
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Robert McCulloch | + 1097 word(s) | 1097 | 2021-04-01 15:00:18 | | | |
| 2 | Conner Chen | Meta information modification | 1097 | 2021-07-09 05:03:08 | | |
Video Upload Options
1. Patient-Specific Instrumentation and Reconstruction
In recent years, CATS has been developed along with 3D printing technology, which represents the possibility to personalize reconstruction with custom-made prostheses [1][2][3] and improve accuracy in bone cutting using patient-specific instruments (PSI) [4][5].
Manufacturing of 3D-printed PSI jigs and custom-made prostheses is based upon the principle of rapid prototyping (RP). RP is a process that directly produces a physical object with a defined structure and shape on the basis of virtual/mathematical model data. Rapid prototyping was first used in the late 1980s and was developed to apply the precision and functionality of computer-assisted design (CAD) to manufacturing. With this technology, a prototype could be quickly produced and accurately represents the engineers draft [6][7]. Electron beam melting (EMB) is the optimal technology to fabricate metallic components with complex shapes and porous structures and has a pivotal role either in the PSI technology or prosthesis manufacturing. EBM involves the generation of an electron beam focused on a powder layer that is added to a previous one. Each layer (50–100 µm thickness) is pre-heated, and using an electron beam a high temperature is generated (up to 600–800 °C), allowing the fusion of the powder according to the 3D CAD project. This process is repeated layer by layer, building the model in a vacuum chamber [8].
Traditional methods of manufacturing orthopaedic implants used subtractive machining, where the material is removed from a metal block until the planned shape has been achieved. The process of RP is different to this and uses the process of additive manufacturing. This is when a construct is made based upon a digital model by plastic or metal being deposited in layers. This provides versatility when constructing complex geometric shapes. Changes in design require no new equipment; therefore, it is ideally suited to low volume patient-specific instrumentation or implants [8][9][10].
In surgery, 3D printing is utilised in the production of the following [9][10]:
-
Anatomical models.
-
Patient-specific instrumentation (PSI).
-
Custom-made implants and devices.
2. Anatomical Models
Anatomical model fabrication is the most common use of 3D printing in surgery.
In the planning stage, 3D models are used to represent the anatomy, simulate surgical procedure and test surgical tools (Figure 1 and Figure 2). Studying a patient’s anatomy with a replication of the structures gives a better comprehension compared to 2D images on a computer screen [11][12]. The 3D-printed model can be shown to patients in order to explain the pathology, surgical planning and assist the consenting process [13][14].
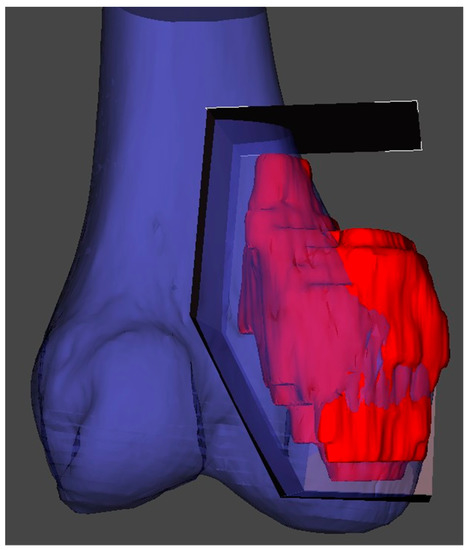
Figure 1. 3D reconstruction and planning of a resection for a distal femoral sarcoma.
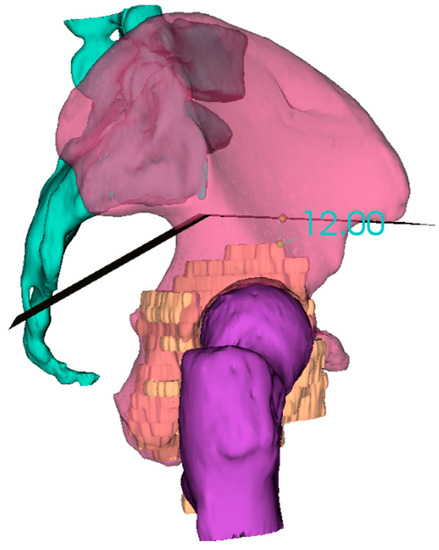
Figure 2. 3D reconstruction and planning of a resection for a pelvic sarcoma.
3. PSI (Patient-Specific Instrumentation)
PSI technology is the most popular medical application of 3D printing and has been developed as an alternative to intraoperative navigation. PSI is designed on a 3D model based on CT and MRI fusion according to the surgical approach, desired resection margins and reconstruction method. The unique anatomy of the patient and the PSI shape allows to place the guide only in a pre-defined position. These contact surfaces must be defined by both surgeon and engineer, considering the surgical approach, the bone exposure and tumour extension. These features make the application of PSI more useful for bone deformities and tumours where the anatomy is often abnormal. The final PSI is commonly printed in nylon or polyamide and provided to the surgeon sterile or to be sterilized (Figure 3). Thus, this method reproduces surgical planning with more accuracy than a free-hand approach. Furthermore, it encourages surgeons to perform a “virtual” surgery in advance, leading to a better comprehension of the possible pitfalls and to a tailored treatment for each patient. This complex procedure in some way has changed surgeons’ mindset due to the network that must be established with a team of engineers.
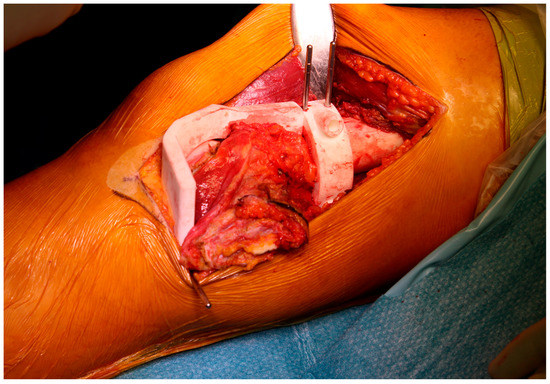
Figure 3. Final patient-specific instrumentation (PSI) printed in nylon and secured intraoperatively.
Limb salvage surgery has become the rule for most patients with bone tumours. However, resection of these tumours needs to achieve wide margins to limit local recurrence and at the same time preserve function [15][16]. In pelvic tumours, surgery is more difficult due to the complex three-dimensional (3D) anatomy of the pelvic bone and the presence of neurovascular structures as well as viscera. This situation leads to a higher local recurrence and complication rate in the pelvis compared with long bones [4][17][18]. In long bones, the use of PSI is most useful in joint preserving resection, in particular in children or young patients in which a reconstruction with a bone allograft can be carried out [15][16].
The performance and accuracy of PSI has been demonstrated; it improves alignment and surgical and operating theatre times [19][20][21][22][23][24][25][26], as well as reducing the risk of contamination and wasting of instrument trays, even if some trials have not displayed significant advantages [27]. Nonetheless, in tumour surgery, the possibility to reduce surgical time whilst performing a more precise cut on the bone and reproducing it on an allograft (or a prosthesis) may improve the outcome of the oncological patient [28].
4. Custom-Made Implants and Devices
Custom-made implants have been used since the 1950s, when a complex reconstruction was performed in a limb salvage procedure [29]. The physical properties of titanium alloy are the most adaptable to bone defect reconstruction. Titanium has high mechanical resistance similar to stainless steel, a low density (50% less than stainless steel) and a low elasticity Young’s modulus, giving the necessary flexibility associated with a high mechanical capacity [30][31][32]. Furthermore, the superficial part of titanium is covered by a free oxygen layer that increase the biocompatibility and biological integration with the surrounding tissues [33][34][35][36].
Many studies have shown that the porosity of titanium is one of the most relevant features influencing bone ingrowth; Frosch et al. has demonstrated that 300–600 µm is the better porosity for osteoconduction [37].
A more recent development is a particular configuration of titanium called trabecular titanium; the main advantage of the trabecular structure is the rough surface that amplifies the contact with host bone, increasing the integration.
References
- Sun, W.; Li, J.; Li, Q.; Li, G.; Cai, Z. Clinical effectiveness of hemipelvic reconstruction using computer-aided custom-made prostheses after resection of malignant pelvic tumors. J. Arthroplast. 2011, 26, 1508–1513.
- Shah, F.A.; Snis, A.; Matic, A.; Thomsen, P.; Palmquist, A. 3D printed Ti6Al4V implant surface promotes bone maturation and retains a higher density of less aged osteocytes at the bone-implant interface. Acta Biomater. 2016, 30, 357–367.
- Merema, B.J.; Kraeima, J.; Ten Duis, K.; Wendt, K.W.; Warta, R.; Vos, E.; Schepers, R.H.; Witjes, M.J.H.; IJpma, F.F.A. The design, production and clinical application of 3D patient-specific implants with drilling guides for acetabular surgery. Injury 2017, 48, 2540–2547.
- Cartiaux, O.; Paul, L.; Francq, B.G.; Banse, X.; Docquier, P.L. Improved accuracy with 3D planning and patient-specific instruments during simulated pelvic bone tumor surgery. Ann. Biomed. Eng. 2014, 42, 205–213.
- Wong, K.C.; Kumta, S.M.; Sze, K.Y.; Wong, C.M. Use of a patient-specific CAD/CAM surgical jig in extremity bone tumor resection and custom prosthetic reconstruction. Comput. Aided Surg. 2012, 17, 284–293.
- Malik, H.H.; Darwood, A.R.; Shaunak, S.; Kulatilake, P.; El-Hilly, A.A.; Mulki, O.; Baskaradas, A. Three dimensional printing in surgery: A review of current surgical applications. J. Surg. Res. 2015, 199, 512–522.
- McGurk, M.; Amis, A.A.; Potamianos, P.; Goodger, N.M. Rapid prototyping techniques for anatomical modelling in medicine. Ann. R. Coll. Surg. Engl. 1997, 79, 169–174.
- Murr, L.E.; Gaytan, S.M.; Martinez, E.; Medina, F.; Wicker, R.B. Next generation orthopaedic implants by additive manufacturing using electron beam melting. Int. J. Biomater. Biomater. 2012, 24572.
- Tack, P.; Victor, J.; Gemmel, P.; Annemans, L. 3D-printing techniques in a medical setting: A systematic literature review. Biomed. Eng. Online 2016, 21, 115.
- Hoang, D.; Perrault, D.; Stevanovic, M.; Ghiassi, A. Surgical applications of three-dimensional printing: A review of the current literature & how to get started. Ann. Transl. Med. 2016, 4, 456.
- Park, J.H.; Lee, Y.; Shon, O.J.; Shon, H.C.; Kim, J.W. Surgical tips of intramedullary nailing in severely bowed femurs in atypical femur fractures: Simulation with 3D printed model. Injury 2016, 47, 1318–1324.
- Zheng, Y.X.; Yu, D.F.; Zhao, J.G.; Wu, Y.L.; Zheng, B. 3D Printout Models vs. 3D-Rendered Images: Which Is Better for Preoperative Planning? J. Surg. Educ. 2016, 73, 518–523.
- Giannetti, S.; Bizzotto, N.; Stancati, A.; Santucci, A. Minimally invasive fixation in tibial plateau fractures using an pre-operative and intra-operative real size 3D printing. Injury 2017, 48, 784–788.
- Bizzotto, N.; Tami, I.; Tami, A.; Spiegel, A.; Romani, D.; Corain, M.; Adani, R.; Magnan, B. 3D Printed models of distal radius fractures. Injury 2016, 47, 976–978.
- Bellanova, L.; Paul, L.; Docquier, P.L. Surgical guides (patient-specific instruments) for pediatric tibial bone sarcoma resection and allograft reconstruction. Sarcoma 2013, 2013.
- Ma, L.; Zhou, Y.; Zhu, Y.; Lin, Z.; Wang, Y.; Zhang, Y.; Xia, H.; Mao, C. 3D-printed guiding templates for improved osteosarcoma resection. Sci. Rep. 2016, 6, 23335.
- Farfalli, G.L.; Albergo, J.I.; Ritacco, L.E.; Ayerza, M.A.; Muscolo, D.L.; Aponte-Tinao, L.A. Oncologic and clinical outcomes in pelvic primary bone sarcomas treated with limb salvage surgery. Musculoskelet. Surg. 2015, 99, 237–242.
- Cartiaux, O.; Banse, X.; Paul, L.; Francq, B.G.; Aubin, C.; Docquier, P. Computer-assisted planning and navigation improves cutting accuracy during simulated bone tumor surgery of the pelvis. Comput. Aided Surg. 2013, 18, 19–26.
- White, D.; Chelule, K.L.; Seedhom, B.B. Accuracy of MRI vs CT imaging with particular reference to patient specific templates for total knee replacement surgery. Int. J. Med. Robot. 2008, 4, 224–231.
- Kunz, M.; Rudan, J.F.; Wood, G.C.; Ellis, R.E. Registration stability of physical templates in hip surgery. Stud. Health Technol. Inform. 2011, 163, 283–289.
- Schkommodau, E.; Decker, N.; Klapper, U.; Birnbaum, K.; Staudte, H.W.; Radermacher, K. Pedicle Screw Implantation Using the DISOS Template System. In Navigation and Robotics in Total Joint and Spine Surgery; Stiehl, J.B., Konermann, W.H., Haaker, R.C., Eds.; Springer: Berlin, Germany, 2003; pp. 501–505.
- Dobbe, J.G.; Pre’, K.J.; Kloen, P.; Blankevoort, L.; Streekstra, G.J. Computer-assisted and patient-specific 3-D planning and evaluation of a single-cut rotational osteotomy for complex long-bone deformities. Med. Biol. Eng. Comput. 2011, 49, 1363–1370.
- Birnbaum, K.; Schkommodau, E.; Decker, N.; Prescher, A.; Klapper, U.; Radermacher, K. Computer assisted orthopedic surgery with individual templates and comparison to conventional operation method. Spine 2001, 26, 365–370.
- Donati, D.; Di Bella, C.; Frisoni, T.; Cevolani, L.; DeGroot, H. Alloprosthetic composite is a suitable reconstruction after periacetabular tumor resection. Clin. Orthop. Relat. Res. 2011, 469, 1450–1458.
- Paolis, D.M.; Biazzo, A.; Romagnoli, C.; Alì, N.; Giannini, S.; Donati, D.M. The use of iliac stem prosthesis for acetabular defects following resections for periacetabular tumors. Sci. World J. 2013, 717031.
- Enneking, W.K.F.; Dunham, W.K. Resection and reconstruction for primary neoplasms involving the innominate bone. J. Bone. Joint. Surg. 1978, 60, 731–746.
- Renson, L.; Poilvache, P.; Van den Wyngaert, H. Improved alignment and operating room efficiency with patient-specific instrumentation for TKA. Knee 2014, 21, 1216–1220.
- Wong, K.C.; Sze, K.Y.; Wong, I.O.; Wong, C.M.; Kumta, S.M. Patient-specific instrument can achieve same accuracy with less resection time than navigation assistance in periacetabular pelvic tumor surgery: A cadaveric study. Int. J. Comput. Assist. Radiol. Surg. 2015, 11, 307–316.
- Scales, J.T. Prostheses in the management of bone cancer. Br. Med. J. (Clin. Res. Ed.) 1983, 287, 761–762.
- Lewandowski, J.J.; Seifi, M. Metal additive manufacturing: A review of mechanical properties. Annu. Rev. Mater. Res. 2016, 46, 151–186.
- Facchini, L.; Magalini, E.; Robotti, P.; Molinari, A. Microstructure and mechanical properties of Ti-6Al-4V produced by electron beam melting of pre-alloyed powders. Rapid Prototyp. J. 2009, 15, 171–178.
- Liu, P.; Yang, Y.; Liu, R.; Shu, H.; Gong, J.; Yang, Y.; Sun, Q.; Wu, X.; Cai, M. A study on the mechanical characteristics of the EBM-printed Ti-6Al-4V LCP plates in vitro. J. Orthop. Surg. Res. 2014, 9, 106.
- Parthasarathy, J.; Starly, B.; Raman, S.; Christensen, C. Mechanical evaluation of porous titanium (Ti6Al4V) structures with electron beam melting (EBM). J. Mech. Behav. Biomed. Mater. 2010, 3, 249–259.
- Niinomi, M. Mechanical biocompatibilities of titanium alloys for biomedical applications. J. Mech. Behav. Biomed. Mater. 2008, 1, 30–42.
- Bandyopadhyay, A.; Espana, F.; Balla, V.K.; Bose, S.; Ohgami, Y.; Davies, N.M. Influence of porosity on mechanical properties and in vivo response of Ti6Al4V implants. Acta Biomater. 2010, 6, 1640–1648.
- Marin, E.; Pressacco, M.; Fusi, S.; Lanzutti, A.; Turchet, S.; Fedrizzi, L. Characterization of grade 2 commercially pure Trabecular Titanium structures. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 2648–2656.
- Frosch, K.H.; Barvencik, F.; Lohmann, C.H.; Viereck, V.; Siggelkow, H.; Breme, J.; Dresing, K.; Stürmer, K.M. Migration, matrix production and lamellar bone formation of human osteoblast-like cells in porous titanium implants. Cells Tissues Organs. 2002, 170, 214–227.




