1000/1000
Hot
Most Recent

Bisindoles are structurally complex dimers and are intriguing targets for partial and total synthesis. They exhibit stronger biological activity than their corresponding monomeric units. Bisindole alkaloids are naturally occurring alkaloids containing two indole nuclei and are the products of late-stage biosynthetic processes in higher plants by combining two monomeric units. Depending on the monomeric units involved, bisindoles can be a homo- or heterodimer. As a result, bisindole alkaloids comprise much higher structural complexity than both of the monomeric units that comprise them. Alstonia, a major genus in the Apocynaceae family of plants, has more than 150 species and is found all over the world. Robert Brown named it in 1811 in honor of Charles Alston (1685–1760), an eminent botanist at the University of Edinburgh. The Alstonia genus’ trees and shrubs are prevalent in the tropical and subtropical parts of Africa, Asia, and Australia. They contribute significant pharmacological activity, including anticancer, antileishmanial, antimalarial, antitussive, antiviral, antiarthritic, and antibacterial activities.
Nature has been a substantial and sustainable pool of biologically active compounds. Since ancient times natural product extracts (in crude form) have been used in traditional and folk medicines in many countries. In modern times pure (isolated) natural products and their derivatives play an important role in drug discovery, as indicated by their prevalence in approved drugs for clinical use. Out of the 1881 newly FDA-approved drugs over the last four decades (1 January 1981 to 30 September 2019), a significant portion comprising 506 (26.9%) were either natural products or derived from or inspired by natural products [1]. It is expected that the advent of modern and innovative technologies such as computational software, cheminformatics, artificial intelligence, automation, and quantum computing will further boost natural product-based drug discovery. A synergy among these technological milestones would accelerate hit to lead to clinic pathways of drug discovery, and natural products are expected to remain an important source [2]. Moreover, pharmacophores and their unique stereochemical interactions with natural products may stimulate more demanding targets such as protein–protein interactions in the near future and open up a new avenue in modern drug discovery [3]. The majority of biologically active natural products are produced in plants, known traditionally as medicinal plants. Alkaloids, the most important class of natural products with structural diversity and significant pharmacological effects, are mainly found in higher plants such as the Apocynaceae, Ranunculaceae, Papaveraceae, and Leguminosae families [4]. These natural products, along with flavonoids, fatty acids, etc., are the major classes of secondary metabolites that are believed to be parts of the plants’ defense mechanism. To date, many monoterpenoid indole and bisindole alkaloids have been found in the Alstonia genus [5]. Modern clinical application of many of these alkaloids are similar to their traditional or folklore applications; for example, cocaine and morphine were used as anesthetics while caffeine and nicotine were used as stimulants [6]. Recently, Fielding et al. illustrated that several anti-coronavirus alkaloids showed potential therapeutic value against severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) in their in silico studies [7].
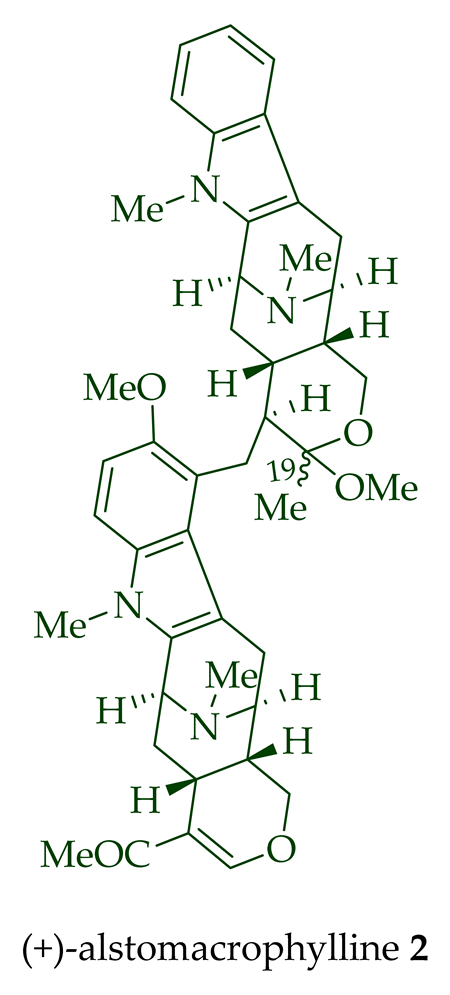
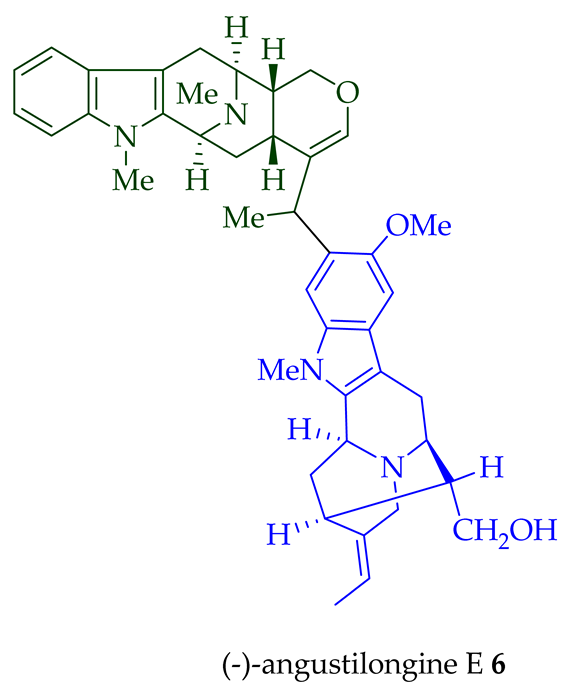
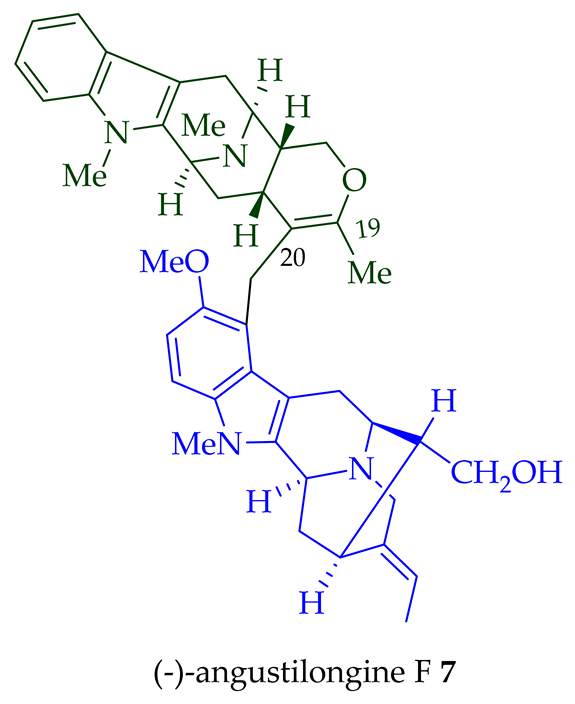
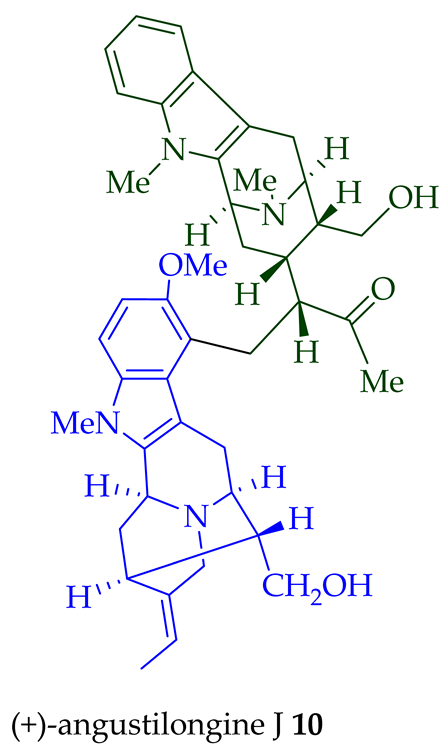
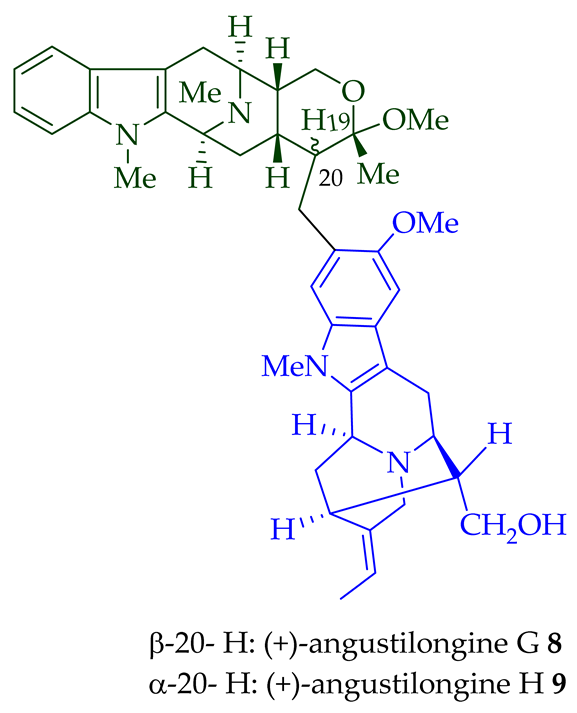
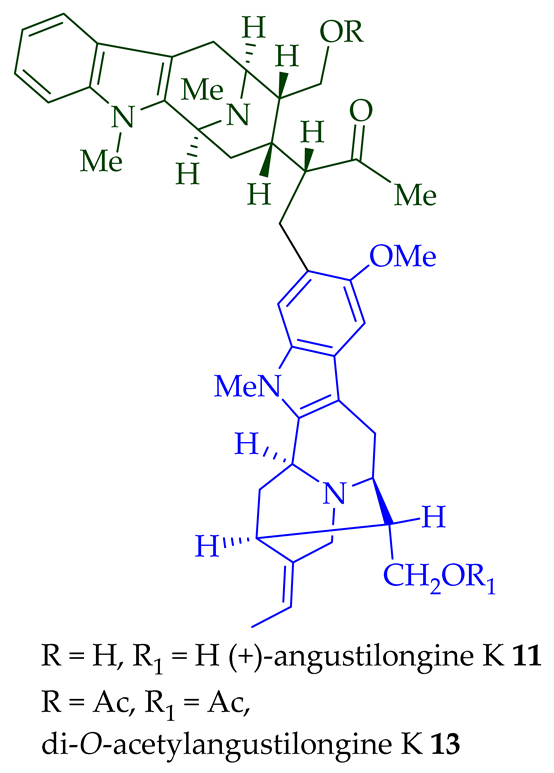
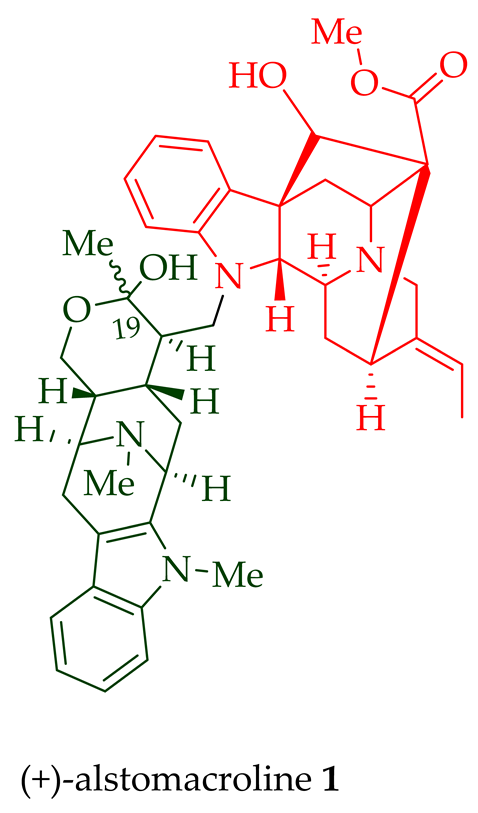
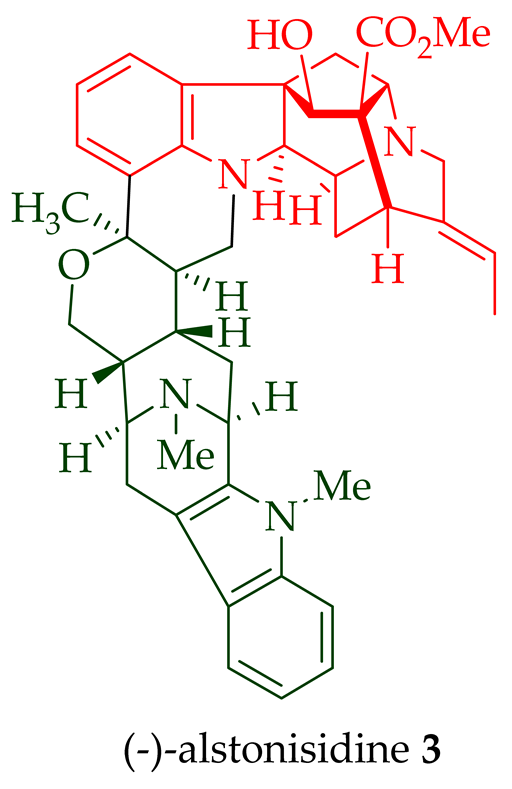
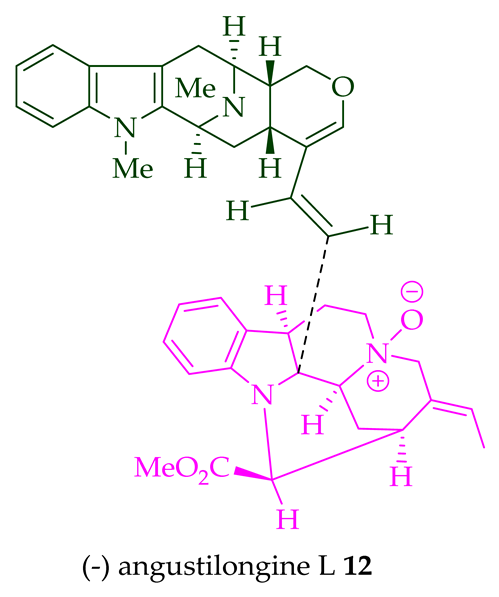
Among various species of the Alstonia genus, A. macrophylla and A. angustifolia are the two major sources of bisindole alkaloids discussed herein (vide infra, Table 1). (+)-Alstomacroline 1, a bisindole alkaloid consisting of a macroline and an ajmaline unit (monomeric units not shown here: please visit https://doi.org/10.3390/molecules26113459 for details), was isolated from the bark of A. macrophylla [12] and the leaves, stem-bark, and root-bark extracts of A. scholaris, A. glaucescens, and A. macrophylla[13][14]. (+)-Alstomacrophylline 2 (macroline–macroline-type) was isolated from the bark of A. macrophylla [12] and the leaves, stem-bark, and root-bark extracts of A. scholaris, A. glaucescens, and A. macrophylla [14]. (-) Alstonisidine 3, which contains a quebrachidine and a macroline unit, was isolated from the bark of A. muelleriana [15][16]. The structure of (-)-alstonisidine 3 was confirmed by X-ray crystallographic data [17]. Yeap et al. recently isolated seven novel bisindoles from the methanol extract of the stem-bark of Malayan A. penangiana [18]. This includes (-)-angustilongine E 6, (-)-angustilongine F 7, (+)-angustilongine G 8, (+)-angustilongine H 9, (+)-angustilongine J 10, (+)-angustilongine K 11, and (-)-angustilongine L 12 (macroline–pleiocarpamine type). (+)-Angustilongine K 11 was converted into (+)-di-O-acetylangustilongine K 13 by stirring it with 10 equivalents of pyridine and 15 equivalents of acetic anhydride for 6 h at room temperature in 95% yield [18]. Among those, angustilongine G 8 and angustilongine H 9 are C-19 methyl substituted [19] bisindoles. The structures of the angustilongines were confirmed by various spectroscopic data, including 1H NMR, 13C NMR, 2D NMR, IR, and HRMS by Yeap et al. [18]. Angustilongine E 6, angustilongine F 7, angustilongine G 8, angustilongine H 9, angustilongine J 10, and angustilongine K 11 are macroline–sarpagine coupled bisindoles. Angustilongine G 8 and angustilongine H 9 differ in stereochemistry only at the C-20 position.
Table 1. Structures of bisindole alkaloids from Alstonia species including semi-synthetic derivatives.
| Macroline-macroline type | ||
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
| Macroline-sarpagine type | ||
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
| Macroline-ajmaline type | ||
 |
 |
|
| Macroline-pleiocarpamine type | ||
 |
 |
 |
 |
 |
 |
 |
||
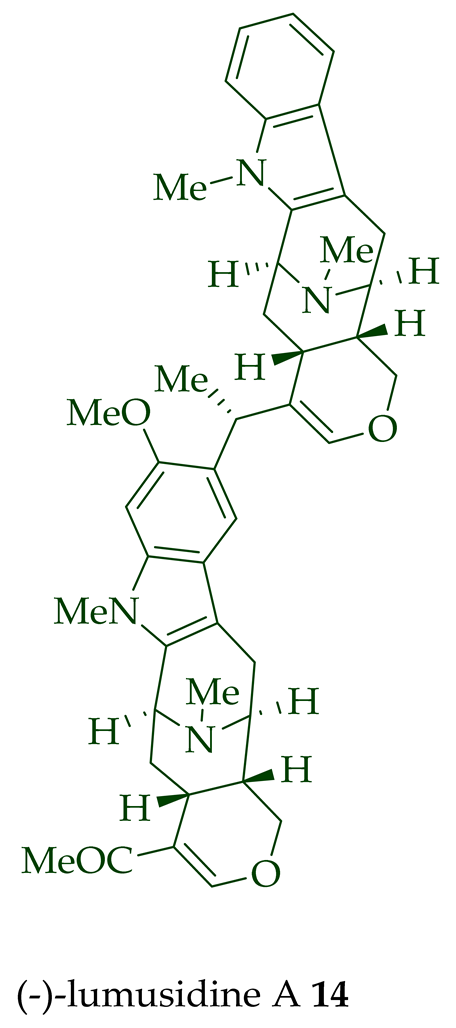
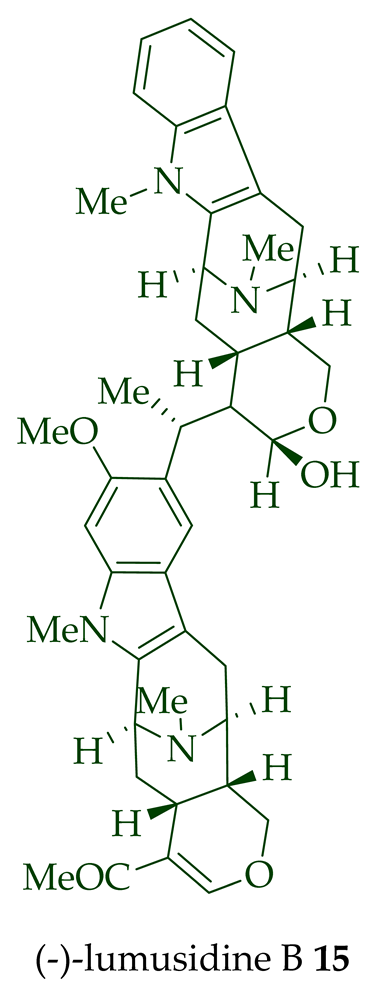
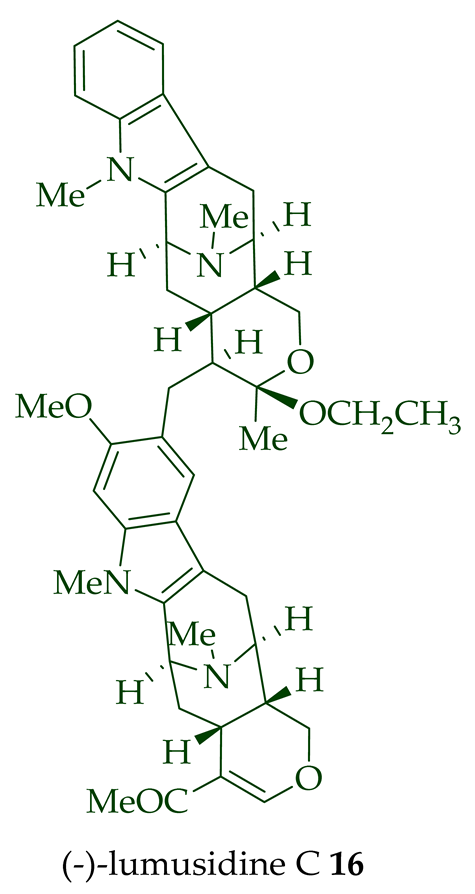
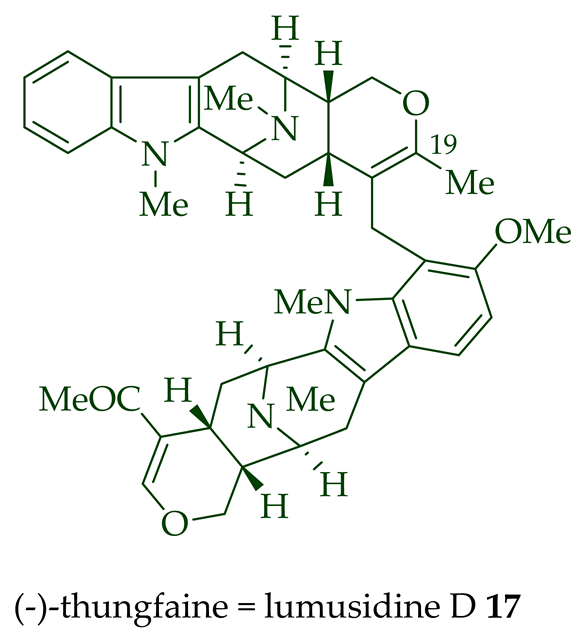
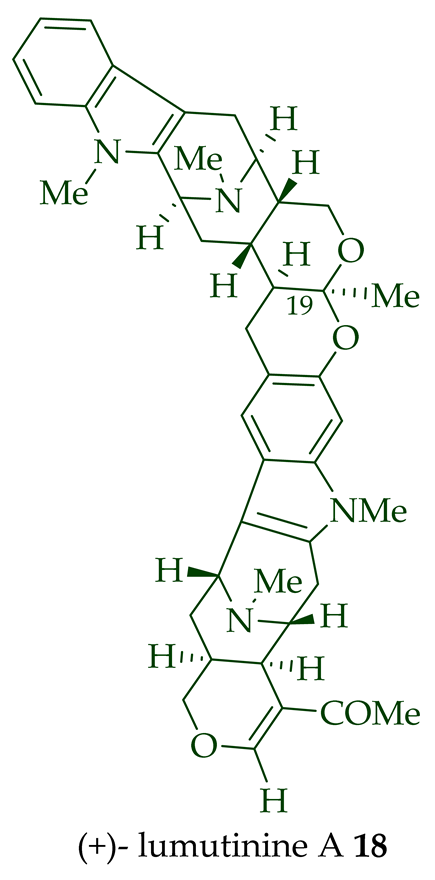
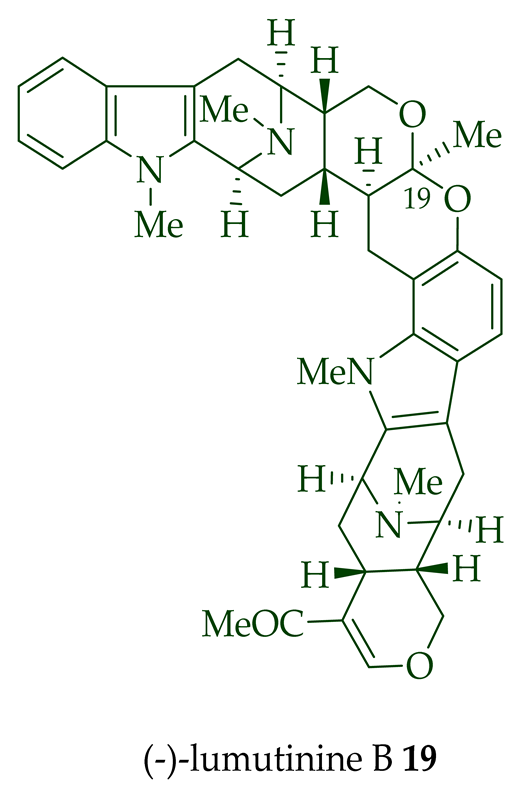
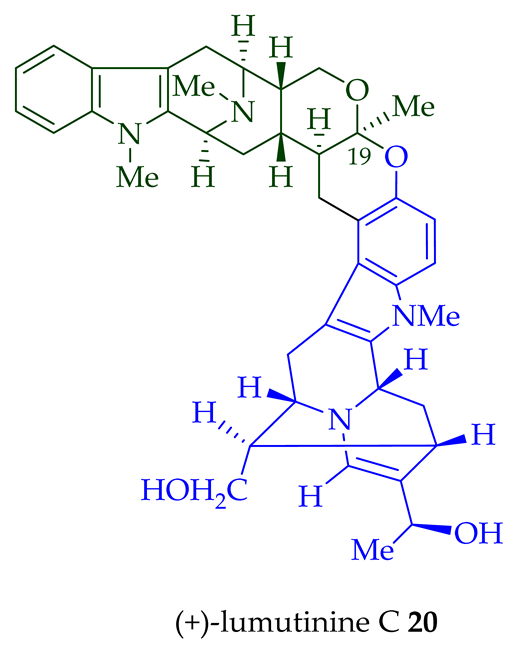
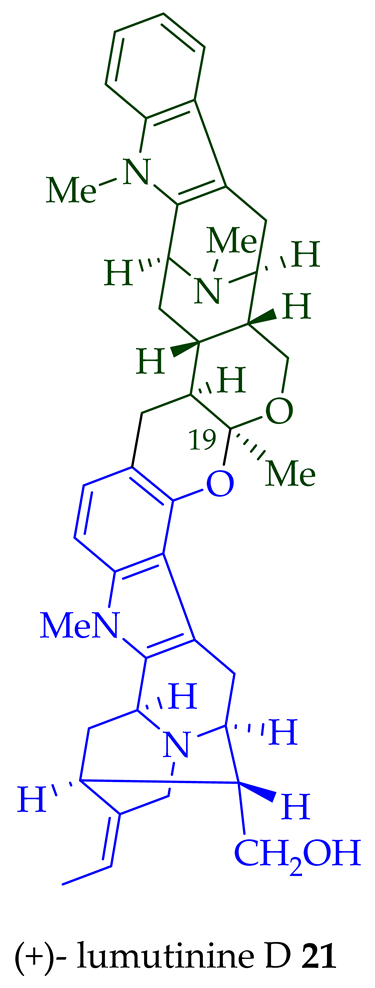
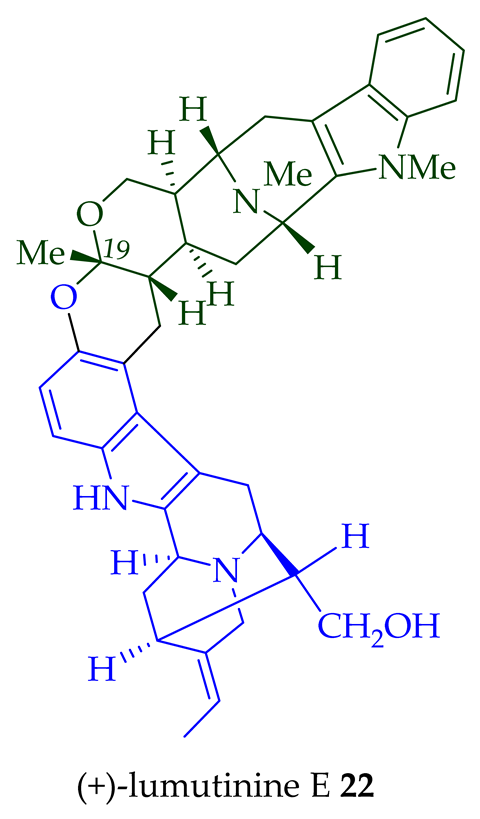
Two macroline units are contained in (-)-lumusidine A 14, (-)-lumusidine B 15, (-)-lumusidine C 16, and (-)-lumusidine D 17 bisindoles. They were isolated from the stem-bark of A. macrophylla and the structures were confirmed via NMR spectroscopy, mass spectrometry, UV spectroscopy, and X-ray crystallography [12]. After isolation, the group of Kam et al. converted oily (-)-lumusidine A 14, (-)-lumusidine B 15, and (-)-lumusidine D 17 into the corresponding crystalline dimethyl diiodide salts (structures not shown) by treatment with an excess of iodomethane for 24 h. The crystalline salts were employed to obtain X-ray crystallographic data to elucidate the exact stereochemical confirmation [12]. (-)-Lumusidine D 17 is also known as thungfaine [20]. (+)-Lumutinine A 18, (-)-lumutinine B 19, (+)-lumutinine C 20, and (+)-lumutinine D 21 are linearly fused bisindoles isolated from the stem-bark of A. macrophylla as a light yellowish oil [21]. (+)-Lumutinine A 18 and (-)-lumutinine B 19 are macroline–macroline-type bisindoles, while (+)-lumutinine C 20, (+)-lumutinine D 21, and (+)-lumutinine E 22 are macroline–sarpagine-type bisindoles. The structures of the lumutinines were elucidated using spectroscopic means including 1D and 2D NMR, IR, as well as mass spectrometric analysis [21]. The structure of (+)-lumutinine D 21 was confirmed by X-ray crystallographic data [22]. (+)-Lumutinine E 22, a macroline–sarpagine-type bisindole, was isolated from the stem-bark of A. angustifolia [23].
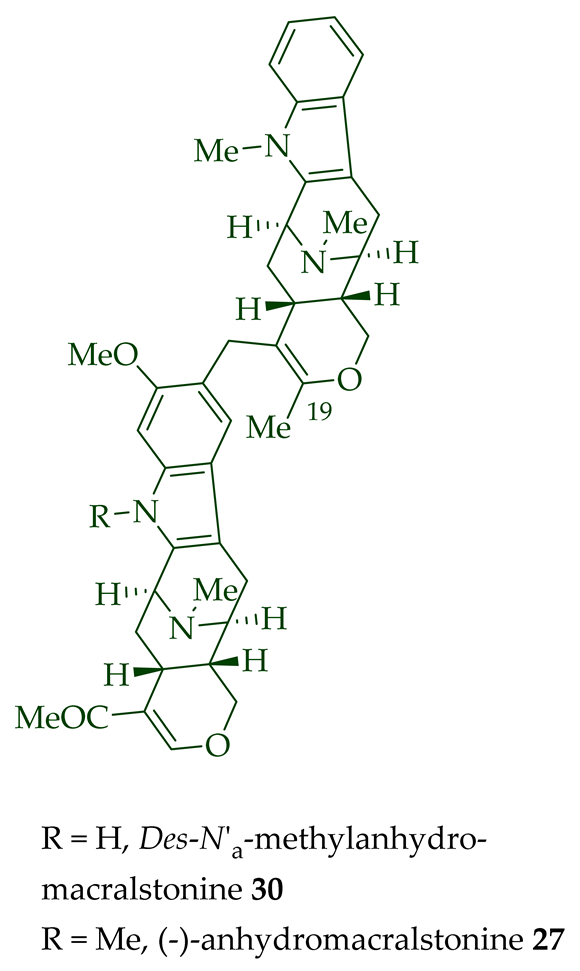
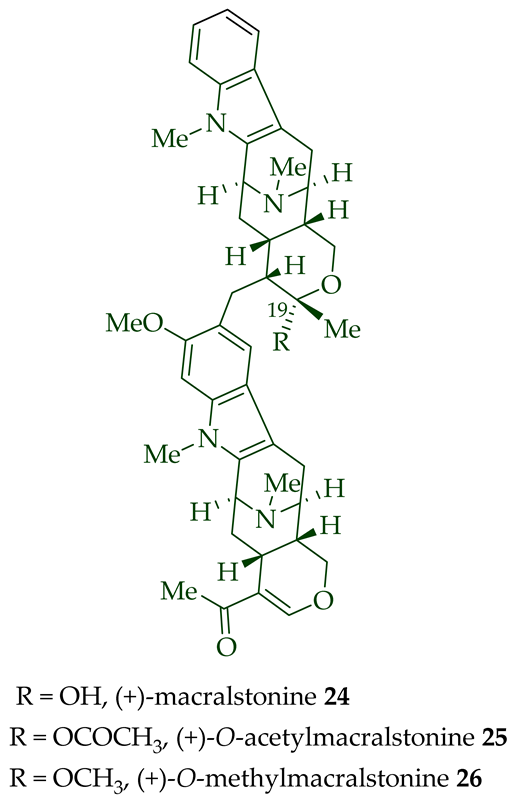
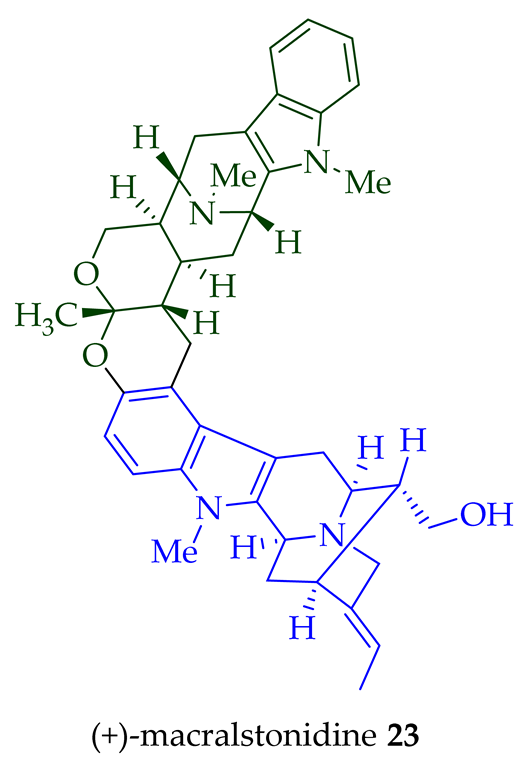
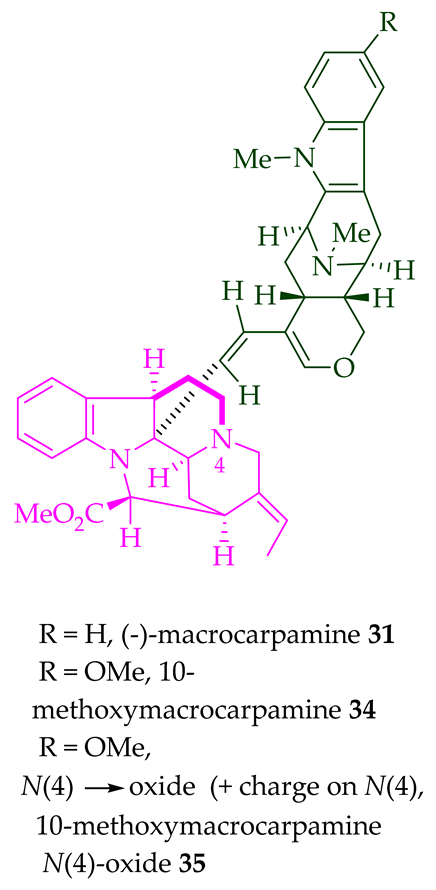
(+)-Macralstonidine 23 (macroline–sarpagine-type) was isolated from the bark of A. macrophylla [24][25], as well as from A. somersentenis [24] and A. spectabilis [26]. (+)-Macralstonine 24 was isolated from the leaves, stem-bark, and root-bark extracts of A. scholaris, A. glaucescens, and A. macrophylla extracts [14], A. macrophylla [24][25][27][28][29], A. muelleriana [30], A. angustifolia [31], as well as from A. glabriflora [26]. The structure of (+)-macralstonine 24 was confirmed by various NMR spectroscopy, mass spectrometry, and X-ray crystallography [28]. The (+)-macralstonine 24-related bisindole, (+)-O-acetylmacralstonine 25, was isolated from the leaves, stem-bark, and root-bark extracts of A. scholaris, A. glaucescens, and A. macrophylla [14]. Also, (+)-O-methylmacralstonine 26 was isolated from the leaves, stem-bark, and root-bark of A. scholaris, A. glaucescens, and A. macrophylla extracts [14]. (-)-Anhydromacralstonine 27 was isolated from the stem-bark of A. angustiloba [23] and contains (-)-alstophylline and (+)-macroline as monomeric units. Another (-)-alstophylline 28 and (+)-macroline monomeric bisindole, (+)-Des-N’a-methylanhydromacralstonine 30, was isolated from the bark of A. muelleriana [14][30], the stem-bark of A. angustifolia [28], and A. glabriflora [26]. (+)-Macrocarpamine 31, a hetero-dimeric bisindole containing a (+)-pleiocarpamine and (-)-anhydromacrosalhinemethine monomeric unit was isolated from the leaves, stem-bark, and root-bark of A. scholaris, A. glaucescens, and A. macrophylla [14], as well as the stem-bark of A. angustifolia [23]. 10-Methoxymacrocarpamine 34 and 10-methoxymacrocarpamine N4′-oxide 35 are structurally related bisindoles. These bisindoles were isolated from A. angustifolia leaves [31].
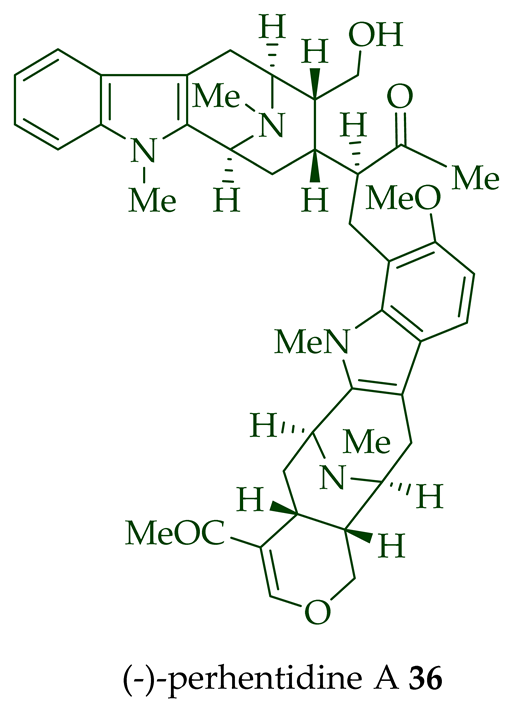
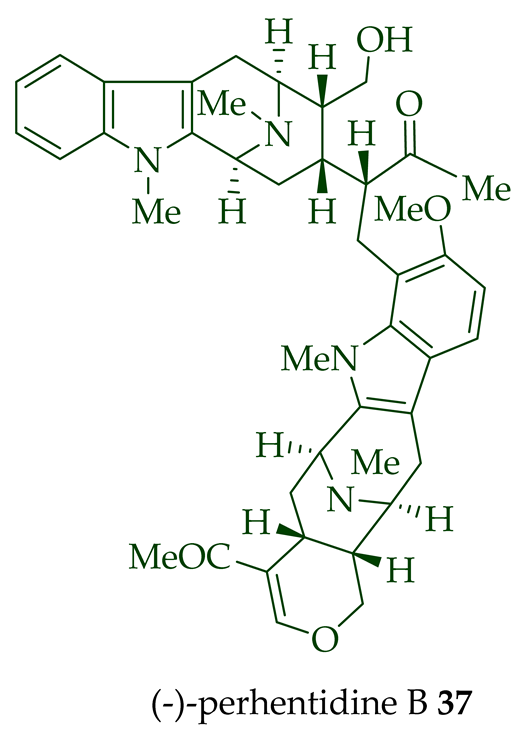
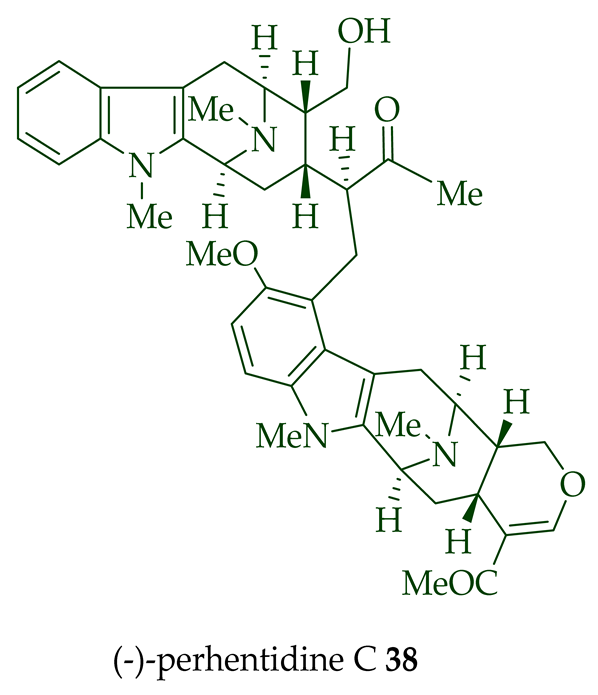
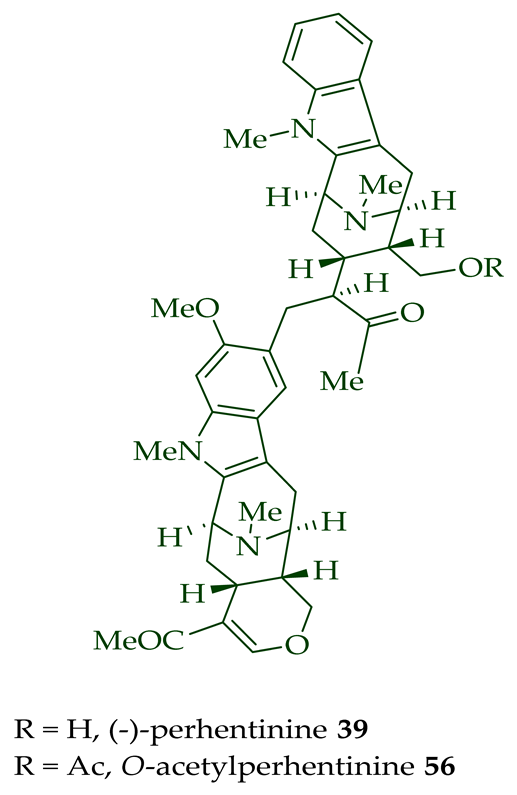
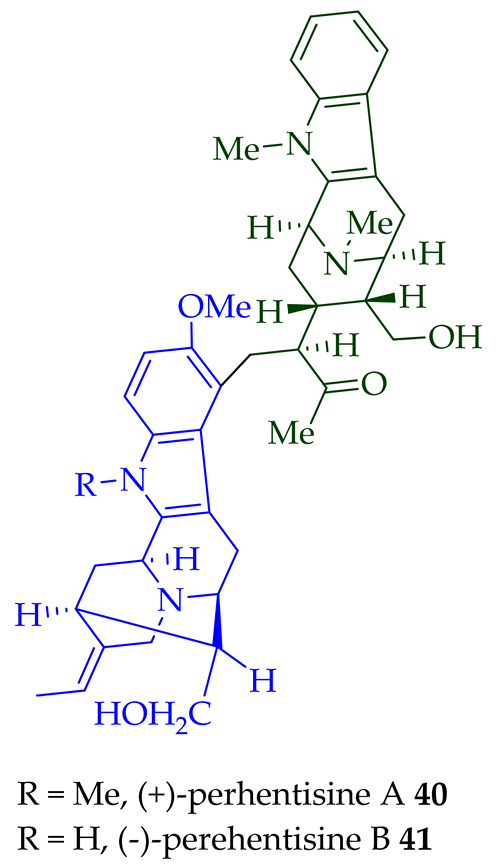
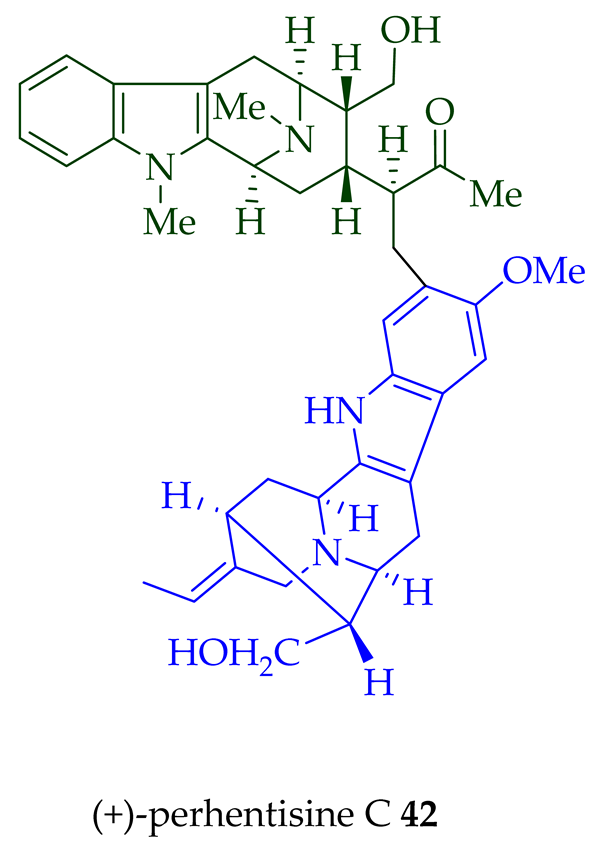
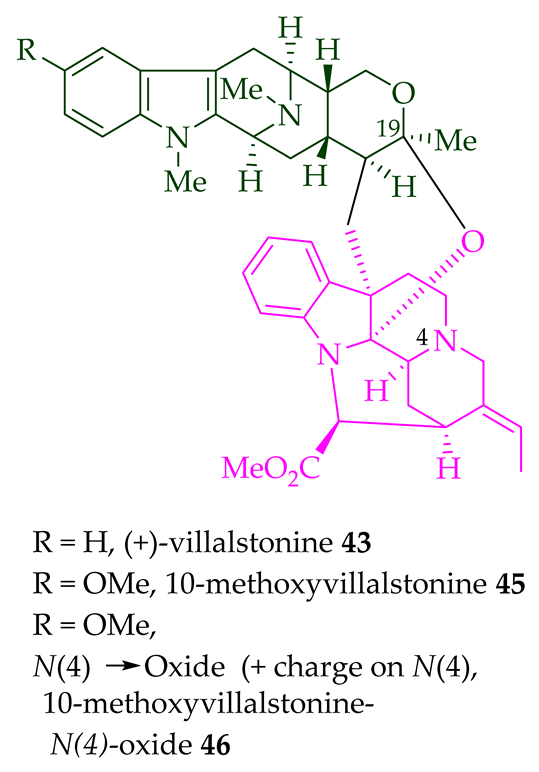
Lim et al. [28] reported the isolation of new macroline–macroline-type bisindoles, (-)-perhentidine A 36, (-)-perhentidine B 37, and (-)-perhentidine C 38 from the ethanolic extract of the stem-bark of Malayan A. macrophylla and A. angustifolia. The structurally related bisindole, (-)-perhentinine 39 (macroline–macroline-type), was isolated from the stem-bark and leaves of A. macrophylla and the leaves of A. angustifolia [12]. The exact structure of (-)-perhentinine 39 was confirmed by X-ray crystallography, converting it into the dimethyl diiodide salt by treating it with an excess of iodomethane. The X-ray crystallographic data of (-)-perhentinine 39 also helped in the structural characterizations of perhentidines A–C (36–38) [28]. Tan et al. isolated three macroline–sarpagine-type bisindoles: (+)-perhentisine A 40, (-)-perhentisine B 41, and (+)-perhentisine C 42 from the stem-bark of A. angustifolia as a light yellow-colored oil together with other bisindoles [23]. The structures of these bisindoles were also elucidated using various NMR and MS techniques [23]. (+)-Villastonine 43, a macroline–pleiocarpamine-type bisindole, was isolated from the stem-bark, root-bark, and leaves of various Alstonia species, including A. spectabilis [24] and A. muelleriana [32] by LeQuesne et al., A. macrophylla [25][27][33], and A. angustifolia [31][34]. Schmid et al. elucidated the structure of (+)-villalstonine 43 by spectroscopic means, accompanied by degradation, and Nordman et al. confirmed the structure by X-ray crystallography [33][35].
Table 2. Isolation of bisindoles from various part' of Alstonia species.
| Bisindoles | Alstonia Species |
Morphology and References |
|---|---|---|
| (+)-Alstomacroline 1 | A. scholaris, A. glaucescens, and A. macrophylla extracts | Leaves, stem-bark, and root-bark [13][14] |
| A. macrophylla | Bark [12] | |
| (+)-Alstomacrophylline 2 | A. macrophylla | Bark [13] |
| A. scholaris, A. glaucescens, and A. macrophylla extracts | Leaves, stem-bark, and root-bark [14] | |
| (−) Alstonisidine 3 | A. muelleriana | Bark [15][16] |
| (−)-Angustilongine E 6 | A. penangiana | Stem-bark [18] |
| (−)-Angustilongine F 7 | A. penangiana | Stem-bark [18] |
| (+)-Angustilongine G 8 | A. penangiana | Stem-bark [18] |
| (+)-Angustilongine H 9 | A. penangiana | Stem-bark [18] |
| (+)-Angustilongine J 10 | A. penangiana | Stem-bark [18] |
| (+)-Angustilongine K 11 | A. penangiana | Stem-bark [18] |
| (−)-Angustilongine L 12 | A. penangiana | Stem-bark [18] |
| (−)-Anhydromacralstonine 27 | A. angustifolia | Stem-bark [23] |
| (+)-Des-N’a-Methylanhydromacralstonine 30 | A. muelleriana | Bark [14] |
| A. angustifolia | Stem-bark [28] | |
| A. muelleriana | Leaves, stem-bark and root-bark [14][30] | |
| (−)-Lumusidine A 14 | A. macrophylla | Stem-bark [12] |
| (−)-Lumusidine B 15 | A. macrophylla | Stem-bark [12] |
| (−)-Lumusidine C 16 | A. macrophylla | Stem-bark [12] |
| (−)-Lumusidine D 17 | A. macrophylla | Stem-bark [12] |
| (+)-Lumutinine A 18 | A. macrophylla | Stem-bark [21] |
| (−)-Lumutinine B 19 | A. macrophylla | Stem-bark [21] |
| (+)-Lumutinine C 20 | A. macrophylla | Stem-bark [21] |
| (+)-Lumutinine D 19 | A. macrophylla | Stem-bark [21] |
| (+)-Lumutinine E 21 | A. angustifolia | Stem-bark [23] |
| (+)-Macralstonidine 23 | A. macrophylla | Bark [24][25] |
| A. somersentenis | Bark [24] | |
| A. spectabilis | Bark [26] | |
| (+)-Macralstonine 24 | A. scholaris, A. glaucescens, and A. macrophylla extracts | Leaves, stem-bark and root-bark [13] |
| (+)-O-Acetylmacralstonine 25 | A. macrophylla | [24][25][27][28][29] |
| A. angustifolia | [31] | |
| A. muelleriana | [30] | |
| A. glabriflora Mgf. | [26] | |
| A. scholaris, A. glaucescens, and A. macrophylla extracts | [14] | |
| (+)-O-Methylmacralstonine 26 | A. scholaris, A. glaucescens, and A. macrophylla extracts | [14] |
| (+)-Macrocarpamine 31 | A. scholaris, A. glaucescens, and A. macrophylla extracts | Leaves, stem-bark, and root-bark [14] |
| A. angustifolia | Stem-bark [23] | |
| 10-Methoxy macrocarpamine 34 | A. angustifolia | Leaves [36] |
| 10-Methoxy macrocarpamine 4′-N-oxide 35 | A. angustifolia | Leaves [36] |
| (−)-Perhentidine A 36 | A. macrophylla and A. angustifolia | Stem-bark [23][28] |
| (−)-Perhentidine B 37 | A. macrophylla and A. angustifolia | Stem-bark [28] |
| (−)-Perhentidine C 38 | A. macrophylla and A. angustifolia | Stem-bark [23][28] |
| (−)-Perhentinine 39 | A. macrophylla | Stem-bark [12] |
| A. angustifolia | Leaves [12] | |
| A. macrophylla | Leaves [12] | |
| (+)-Perhentisine A 40 | A. angustifolia | Stem-bark [23] |
| (−)-Perhentisine B 41 | A. angustifolia | Stem-bark [23] |
| (+)-Perhentisine C 42 | A. angustifolia | Stem-bark [23] |
| A. muelleriana | Leaves and stem-bark [32] | |
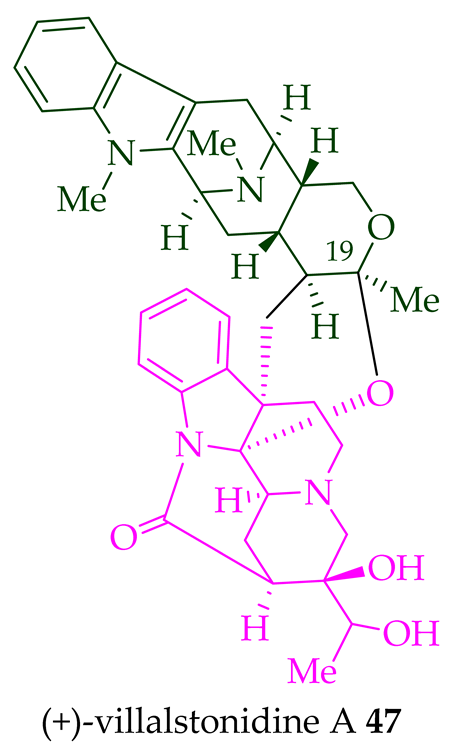
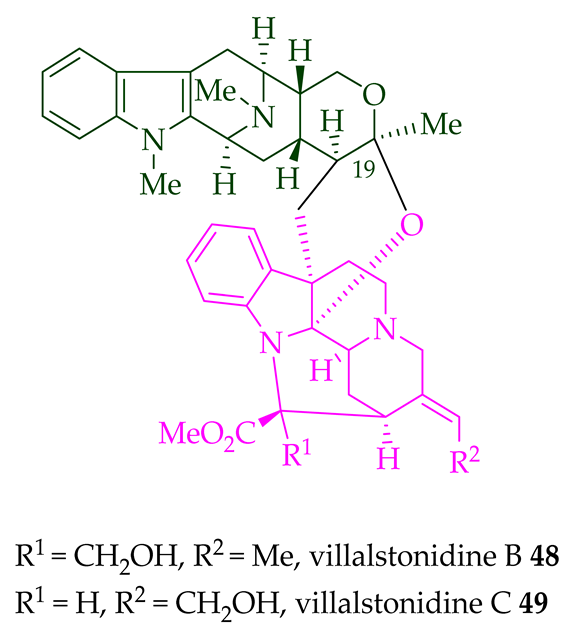
| (+)-Villalstonidine A 47 | A. angustifolia | Stem-bark [23] |
| (+)-Villalstonidine B 48 | A. angustifolia | Stem-bark [23] |
| A. macrophylla | Stem-bark [22] | |
| (+)-Villalstonidine C 49 | A. angustifolia | Stem-bark [23] |
| (+)-Villalstonidine D 50 | A. angustifolia | Stem-bark [23] |
| (+)-Villalstonidine E 51 | A. angustifolia | Stem-bark [23] |
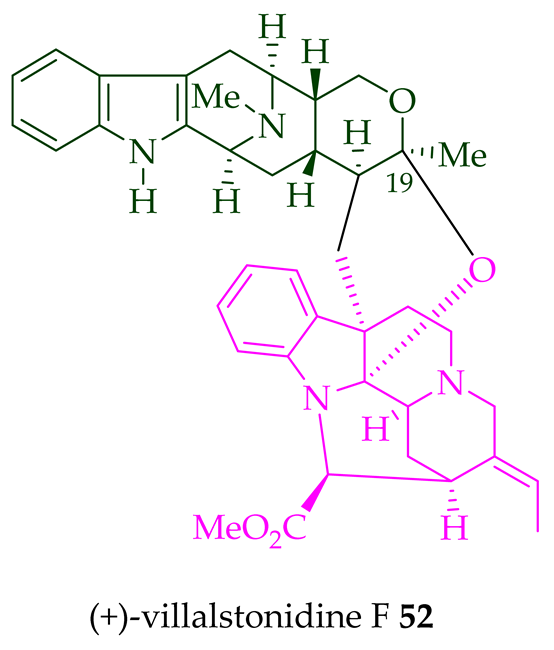
| (+)-Villalstonidine F 52 | A. macrophylla | Stem-bark [22] |
| (+)-Villalstonine 43 | A. angustifolia | Leaves and stem-bark [23][31][34][36] |
| A. macrophylla | [36] | |
| A. villosa | [36] | |
| A. verticillosa | [36] | |
| A. somersentensis | [36] | |
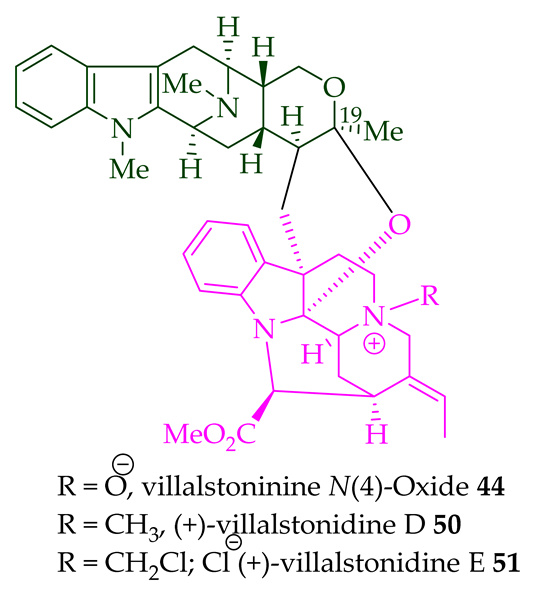
| Villalstonine N(4)-oxide 44 | A. angustifolia | Stem-bark [23] |
| A. scholaris, A. glaucescens, and A. macrophylla extracts | Leaves, stem-bark, and root-bark [14][25][27][33] | |
| (+)-10-Methoxy villalstonine 45 | A. angustifolia | Leaves [36] |
| 10-Methoxy villalstonine 4′-N-oxide 46 | A. angustifolia | Leaves [36] |
Villalstonine N(4)-oxide 44 was isolated from the stem-bark of A. angustifolia [23]. It was also isolated from the leaves, stem-bark, and root-bark of A. scholaris, A. glaucescens, and A. macrophylla extracts [14]. Moreover, two villalstonine 44-related bisindoles, (+)-10-methoxy villalstonine 45 and 10-methoxy villalstonine N(4)-oxide 46, were isolated from the leaves of A. angustifolia [36]. Villalstonidine A 47, villalstonidine B 48, villalstonidine C 49, villalstonidine D 50, villalstonidine E 51, (+)-villalstonidine F 52, and villalstonine N(4)-oxide 44 are macroline–pleiocarpamine-type bisindoles and are close in structure to villalstonine 43. Villalstonidines A–D (47–50) were isolated from the stem-bark of A. angustifolia [23]. Additionally, (+)-villalstonidine F 52 {N(1)-demethylderivative of villalstonine 43} and (+)-villalstonidine B 48 were isolated from the stem-bark of A. macrophylla [22].
Studies from various groups have shown that bisindoles have anticancer activity in different cell lines, including vincristine-resistant KB/VJ300 cells. Bisindoles are also reported to have other biological activities, including antiprotozoal activity against Plasmodium falciparum and antileishmanial activity against promastigotes of Entamoeba histolytica. The reported biological activity of bisindoles from various Alstonia species including the semi-synthetic derivatives reviewed herein are listed in Table 3. (+)-Alstomacroline 1 and (+)-alstomacrophylline 2 bisindoles were active against the K1 (multi-drug resistant) strain of P. falciparum with an IC50 1.12 ± 0.35 μM and IC50 1.10 ± 0.30 μM, respectively [37]. Newly isolated macroline-sarpagine-type bisindoles, (−)-angustilongines E 6, (-)-angustilongine F 7, (+)-angustilongine G 8, (+)-angustilongine H 9, (+)-angustilongine J 10, and (+)-angustilongine K 11 showed in-vitro growth inhibitory activity against human cancer cell lines, inclusive of KB, vincristine-resistant strains of KB, HCCT 116, PC-3, MDA-MB-231, LNCaP, MCF7, HT-29, and A549 cells with IC50 values ranging from 0.02 to 9.0 μM in the study from the group of Kam [18]. Kam et al. [12] also reported the anticancer activity of (−)-lumusidine A 14, (−)-lumusidine B 15, (−)-lumusidine C 16, and (−)-lumusidine D 17. These bisindoles were cytotoxic against KB/VJ300 cells ranging from IC50 0.16 to 5.03 μg/mL (μM) values with 0.12 μM vincristine added [12]. Lumutinine A 18, lumutinine B 19, lumutinine C 20, lumutinine D 21, and lumutinine E 22 exhibited moderate anticancer activity against KB/VJ300 cells ranging in IC50 value from 0.10 to 4.61 μg/mL (μM) again with 0.12 μM vincristine added in the studies from the same group (Kam) [12].
Table 3. Bioactivity of bisindoles (including semisynthetic derivatives) from Alstonia species.
Bisindoles |
Bioactivity |
References |
|---|---|---|
| (+)-Alstomacroline 1 | Antimalarial, with IC50 values of 1.12 ± 0.35 and 10.0 ± 0.4 μM against the K1 strain and T9-96 strain of P. falciparum, respectively. | [14] |
| (+)-Alstomacrophylline 2 | Antimalarial, with an IC50 value of 1.10 ± 0.30 μM against the K1 strain of P. falciparum. | [14] |
| Angustilongines E, F, G, H, J, and K (6–11) | Anticancer, cytotoxic against various human cancer cell lines including KB, vincristine-resistant KB, HCCT 116, PC-3, MDA-MB-231, LNCaP, MCF7, HT-29, and A549 cells with IC50 values ranging from 0.02 to 9.0 μM. | [18] |
| (−)-Lumusidine A, B, and C (14–16) | Anticancer, moderately cytotoxic in KB/VJ300 cells with IC50 values of 0.16, 0.70, and 1.19 μg/mL (μM), respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (−)-Lumusidine D 17 | Anticancer, cytotoxic in KB/VJ300 cells with an IC50 value of 5.03 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. |
[12] |
| Lumutinine A, B, C, D, and E (18–22) |
Anticancer, moderately cytotoxic in KB/VJ300 cells with IC50 0.21, 0.10, 4.61, 3.93, and 2.74 μg/mL (μM) values, respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (+)-Macralstonine 24 | Anticancer, strongly cytotoxic in KB/VJ300 cells with an IC50 1.71 μg/mL (μM) value. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| Antimalarial, active against the K1 strain of P. falciparum with an IC50 8.92 ± 2.95 μM value. | [14] | |
| (−)-Anhydromacralstonine 27 | Anticancer, moderately cytotoxic in KB/VJ300 cells with an IC50 value of 0.44 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. |
[12] |
| (+)-O-Acetyl macralstonine 25 | Antimalarial, with IC50 values 0.53 ± 0.09 and 12.4 ± 1.6 (μM) against the K1 strain and T9-96 strain of P. falciparum, respectively. | [14] |
| (+)-O-Methyl macralstonine 26 | Antimalarial, active against the K1 strain of P. falciparum with an IC50 0.85 ± 0.20 μM value. | [14] |
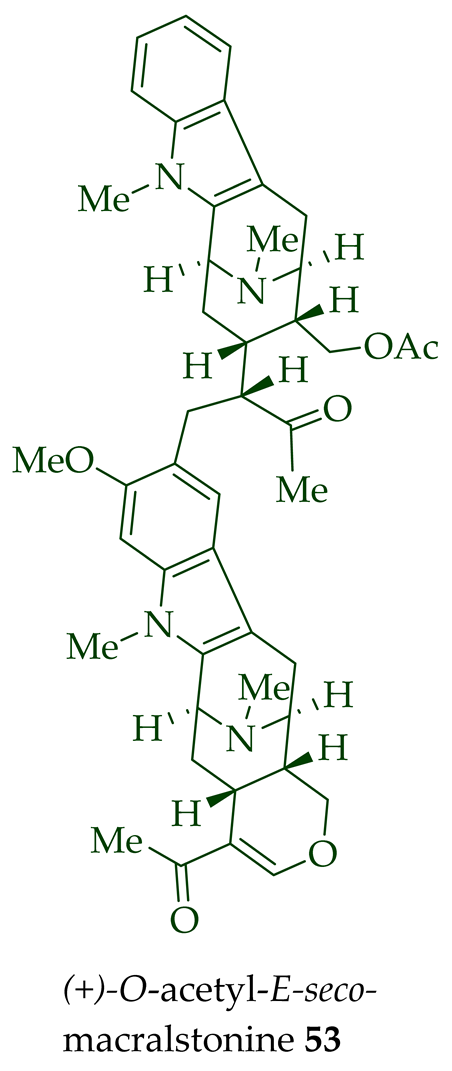
| O-Acetyl-E-seco-macralstonine 53 | Anticancer, strongly cytotoxic in KB/VJ300 cells with an IC50 value of 0.27 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (−)-Perhentidine A 36 and (-)-perhentidine B 37 | Anticancer, strongly cytotoxic in KB/VJ300 cells with IC50 values of 2.29 and 0.84 μg/mL (μM), respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
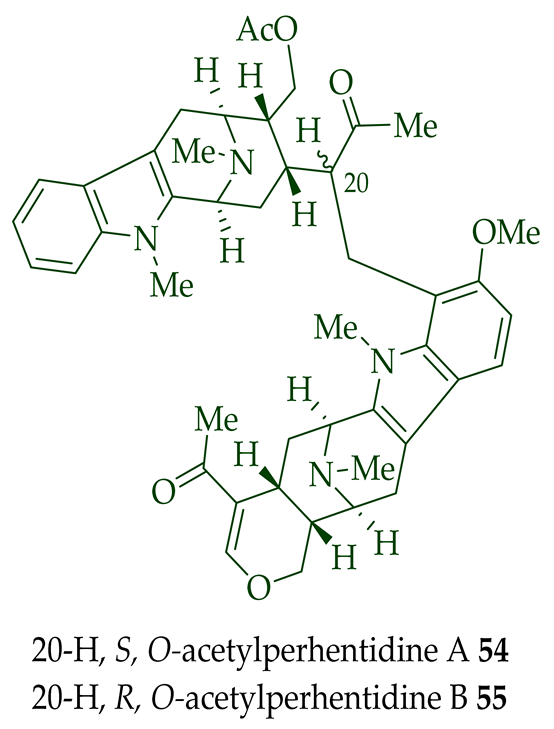
| (−)-O-Acetylperhentidine A 54 and (-)-O-Acetylperhentidine B 55 | Anticancer, strongly cytotoxic in KB/VJ300 cells with IC50 0.36 and 0.28 μg/mL (μM) values, respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (−)-Perhentinine 39 and O-Acetylperhentinine 56 | Anticancer, cytotoxic in KB/VJ300 cells with IC50 values of 0.52 and 0.30 μg/mL (μM), respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. |
[12] |
| (+)-Macralstonidine 23 | Anticancer, moderately cytotoxic in KB/VJ300 cells with an IC50 value of 0.13 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (+)-Macrocarpamine 21 | Anticancer, strongly cytotoxic in KB/VJ300 cells with an IC50 value of 0.53 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| Strong antimalarial activity against the K1 strain of P. falciparum with an IC50 value of 0.36 ± 0.06 μM. Active against the T9-96 strain of P. falciparum with an IC50 >39 μM value. |
[14] | |
| Strong antiprotozoal activity in vitro against E. histolytica and P. falciparum with ED50 8.12 (95% C.I.) μM and ED50 9.36 (95% C.I.) μM values, respectively. | [38] | |
| (+)-Villalstonine 43 | Anticancer, cytotoxic in KB/VJ300 cells with an IC50 value of 0.42 μg/mL (μM). The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| Anticancer, cytotoxic against the HT-29 cell line with an ED50 8.0 μM value (paclitaxel was used as the positive control). | [34] | |
| Antimalarial, with IC50 values of 0.27 ± 0.06 and 0.94 ± 0.07 μM against the K1 strain and T9-96 strain of P. falciparum, respectively. | [14] | |
| Antiamoebic activity against E. histolytica with an ED50 of 2.04 μM. | [38] | |
| Villalstonine N(4)-oxide 44 | Antileishmanial activity against promastigotes of L. mexicana with an IC50 value of 80.3 μM (amphotericin B was used as the positive control). | [34] |
| Antimalarial, active against the K1 strain of P. falciparum with an IC50 10.7 ± 1.9 (μM) value. | [14] | |
| (+)-Villalstonidine B 48 and (+)-villalstonidine F 52 |
Anticancer, strongly cytotoxic in KB/VJ300 cells with IC50 values of 0.35 and 5.64 μg/mL (μM), respectively. The assay with 0.12 μM added vincristine did not influence KB/VJ300 cell growth. | [12] |
| (+)-Villalstodinine D 50 | Antileishmanial, active against promastigotes of L. mexicana with an IC50 value of 120.4 μM (amphotericin B was used as the positive control). | [34] |
| (+)-Villalstonidine E 51 | Anticancer, cytotoxic against HT-29 cell lines with an ED50 6.5 μM value (paclitaxel was used as the positive control). | [34] |
| Antileishmanial against promastigotes of L. mexicana with an IC50 78 μM value (amphotericin B was used as the positive control). | [34] |
(+)-Macralstonine 24 was active against the K1 strain of P. falciparum (IC50 8.92 ± 2.95 μM). Notably, the derivatives of (+)-macralstonine 24 were more active than (+)-macralstonine 24 itself. (+)-O-methyl macralstonine 26 and (+)-O-acetyl macralstonine 25 demonstrated more potent activity against the K1 strain of P. falciparum with IC50 values of 0.85 ± 0.20 μM and IC50 0.53 ± 0.09 μM, respectively [14]. Likely, the functionalization facilitated the transportation of these bisindoles through the cell membranes of parasites and red blood cells, which would have enhanced the activity as lipophilicity rose [14]. (+)-O-acetyl macralstonine 25 and (+)-alstomacroline 1 were also somewhat active against the T9-96 strain of P. falciparum with IC50 values of 12.4 and 10.2 μM, respectively [14]. Heterodimeric alkaloid (−)-anhydromacralstonine 27 showed moderate cytotoxicity (IC50 value of 0.44 μg/mL (μM) in KB/VJ300 cells with 0.12 μM of vincristine added [12]). Another semisynthetic analog of macralstonine 24, the related O-acetyl-E-seco-macralstonine 53, showed strong anticancer activity. It was prepared by the reaction of macralstonine 24 with acetic anhydride/pyridine in DCM [12]. It demonstrated potent activity against vincristine-resistant KB/VJ300 cells with an IC50 value of 0.27 μg/mL (μM), with 0.12 μM of vincristine added to the assay [12]. (+)-Macralstonidine 23 was found to exhibit moderately active anticancer activity. It was active against KB/VJ300 cells with an IC50 value of 0.13 μg/mL (μM) [12].
(−)-Macrocarpamine 31 exhibited antiprotozoal and anticancer activity in various studies. It showed significant antiprotozoal activity in vitro in studies from Wright et al. against E. histolytica and P. falciparum with ED50 values of 8.12 (95% C.I.) μM and 9.36 (95% C.I.) μM, respectively [38]. Keawpradub et al. reported significant activity of (−)-macrocarpamine 31 against the K1 strain of P. falciparum with an IC50 value of 0.36 μM [14]. Furthermore, (−)-macrocarpamine 31 showed antimalarial activity against the T9-96 strain of P. falciparum with an IC50 > 39 μM. The ancient folklore use of the extracts from A. angustifolia in Malaya for treatment of malaria and dysentery is supported by these in vitro studies [39]. (−)-Macrocarpamine 31 was strongly cytotoxic in KB/VJ300 cells with an IC50 value of 0.53 μg/mL (μM) [12]. (−)-Perhentidine A 36 and (−)-perhentidine B 37 showed strong cytotoxicity against KB/VJ300 cells with IC50 values of 2.29 and 0.84 μg/mL (μM), respectively [12]. O-Acetylperhentidine A 54 and O-acetylperhentidine B 55 also exhibited strong cytotoxicity against KB/VJ300 cells with IC50 values of 0.36 and 0.28 μg/mL (μM), respectively [12]. (−)-Perhentidine A 36 and (−)-perhentidine B 37 were treated individually by dropwise addition of acetic anhydride in a py/DCM solution, followed by stirring at room temperature for 2 h to furnish the semisynthetic (−)-O-acetylperhentidine A 54, and (−)-O-acetylperhentidine B 55, respectively [12]. (−)-Perhentinine 39 and O-acetylperhentinine 56 were cytotoxic against KB/VJ300 cells with IC50 values of 0.52 and 0.30 μg/mL (μM), respectively, in the studies by Kam et al. [12].
(+)-Villalstonine 43 has demonstrated various biological activities including anticancer, antimalarial, and antiamoebic activity. (+)-Villastonine 43 was 1/15th as potent as chloroquine (antimalarial drug) against malaria [40]. It exhibited potent antiplasmodial activity against the multidrug-resistant K1 strain of P. falciparum with an IC50 value of 0.27 μM [14]. Wright et al. tested this compound for antiamoebic activity against E. histolytica [38]. (+)-Villalstonine 43 showed six times less activity (ED50 11.8 μM) than the antiamoebic drug emetine (ED50 2.04 μM). These results also concur with the use of various parts of the A. angustifolia plant from ancient times to treat malaria and amoebic dysentery [38]. Moreover, (+)-villalstonine 43 was cytotoxic against KB cells with an ED50 value of 11.6 μM [38]. (+)-Villalstonine 43 was also potent against the T9-96 strain of P. falciparum with an IC50 value of 0.94 μM. It also showed anticancer activity against the HT-29 cell line with an ED50 value of 8.0 μM (paclitaxel was the positive control). Also, it was cytotoxic against vincristine-resistant KB/VJ300 cells with an IC50 value of 0.42 μg/mL (μM) [12]. On the other hand, the derivative of (+)-villalstonine 43, villalstonine N(4)-oxide 44 was less potent (IC50 10.7 ± 1.9) than (+)-villalstonine 43 itself. The increase in the ionic charge (decrease in lipophilicity) might have reduced the ability of villalstonine N(4)-oxide 44 to cross through the cell membranes of red blood cells or the parasites, which if correct, would explain the weaker activity [14]. Villalstonine N(4)-oxide 44 is also an antileishmanial bisindole. It was active against promastigotes of Leishmania mexicana with an IC50 value of 80.3 μM [34]. (+)-Villalstonine 43-related alkaloids (+)-villalstonidine B 48 and (+)-villalstonidine F 52 were found to be strongly cytotoxic against KB/VJ300 cells with IC50 values of 0.35 and 5.64 μg/mL (μM), respectively [12]. (+)-Villalstonidine D 50 exhibited antileishmanial activity. It was active against promastigotes of L. mexicana with an IC50 value of 120.4 μM in a study from Pan et al. [34]. Another (+)-villalstonine 43-related alkaloid, (+)-villalstonidine E 51, demonstrated anticancer and antimalarial activity. It was cytotoxic against the HT-29 cell lines with an ED50 value of 6.5 μM and was active against promastigotes of L. mexicana with an IC50 value of 78 μM in the study from the same group [34]. Many bisindoles are yet to be screened for their activity because of the paucity of material; however, their significant role in future drug discovery should be considered.
Many Alstonia species are rich in indole alkaloids including bisindoles. Bisindole alkaloids including semisynthetic derivatives have been found to possess significant bioactivity, including anticancer, antileishmanial, and antimalarial properties, and thus are promising leads for nature-inspired drug discovery and development. Unnatural medicinal compounds formed by combining bioactive mismatched monomeric units can furnish novel medicinal compounds. Incorporating unnatural enantiomers of monomeric alkaloids into bisindoles can provide access to novel and unnatural bioactive compounds that may have better activity and in vivo stability depending on their metabolism. Several bisindoles along with their corresponding monomeric units has been successfully synthesized (please visit https://doi.org/10.3390/molecules26113459 for details) however; many bisindoles still await their total synthesis as well as biological screening.