
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anna Gliszczyńska | + 2621 word(s) | 2621 | 2021-06-21 06:16:17 | | | |
| 2 | Peter Tang | Meta information modification | 2621 | 2021-06-28 04:26:21 | | |
Video Upload Options
Methoxylated derivatives of cinnamic acid play an important role in the formation of the pro-health potential of food products. Numerous reports present them as molecules with strong antimicrobial, antidiabetic, anticancer as well as hepato-, cardio-, and neuroprotective activities.
1. Introduction
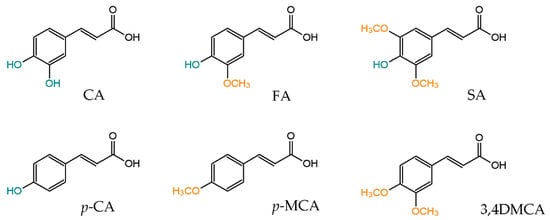
2. Biological Activity of Methoxylated Derivatives of Cinnamic Acid
|
Activity |
Molecule |
Research Model |
Active Dose |
Mechanism of Action |
Refs. |
|---|---|---|---|---|---|
|
Hepatoprotective |
FA |
rats/EtOH |
20 mg/kg b.w. |
ALP↓, GGT↓, ALT↓, AST↓ |
[28] |
|
MFA |
mice/EtOH |
5–20 mg/kg b.w. |
ALT↓, AST↓, SOD↑, CAT↑, GSH↑ GSH-Px↑, T-AOC↑ |
[40] |
|
|
p-MCA |
rats/CCl4 |
1–5 μM |
GSH↑, GSH-Px↑, GST↑, GR↑ |
[16] |
|
|
p-MCA |
rats/CCl4 |
50 mg/kg |
ALP↓, ALT↓, GGT↓ |
[39] |
|
|
Antydiabectic |
p-MCA |
INS-1 cell line |
100 μM |
insulin↑ |
[34] |
|
p-MCA |
INS-1 cell line |
100 μM |
ions Ca2+↑, insulin↑ |
[35] |
|
|
p-MCA |
Wistar rats |
5 mg/kg |
insulin↑ |
[34] |
|
|
p-MCA |
rats/STPZ |
40 mg/kg |
insulin↑, glukogenesis↓ |
[36] |
|
|
p-MCA |
rats/STPZ |
40–100 mg/kg |
insulin↑ |
[37] |
|
|
p-MCA |
rats/STPZ |
10–40 mg/kg |
Insulin↑ |
[38] |
|
|
Neuroprotective |
FA |
rats-ROT |
50 mg/kg m.c. |
neuron necrosis↓, IL-1β↓, IL-6↓, TNF-α↓, CAT↑, SOD↑ |
[30] |
|
p-MCA |
rat cortical cells/glutamate |
1 μM |
- |
[41] |
|
|
Ep-MCA |
rat cortical cells/glutamate |
0.01–1 μM |
ions Ca2+↑, glutamatergic antagonism |
[42] |
|
|
Ep-MCA |
mice-ICR |
0.01–2 mg/kg |
- |
[43] |
|
|
3,4-DMCA |
SH-SY5Y cell line |
400 nM (Kd) |
cell viability↑ |
[44] |
|
|
Chemopreventive and anticancer |
FA |
MDA-MB-231 mices |
100 mg/kg |
apoptosis↑ metastatic potential↓ |
[23] |
|
FA |
mice/UV radiation |
50 mg/kg |
DNA protection |
[45] |
|
|
FA |
PC-3, LNCaP cell line |
300 μM 500 μM |
cell proliferation↓ |
[25] |
|
|
FA |
rats/DMAB |
40 mg/kg |
- |
[46] |
|
|
FA |
rats/4NQO |
500 ppm |
- |
[47] |
|
|
p-MCA |
HepG2 cell line |
27.1 g/mL (IC50) |
apoptosis↑ |
[14] |
|
|
p-MCA |
rats |
40 mg/kg |
- |
[48] |
|
|
p-MCA |
HCT-116 cell line |
10 μM (IC50) |
apoptosis↓ |
[49] |
|
|
Ep-MCA |
mice/DMAB |
23.4 mg/kg |
apoptosis↓ |
[50] |
|
|
Ep-MCA |
MCF-7 cell line |
360 g/mL (IC50) |
- |
[51] |
|
|
Ep-MCA |
rats/DMH |
40 mg/kg |
- |
[52] |
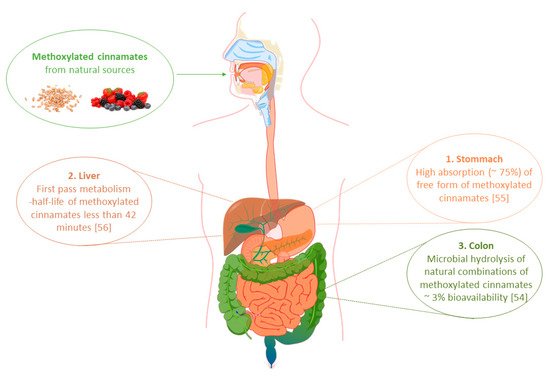
3. Bioavailability of Phenylpropenoic Acids
References
- Kulbat, K. The role of phenolic compounds in plant resistance. Biotechnol. Food Sci. 2016, 80, 97–108.
- Gharras, H. Polyphenols: Food sources, properties and applications—A review. Int. J. Food Sci. Technol. 2009, 44, 2512–2518.
- Ou, S.; Kwok, K. Ferulic acid: Pharmaceutical functions, preparation and applications in foods. J. Sci. Food Agric. 2004, 84, 1261–1269.
- Grosso, G.; Stepaniak, U.; Micek, A.; Kozela, M.; Stefler, D.; Bobak, M.; Pajak, A. Dietary polyphenol intake and risk of hypertension in the Polish arm of the HAPIEE study. Eur. J. Nutr. 2018, 57, 1535–1544.
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278.
- Russell, W.R.; Labat, A.; Scobbie, L.; Duncan, G.J.; Duthie, G.G. Phenolic acid content of fruits commonly consumed and locally produced in Scotland. Food Chem. 2009, 115, 100–104.
- Yadav, R.P.; Tarun, G. Versatility of turmeric: A review the golden spice of life. J. Pharmacogn. Phytochem. 2017, 6, 41–46.
- Dykens, L.; Rooney, L.W. Phenolic Compounds in Cereal Grains and Their Health Benefits. Cereal Food World 2007, 52, 105–111.
- Andrade, P.B.; Leitão, R.; Seabra, R.M.; Oliveira, M.B.; Ferreira, M.A. 3,4-Dimethoxycinnamic acid levels as a tool for differentiation of Coffea canephora var robusta and Coffea arabica. Food Chem. 1998, 61, 511–514.
- Clifford, M.N. Review Chlorogenic acids and other cinnamates—Nature, occurrence and dietary burden. J. Sci. Food Agric. 1999, 372, 362–372.
- Pei, K.; Ou, J.; Huang, J.; Ou, S. p-Coumaric acid and its conjugates: Dietary sources, pharmacokinetic properties. J. Sci. Food Agric. 2016, 96, 2952–2962.
- Zhao, Z.; Moghadasian, E.M.H. Bioavailability of hydroxycinnamates: A brief review of in vivo and in vitro studies. Phytochem. Rev. 2010, 9, 133–145.
- Helena, J.; Ferreira, R.S.; Pereira, E.P.; Braga de Souza, S.; Marylin, M.; Almeida, M.M.A.D.; Creusa, C.; Butt, A.M.; Caiazzo, E.; Diogenes, V.; et al. Amburana cearensis: Pharmacological and Neuroprotective Effects of Its Compounds. Molecules 2020, 25, 3394.
- Liu, B.; Liu, F.; Chen, C.; Gao, H. Supercritical carbon dioxide extraction of ethyl p-methoxycinnamate from Kaempferia galanga L. rhizome and its apoptotic induction in human HepG2 cells. Nat. Prod. Res. 2010, 24, 1927–1932.
- He, Z.H.; Yue, G.G.L.; Lau, C.B.S.; Ge, W.; But, P.P.H. Antiangiogenic effects and mechanisms of trans-ethyl p-methoxycinnamate from Kaempferia galanga L. J. Agric. Food Chem. 2012, 60, 11309–11317.
- Eun, J.L.; So, R.K.; Kim, J.; Young, C.K. Hepatoprotective phenylpropanoids from Scrophularia buergeriana roots against CCl4-induced toxicity: Action mechanism and structure-activity relationship. Planta Med. 2002, 68, 407–411.
- Ohishi, N.; Yagi, K. Action of Ferulic Acid and Its Derivatives as Antioxidants. J. Nutr. Sci. Vitaminol. 1979, 25, 127–130.
- Graf, E. Antioxidant Potential of Ferulic Acid. Free Radic. Biol. Med. 1992, 13, 435–448.
- Itagaki, S.; Kurokawa, T.; Nakata, C.; Saito, Y.; Oikawa, S.; Kobayashi, M.; Hirano, T.; Iseki, K. In vitro and in vivo antioxidant properties of ferulic acid: A comparative study with other natural oxidation inhibitors. Food Chem. 2009, 114, 466–471.
- Shi, C.; Zhang, X.; Sun, Y.; Yang, M.; Song, K.; Zheng, Z. Antimicrobial Activity of Ferulic Acid Against Cronobacter sakazakii and Possible Mechanism of Action. Foodborne Pathog. Dis. 2016, 13, 196–204.
- Fong, Y.; Tang, C.C.; Hu, H.T.; Fang, H.Y.; Chen, B.H.; Wu, C.Y. Inhibitory effect of trans-ferulic acid on proliferation and migration of human lung cancer cells accompanied with increased endogenous reactive oxygen species and β-catenin instability. Chin. Med. 2016, 11, 1–13.
- Zhang, X.; Jiang, R.; Li, H. Ferulic acid exerts antitumor activity and inhibits metastasis in breast cancer cells by regulating epithelial to mesenchymal transition. Oncol. Rep. 2016, 36, 271–278.
- Hemaiswarya, S.; Doble, M. Phytomedicine Combination of phenylpropanoids with 5-fluorouracil as anti-cancer agents against human cervical cancer (HeLa) cell line. Eur. J. Integr. Med. 2013, 20, 151–158.
- Eroglu, C.; Seçme, M.; Barger, G.; Dodurga, Y. Assessment of the anticancer mechanism of ferulic acid via cell cycle and apoptotic pathways in human prostate cancer cell lines. Tumor Biol. 2015, 36, 9437–9446.
- Dodurga, Y.; Eroğlu, C.; Seçme, M.; Elmas, L.; Avci, Ç.B.; Şatıroğlu-Tufan4, N.L. Anti-proliferative and anti-invasive effects of ferulic acid in TT medullary thyroid cancer cells interacting with URG4/URGCP. Tumor Biol. 2016, 37, 1933–1940.
- Kawabata, K.; Yamamoto, T.; Hara, A.; Shimizu, M. Modifying effects of ferulic acid on azoxymethane-induced colon carcinogenesis in F344 rats. Cancer Lett. 2000, 157, 15–21.
- Vinayagam, R.; Jayachandran, M.; Xu, B. Antidiabetic Effects of Simple Phenolic Acids: A Comprehensive Review. Phyther. Res. 2016, 30, 184–199.
- Rukkumani, R.; Aruna, K.; Varma, P.S. Hepatoprotective Role of Ferulic Acid: A Dose-Dependent Study. J. Med. Food 2004, 7, 456–461.
- Kwon, E.Y.; Do, G.M.; Cho, Y.Y.; Park, Y.B.; Jeon, S.M.; Choi, M.S. Anti-atherogenic property of ferulic acid in apolipoprotein E-deficient mice fed Western diet: Comparison with clofibrate. Food Chem. Toxicol. 2010, 48, 2298–2303.
- Ojha, S.; Hayate, J.; Azimullah, S.; Khair, S.B.; Haque, M. Neuroprotective potential of ferulic acid in the rotenone model of Parkinson’s disease. Drug Des. Devel. Ther. 2015, 9, 5499–5510.
- Feng, Q.; Xi, H. Guanxin Ⅱ (冠心Ⅱ号) for the Management of Coronary Heart Disease. Chin. J. Integr. Med. 2009, 15, 472–476.
- Adisakwattana, S.; Sookkongwaree, K.; Roengsumran, S.; Petsom, A.; Ngamrojnavanich, N.; Chavasiri, W.; Deesamer, S.; Yibchok-Anun, S. Structure-activity relationships of trans-cinnamic acid derivatives on α-glucosidase inhibition. Bioorg. Med. Chem. Lett. 2004, 14, 2893–2896.
- Adisakwattana, S. Cinnamic acid and its derivatives: Mechanisms for prevention and management of diabetes and its complications. Nutrients 2017, 9, 163.
- Adisakwattana, S.; Moonsan, P.; Yibchok-Anun, S. Insulin-releasing properties of a series of cinnamic acid derivatives in vitro and in vivo. J. Agric. Food Chem. 2008, 56, 7838–7844.
- Adisakwattana, S.; Hsu, W.H.; Yibchok-Anun, S. Mechanisms of p-Methoxycinnamic acid-induced increase in insulin secretion. Horm. Metab. Res. 2011, 43, 766–773.
- Adisakwattana, S.; Roengsamran, S.; Hsu, W.H.; Yibchok-Anun, S. Mechanisms of antihyperglycemic effect of p-methoxycinnamic acid in normal and streptozotocin-induced diabetic rats. Life Sci. 2005, 78, 406–412.
- Yibchok-Anun, S.; Adisakwattana, S.; Moonsan, P.; Hsu, W.H. Insulin-secretagogue activity of p-methoxycinnamic acid in rats, perfused rat pancreas and pancreatic β-cell line. Basic Clin. Pharmacol. Toxicol. 2008, 102, 476–482.
- Ambika, S.; Saravanan, R.; Thirumavalavan, K. Antidiabetic and antihyperlipidemic effect of p-hydroxycinnamic acid on streptozotocin-induced diabetic Wistar rats. Biomed. Aging Pathol. 2013, 3, 253–257.
- Fernandez-Martinez, E.; Bobadilla, R.; Morales-Rios, M.; Muriel, P.; Perez-Alvarez, V. Trans-3-Phenyl-2-Propenoic Acid (Cinnamic Acid) Derivatives: Structure-Activity Relationship as Hepatoprotective Agents. Med. Chem. 2007, 3, 475–479.
- Li, C.; Li, L.; Yang, C.; Zhong, Y.; Wu, D.; Shi, L.; Chen, L.; Li, Y. Biochemical and Biophysical Research Communications Hepatoprotective effects of Methyl ferulic acid on alcohol-induced liver oxidative injury in mice by inhibiting the NOX4/ROS-MAPK pathway. Biochem. Biophys. Res. Commun. 2017, 493, 277–285.
- Kim, S.R.; Kim, Y.C. Neuroprotective phenylpropanoid esters of rhamnose isolated from roots of Scrophularia buergeriana. Phytochemistry 2000, 54, 503–509.
- Kim, S.R.; Sung, S.H.; Jang, Y.P.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. E-p-Methoxycinnamic acid protects cultured neuronal cells against neurotoxicity induced by glutamate. Br. J. Pharmacol. 2002, 135, 1281–1291.
- Kim, S.R.; Kang, S.Y.; Lee, K.Y.; Kim, S.H.; Markelonis, G.J.; Oh, T.H.; Kim, J.C. Anti-amnestic activity of E-p-methoxycinnamic acid from Scrophularia buergeriana. Cogn. Brain Res. 2003, 17, 454–461.
- Zanyatkin, I.; Stroylova, Y.; Tishina, S.; Stroylov, V.; Melnikova, A.; Haertle, T.; Muronetz, V. Inhibition of Prion Propagation by 3,4-Dimethoxycinnamic Acid. Phyther. Res. 2017, 31, 1046–1055.
- Maurya, D.K.; Paul, T.; Devasagayam, A. Ferulic Acid Inhibits Gamma Radiation-Induced DNA Strand Breaks and Enhances the Survival of Mice. Cancer Biother. Radiopharm. 2013, 28, 51–57.
- Baskaran, N.; Manoharan, S.; Balakrishnan, S.; Pugalendhi, P. Chemopreventive potential of ferulic acid in 7,12-dimethylbenz[a]anthracene-induced mammary carcinogenesis in Sprague—Dawley rats. Eur. J. Pharmacol. 2010, 637, 22–29.
- Mori, H.; Kawabata, K.; Yoshimi, N.; Tanaka, T.; Murakami, T.; Okada, T.; Murai, H. Chemopreventive effects of ferulic acid on oral and rice germ on large bowel carcinogenesis. Anticancer Res. 1999, 19, 3775–3778.
- Sivagami, G.; Karthikkumar, V.; Balasubramanian, T.; Nalini, N. The modulatory influence of p-methoxycinnamic acid, an active rice bran phenolic acid, against 1,2-dimethylhydrazine-induced lipid peroxidation, antioxidant status and aberrant crypt foci in rat colon carcinogenesis. Chem. Biol. Interact. 2012, 196, 11–22.
- Gunasekaran, S.; Venkatachalam, K.; Namasivayam, N. P-Methoxycinnamic acid, an active phenylpropanoid induces mitochondrial mediated apoptosis in HCT-116 human colon adenocarcinoma cell line. Environ. Toxicol. Pharmacol. 2015, 40, 966–974.
- Santoso, B.S.A.; Khotib, J. Oil Fraction from Kaempferia galanga Alcoholic Extract Increases Apoptosis Activity in Mice Colon Cancer. In Proceedings of the Research and Application on Traditional Complementary and Alternative Medicine in Health Care (TCAM), Surakarta, Indonesia, 22–23 June 2012; pp. 66–72.
- Omar, M.N.; Hasali, N.H.M.; Yarmo, M.A. Cytotoxicity activity of biotransformed ethyl p-methoxycinnamate by Aspergillus niger. Orient. J. Chem. 2016, 32, 2731–2734.
- Gunasekaran, S.; Venkatachalam, K.; Namasivayam, N. Anti-inflammatory and anticancer effects of p-methoxycinnamic acid, an active phenylpropanoid, against 1,2-dimethylhydrazine-induced rat colon carcinogenesis. Mol. Cell. Biochem. 2019, 451, 117–129.
- Kern, S.M.; Bennett, R.; Mellon, F.; Kroon, P.; Garcia, M. Absorption of Hydroxycinnamates in Humans after High-Bran Cereal Consumption. J. Agric. Food Chem. 2003, 51, 6050–6055.
- Zhao, Z.; Egashira, Y.; Sanada, H. Ferulic acid is quickly absorbed from rat stomach as the free form and then conjugated mainly in liver. J. Nutr. 2004, 134, 3083–3088.
- Yang, C.; Tian, Y.; Zhang, Z.; Xu, F.; Chen, Y. High-performance liquid chromatography–electrospray ionization mass spectrometry determination of sodium ferulate in human plasma. J. Pharm. Biomed. Anal. 2007, 43, 945–950.
- Wen, X.; Walle, T. Methylated flavonoids have greatly improved intestinal absorption and metabolic stability. Drug Metab. Dispos. 2006, 34, 1786–1792.
- Rook, M. p-Methoxycinnamate and Its Metabolite. J. Pharm. Sci. 1968, 57, 6–9.




