costantini, D.; Mousseau, T.A.; Møller, A.P. Ionizing radiation, antioxidant response and oxidative damage: A meta-analysis. Sci. Total Environ. 2016, 548–549, 463–471.
- De Rosa, M.; Zarrilli, S.; Paesano, L.; Carbone, U.; Boggia, B.; Petretta, M.; Maisto, A.; Cimmino, F.; Puca, G.; Colao, A.; et al. Traffic pollutants affect fertility in men. Hum. Reprod. 2003, 18, 1055–1061.
- Carré, J.; Gatimel, N.; Moreau, J.; Parinaud, J.; Léandri, R. Does air pollution play a role in infertility? A systematic review. Environ. Health 2017, 16, 82.
- Rubes, J.; Selevan, S.G.; Evenson, D.P.; Zudova, D.; Vozdova, M.; Zudova, Z.; Robbins, W.A.; Perreault, S.D. Episodic air pollution is associated with increased DNA fragmentation in human sperm without other changes in semen quality. Hum. Reprod. 2005, 20, 2776–2783.
- Rengaraj, D.; Kwon, W.-S.; Pang, M.-G. Effects of Motor Vehicle Exhaust on Male Reproductive Function and Associated Proteins. J. Proteome Res. 2015, 14, 22–37.
- Bahrami, N.; Goudarzi, M.; Hosseinzadeh, A.; Sabbagh, S.; Reiter, R.J.; Mehrzadi, S. Evaluating the protective effects of melatonin on di(2-ethylhexyl) phthalate-induced testicular injury in adult mice. Biomed. Pharmacother. 2018, 108, 515–523.
- Al-Saleh, I.; Coskun, S.; Al-Doush, I.; Al-Rajudi, T.; Al-Rouqi, R.; Abduljabbar, M.; Al-Hassan, S. Exposure to phthalates in couples undergoing in vitro fertilization treatment and its association with oxidative stress and DNA damage. Environ. Res. 2019, 169, 396–408.
- Lafuente, R.; García-Blàquez, N.; Jacquemin, B.; Checa, M.A. Outdoor Air Pollution and Sperm Quality. Fertil. Steril. 2016, 106, 880–896.




