
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kristina Habschied | + 2238 word(s) | 2238 | 2021-06-15 10:03:53 | | | |
| 2 | Nora Tang | + 36 word(s) | 2274 | 2021-06-15 10:37:10 | | |
Video Upload Options
Polyphenolic molecules can be found in different stages during the brewing process and react with proteins: during wort boiling, they form the hot break; during cooling, they form the cold break; and during post-fermentation, they are involved in the formation of chill haze and permanent hazes and facilitate the removal of undesirable compounds with filtration. However, they tend to react with proteins in packaged beer and form undesirable haze after the expiration date. As mentioned before, polyphenols can enter beer from hop and malt.
1. Introduction
Flavor stability is, besides colloidal stability, one of the most important indicators of beer quality. Some types of beer (i.e., Indian Pale Ale) are bound to contain strong hoppy notes and bitterness originating from polyphenols in hops or barley. However, the emergence of undesirable notes can be a result of a faulty lead brewing process, whether due to the over-dosage of hop (acids), low temperatures while dry hopping, polyphenols, or simply staling. The ongoing craft beer boom has aimed to present different types of beer to the broader public, and this includes hoppy and bitter beers (mostly ales) that differ from the industrial lagers by increased bitterness [1].
As such, polyphenols can provide both bitterness and astringency, depending on their degree of isomerization [2]. The consumers’ perception of bitterness depends on different factors and is very complex. The range of molecules that elicit bitter responses is very wide [3][4]; for example, the reactivity of flavanols with metal ions and molecular oxygen results in flavanol polymers.
Astringency, on the other hand, is much easier to explain. It is described as a mouthfeel characterized by a drying, puckering, or rough sensation across the oral cavity, especially on the tongue [5]. It occurs when large molecular weight polyphenols react with proline-rich proteins found in saliva. Upon their interaction, they precipitate onto the surface of the mouth, which leads to the feeling of a coating dryness [6].
[7][8], and flavor is comprised of four different components: odor, aroma, taste, and mouthfeel. By definition, the odor is the perception of volatiles by the olfactory mucous membrane in the nasal cavity, after sniffing through the nose and entering the nasal passage. Aroma is also connected with volatile compounds, which volatilize by body heat after putting the beverage in the mouth. Soluble substances in the mouth recognized by the receptors located on the surface of the tongue form the reception of taste [8][9][10].
Mouthfeel is a sensation of the haptic perception of the food product on the surface of the oral cavity (the sparkling of carbon dioxide, the oiliness of fats, and astringency) [9][11][12][13]. All of these components intertwine and sum up the perceived flavor as a result of complex reactions between the senses, as can be seen from Figure 1. A good example is when beers with higher CO2content taste more sour but less astringent.
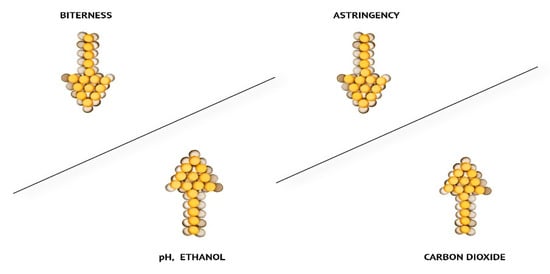
Figure 1. Changes in taste perception (higher levels of CO2 in beer decrease astringency; higher C2H5OH concentration and higher beer pH increase the bitterness perception) [12].
2. Raw Materials and Polyphenols
Even though beer is produced from four basic components, water, malt, hops, and yeast, phenolic compounds in beer commonly originate from hops (30%), malt (70%) [14], or can form during chemical transformations in the brewing process.
Phenolic acids are not as responsible for beer flavor, as they influence flavor precursors. Bottom-fermented light beers do not withstand heavy phenolic notes, which are, in this case, noted as off-flavor. Top-fermented beers, such as German Weizen or Belgian white beers, are designated by the volatile monophenols [15][16][17]. However, raw material also represents an important parameter [18].
According to [19][20], phenolic compounds quantified in beer are somewhat higher than in white wine and lower than in red wine. This inconsistency can be attributed to the varying quality of raw materials, yeast strains, and brewing process parameters [18].
Common phenolic compounds that can be found in barley (in free, esterified, or bound form) are benzoic and cinnamic acid derivatives, proanthocyanidins, quinines, flavonols, chalcones, flavones, flavanones, and amino phenolic compounds [21][22][23][24][25][26][27][28][29][30]. Ferulic and p-coumaric acid are the low-molecular weight (LMW) phenolic acids in barley grains. They can be detected in the husk, pericarp, testa, and aleurone, and even in the endosperm matrix. Phenolic acids such as vanillic, sinapinic, and p-hydroxybenzoic acids [23][28][31][32][33][34]; flavan-3-ols from monomers ((+)-catechin and (−)-epicatechin), dimers (prodelphinidin B3 and procyanidin B3), and trimers (procyanidin C2); and higher-molecular weight flavonoid-derived tannins are common in barley as well [23][35][36].
Polyphenols are secondary plant metabolites and play an important role as protective agents. There are many classes of polyphenols, but only prenylflavonoids are characteristically present in the hop plant. Bitter acids (multifidol glucosides) are also indigenous to hop plants [37]. The most common classes are presented in the following sections adapted from an extensive review by Knez Hrnčič et al.
Prenylflavonoids represent a class of flavonoids with at least one prenyl or geranyl substituent in the ring [38]. It is presumed that the desmethylxanthohumol represents a precursor for the majority of flavonoids in hops [39]. It has been studied as an anticancer agent, but poses many other positive properties against pathogenic fungi, malaria, and HIV-1 viruses [40]. Another chemical compound with anticarcinogenic properties belonging to this group is 8-prenylnaringenin [41].
)-catechin is the third most abundant compound in hop cones possessing antioxidative and vasodilative features [40]. Flavanol(−)-epicatechin and (+)-gallocatechin can be found in hops as well [42]. Flavanols catechin and epicatechin show antioxidative and anti-inflammatory properties [43]. A mixture of hop proanthocyanidins shows more pronounced antioxidative properties than individual flavanols and proanthocyanidins [40].
Quercetin and kaempferol are the most mentioned antioxidant flavanols that can be found in fruits and vegetables, but also in hops [40][42]. Plants usually contain flavonols in the form of glycosides [44], which, in the case of quercetin, presents its bioavailable form, and the least bioavailable form of quercetin is rutin, the most common form of quercetin in hops It is widely recognized for its anti-aging properties [45]. Both, quercetin and kaempferol can inhibit the growth of various cancer cells [46].
1-(2-methyl propanoyl)phloroglucinol-glucopyranoside, multifidol glucosides, 1-(3-methyl butyryl) phloroglucinol, and 5-(2 Methylpropanoyl) phloroglucinol are constituents of hops. Authors [47] have investigated the human recognition threshold concentrations and the lowest recognitions levels for 11 bitter tastants from the hop hard resin fraction, and the lowest concentration was determined for co-multifidol glucopyranoside (5 μmol/L) showing that minor hop compounds may act as significant taste-carriers [47].
Ferulic acid from hops is designated as a highly antioxidative polyphenolic compound that prevents lipid peroxidation, apoptotic cell death of healthy cells, and acts as a free radical scavenger [42][48]. It is important for the brewing industry because it retards the degradation of iso-α-acids and actively prevents beer spoilage [49]. Ferulic acid has many properties that enhance the toxicity of certain chemicals, carcinogenic agents, and ionizing radiation. Resveratrol is reportedly an anti-inflammatory and anticancer agent, and also acts preventively on cardiovascular diseases development [50][51].
The malting process itself induces modifications in the composition of barley grain including the degradation of endogenous phenolic compounds [23][29][32][52][53][54][55]. According to several authors, the concentration of phenolic compounds in malt can be higher than in barley. However, the main groups remain the same, which indicates that the extraction of flavonoids and phenolic acids in malt is more pronounced [28][29][32]. Behavior of polyphenols after malting and brewing is described in the sections below.
Hops are used as a spice in the brewing process and only 200–600 g/hL of beer. Many hop varieties contribute to beer with two main characteristics: bitterness and aromas. Among many different compounds, resins and essential oils are the most important compounds for the brewing industry, but phenolic compounds, important for the flavor of the finished beer, are significant as well [56]. The α-acids get extracted during wort boiling when they undergo oxidative isomerization to iso-α-acids (iso-humulones), also recognized as bitter compounds of beer (Figure 2).
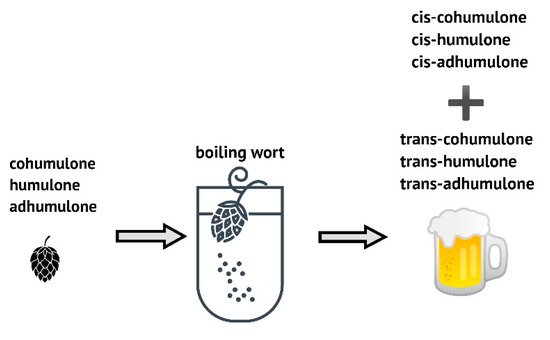
Figure 2. Scheme of α-acids isomerization to trans- and cis-forms.
Beer contains from 20–60 mg/L of iso-humulones, and oxidized β-acids sum up to the rest of the bitterness sensation. Besides resins, hops contain 0.5–3.0% of essential oils, which provide certain beer flavors. Phenolic compounds in beer are mostly tannins, flavonoids, and polyphenols. A significant portion of phenolic compounds are in the monomeric form (p-coumaric, ferulic, chlorogenic, caffeic acids, and gallic).
Humulones and linalool, the two-hop constituents, tend to prevent beer gushing [57] if added during wort boiling [58].
Polyphenolic molecules can be found in different stages during the brewing process and react with proteins: during wort boiling, they form the hot break; during cooling, they form the cold break; and during post-fermentation, they are involved in the formation of chill haze and permanent hazes and facilitate the removal of undesirable compounds with filtration. However, they tend to react with proteins in packaged beer and form undesirable haze after the expiration date [59].
Mashing-in time also plays an important factor in phenolic acid release, but viscosity and grist coarseness are important as well [60][61]. Decoction results with worts with higher total polyphenol content [62]. Several authors [63] reported that using lauter tun can result in lower phenolic compounds levels in wort after separation. Research by [64] noted a significant increase of total polyphenols during lautering, probably due to their extraction from phenol-rich spelt material.
It is predicted that during wort boiling, about 70% of polyphenols are extracted from hops [14][65], but according to their polarity and their tendency to form complexes with wort proteins, this number is variable [65]. Hot trub contains 40–70% of proteins, 7–32% of bitter substances, 20–30% of organic substances, and 5% of ash [66]. Higher oligomeric phenolic compounds are prone to form complexes with proteins, and small phenolic molecules like phenolic acids easily get adsorbed to hot trub [67]. However, several phenolic acids and catechin, except for ferulic acid (35% decrease during warm rest), were not affected [64].
Prenylflavanones showed higher levels in beer than prenylchalcones, even though raw hops contain very low amounts of prenylflavanones [68][69][70]. Xanthohumol losses commonly occur due to adsorption to hot trub, yeast, and cold trub. Xanthohumol isomerization assumedly occurs because xanthohumol binds to higher molecular weight (300 to 600 kDa) roasted substances [71][72][73][74][75], which can serve as xanthohumol carriers, and reduce its losses during fermentation, filtration, and stabilization process [75]. Lower levels of PVPP remove phenolic compounds with higher degrees of hydroxylation and oligomerization, and at higher levels, PVPP removes all polyphenols [76][77].
Storage time can significantly impact beer quality, such as colloidal stability and flavor. In a study that followed changes during a six-month storage time, the concentrations of (+)-catechin, (−)-epicatechin, proanthocyanidin B3, and prodelphinidin showed a significant reduction [78], but monomeric flavanols showed increased stability over dimers during six months of storage [78][79]. A study on the influence of acetaldehyde from beer on haze formation and stability of beer phenolics reported that beer pH is influenced by acetaldehyde, which results in a reduction of catechin content and haze formation [80]. Prenylated flavonoids exhibit stability during beer storage, resulting in almost no change in beer stored for 10 years at 20 °C in a brown glass bottle [81].
Phenolic acids show high sensory thresholds. For example, cinnamic acid has a threshold of 67 to 139 mg/L and benzoic acid derivatives are 206 to 315 mg/L for [82]; remaining decarboxylation products exhibit significant flavor activity [16]. Some volatile monophenols bring about the spicy, clove-like, sweet, and vanilla-like notes to a beer, but at higher concentrations, they become unpleasant [16]. However, yeast strain and fermentation conditions influence and amplify the phenolic flavor of wheat beers more than temperature conditions [15][83].
3. Bitterness
Bitterness is an important property of beers and according to several authors, about 80% of bitterness originates from hops during boiling [84][85][86]. The ratio of trans/cis stereoisomers for standard beers is close to 3:7 beers [87], or 68:32 in favor of the cis-compounds. However, bitter taste in beer is modified with residual sugars and results in pleasant bitterness for the consumer [88]. Isomerized α-acids have tensioactive properties, which stabilize the beer foam, and they act inhibitory to Gram-positive bacteria, while lactic acid bacteria in beer exhibit resistance to iso-α-acids.
Bittering procedures have evolved and have transferred the use of hops into almost all brewing stages, such as post-fermentation bittering, or dry hopping. The availability of the hop extends to different forms (cones, pellets, plugs) that can be added at different stages of the brewing process. Dry-hopping is a method of soaking hops in beer during fermentation or conditioning in order to add different aromas and flavors to the finished beer. Moreover, they are recognized as important acceptance factors in different beverages, including beer [89].
4. Astringency
As described in the introduction, astringency is a complex sensory property, characterized by drying, roughing, and puckering of the skin or mucosal surface in the mouth. Immediate perception is not always possible; however, it is usually recognized in the mouth after swallowing the content [90]. According to Siebert and Chassy [91], several compounds provide this sensation: salts of multivalent metallic cations (alum), dehydrating agents (ethanol or acetone), mineral and organic acids, and plant tannins (polyphenols). The reaction between saliva proteins that lubricate the mouth and polyphenols derived from foods results in an astringent sensation.
Certain phenolic compounds (ferulic acid, p-coumaric acid, and protocatechuic acid) reportedly aid astringency [92]. On the other hand, flavanols, catechin, and epicatechin add bitterness [2][89].
Research conducted by [93] investigated the addition of purified polyphenols and oxidized counterparts to water and beer. The conducted sensory analysis confirmed that the addition of polyphenols to beer induced harsh bitterness and increased astringency. A similar report was submitted by [5]. Sensory panelists graded beer with iso-α acids and 100 or 200 mg/L of polyphenols from spent hop as more bitter.
Removal of acrospires from malt by polishing reduced the astringent components, resulting in smooth-tasting beer [94][95][96]. If subcritical water is applied to malt, from which beer was produced, such beer had reduced astringent components and aftertaste.
Lower pH values result in lesser polyphenols (tannins) that get extracted to beer, and this beer is then less astringent. Mg2+in concentration over 15 mg/L can affect a sour or bitter astringency found in beer [56].
References
- Mastanjević, K.; Krstanović, V.; Lukinac, J.; Jukić, M.; Lučan, M.; Mastanjević, K. Craft brewing—Is it really about the sensory revolution? Kvasny Prumysl 2019, 65, 13–16.
- Peleg, H.; Gacon, K.; Schlich, P.; Noble, A. Bitterness and astringency of flavan-3-ol monomers, dimers and trimers. J. Sci. Food Agric. 1999, 79, 1123–1128.
- Drewnowski, A. The science and complexity of bitter taste. Nutr. Rev. 2001, 59, 163–169.
- Walters, E.; Roy, G. Taste Interactions of Sweet and Bitter Compounds. In Flavor-Food Interactions; American Chemical Society: Washington, DC, USA, 1996; pp. 130–142.
- McLaughlin, I.R.; Lederer, C.; Shellhammer, T.H. Bitterness-Modifying Properties of Hop Polyphenols Extracted from Spent Hop Material. J. Am. Soc. Brew. Chem. 2008, 66, 174–183.
- Luck, G.; Liao, H.; Murray, N.; Grimmer, H.; Warminski, E.; Williamson, M.; Lilley, T.; Haslam, E. Polyphenols, astringency and proline—Rich proteins. Phytochemistry 1994, 37, 357–371.
- Amerine, M.A.; Pangborn, R.M.; Roessler, E.B. Principles of Sensory Evaluation of Food; Academic Press: New York, NY, USA, 1965.
- Meilgaard, M.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 4th ed.; CRC Press: Boca Raton, FL, USA, 2007.
- Schmelzle, A. The beer aroma wheel. Updating beer flavor terminology according to sensory standards. Brew. Sci. 2009, 62, 26–32.
- Saison, D.; De Schutter, D.P.; Uyttenhove, B.; Delvaux, F.; Delvaux, F.R. Contribution of staling compounds to the aged flavor of lager beer by studying their flavour thresholds. Food Chem. 2009, 114, 1206–1215.
- Langstaff, S.A.; Lewis, M.J. The mouthfeel of beer—A review. J. Inst. Brew. 1993, 99, 31–37.
- Meilgaard, M.C.; Dalgliesh, C.E.; Clapperton, J.F. Beer flavor terminology. J. Am. Soc. Brew. Chem. 1979, 37, 47–52.
- Langstaff, S.A.; Guinard, J.X.; Lewis, M.J. Sensory evaluation of the mouthfeel of beer. J. Am. Soc. Brew. Chem. 1991, 49, 54–59.
- Jerkovic, V.; Callemien, D.; Collin, S. Determination of stilbenes in hop pellets from different cultivars. J. Agric. Food Chem. 2005, 53, 4202–4206.
- Vanbeneden, N.; Gils, F.; Delvaux, F.; Delvaux, F.R. Formation of 4-vinyl and 4-ethyl derivatives from hydroxycinnamic acids: Occurrence of volatile phenolic flavour compounds in beer and distribution of pad1-activity among brewing yeasts. Food Chem. 2008, 107, 221–230.
- Sterckx, F.L.; Missiaen, J.; Saison, D.; Delvaux, F.R. Contribution of monophenols to beer flavour based on flavour thresholds, interactions and recombination experiments. Food Chem. 2011, 126, 1679–1685.
- Langos, D.; Granvogl, M.; Schieberle, P. Characterization of the key aroma compounds in two Bavarian wheat beers by means of the sensomics approach. J. Agric. Food Chem. 2013, 61, 11303–11311.
- Wannenmacher, J.; Gastl, M.; Becker, T. Phenolic Substances in Beer: Structural Diversity, Reactive Potential and Relevance for Brewing Process and Beer Quality. Compr. Rev. Food Sci. Food Saf. 2018, 17, 953–988.
- Neveu, V.; Perez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024.
- Bamforth, C.W. Beer: Tapping into the Art and Science of Brewing; Insight Books Plenum Publishing Corporation: New York, NY, USA; London, UK, 2003; pp. 60–65.
- Bonoli, M.; Marconi, E.; Caboni, M.F. Free and bound phenolic compounds in barley (Hordeum vulgare L.) flours: Evaluation of the extraction capability of different solvent mixtures and pressurized liquid methods by micellar electrokinetic chromatography and spectrophotometry. J. Chromatogr. A 2004, 1057, 1–12.
- Bonoli, M.; Verardo, V.; Marconi, E.; Caboni, M.F. Antioxidant phenols in barley (Hordeum vulgare L.) flour: Comparative spectrophotometric study among extraction methods of free and bound phenolic compounds. J. Agric. Food Chem. 2004, 52, 5195–5200.
- Dvořáková, M.; Guido, L.F.; Dostálek, P.; Skulilová, Z.; Moreira, M.M.; Barros, A.A. Antioxidant properties of free, soluble ester and insoluble-bound phenolic compounds in different barley varieties and corresponding malts. J. Inst. Brew. 2008, 114, 27–33.
- Nardini, M.; Cirillo, E.; Natella, F.; Mencarelli, D.; Comisso, A.; Scaccini, C. Detection of bound phenolic acids: Prevention by ascorbic acid and ethylenediaminetetraacetic acid of degradation of phenolic acids during alkaline hydrolysis. Food Chem. 2002, 79, 119–124.
- Nardini, M.; Ghiselli, A. Determination of free and bound phenolic acids in beer. Food Chem. 2004, 84, 137–143.
- Nardini, M.; Natella, F.; Scaccini, C.; Ghiselli, A. Phenolic acids from beer are absorbed and extensively metabolized in humans. J. Nutr. Biochem. 2006, 17, 14–22.
- Gerhäuser, C. Beer constituents as potential cancer chemopreventive agents. Eur. J. Cancer 2005, 41, 1941–1954.
- Maillard, M.N.; Soum, M.H.; Boivin, P.; Berset, C. Antioxidant activity of barley and malt: Relationship with phenolic content. LWT—Food Sci. Technol. 1996, 29, 238–244.
- Lu, J.; Zhao, H.; Chen, J.; Fan, W.; Dong, J.; Kong, W.; Sun, J.; Cao, Y.; Cai, G. Evolution of phenolic compounds and antioxidant activity during malting. J. Agric. Food Chem. 2007, 55, 10994–11001.
- Carvalho, D.O.; Curto, A.F.; Guido, L.F. Determination of Phenolic Content in Different Barley Varieties and Corresponding Malts by Liquid Chromatography-diode Array Detection-Electrospray Ionization Tandem Mass Spectrometry. Antioxidants 2015, 4, 563–576.
- Whittle, N.; Eldridge, H.; Bartley, J.; Organ, G. Identification of the polyphenols in barley and beer by HPLC/MS and HPLC/electrochemical detection. J. Inst. Brew. 1999, 105, 89–99.
- Goupy, P.; Hugues, M.; Boivin, P.; Amiot, M.J. Antioxidant composition and activity of barley (Hordeum vulgare) and malt extracts and of isolated phenolic compounds. J. Sci. Food Agric. 1999, 79, 1625–1634.
- Liu, Q.; Yao, H. Antioxidant activities of barley seeds extracts. Food Chem. 2007, 102, 732–737.
- Kim, M.-J.; Hyun, J.-N.; Kim, J.-A.; Park, J.-C.; Kim, M.-Y.; Kim, J.-G.; Lee, S.-J.; Chun, S.-C.; Chung, I.-M. Relationship between phenolic compounds, anthocyanins content and antioxidant activity in colored barley germplasm. J. Agric. Food Chem. 2007, 55, 4802–4809.
- Holtekjølen, A.K.; Kinitz, C.; Knutsen, S.H. Flavanol and bound phenolic acid contents in different barley varieties. J. Agric. Food Chem. 2006, 54, 2253–2260.
- Dvorakova, M.; Moreira, M.M.; Dostalek, P.; Skulilova, Z.; Guido, L.F.; Barros, A.A. Characterization of monomeric and oligomeric flavan-3-ols from barley and malt by liquid chromatography-ultraviolet detection-electrospray ionization mass spectrometry. J. Chromatogr. A 2008, 1189, 398–405.
- Knez Hrnčič, M.; Španinger, E.; Košir, I.J.; Knez, Ž.; Bren, U. Hop Compounds: Extraction Techniques, Chemical Analyses, Antioxidative, Antimicrobial, and Anticarcinogenic Effects. Nutrients 2019, 11, 257.
- Plazar, J. Mechanism of Antigenotoxic Activity of Xanthohumol and Related Prenylflavonoids from Hops (Humulus lupulus L.). Ph.D. Thesis, Nacionalni Inštitut za Biologijo, Ljubljana, Slovenia, 2007.
- Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R. The pharmacognosy of Humulus lupulus L. (hops) with an emphasis on estrogenic properties. Phytomedicine 2006, 13, 119–131.
- Biendl, M.; Pinzl, C. Hops and Health Uses-Effects-History, 2nd ed.; German Hop Museum Wonlzach: Wonlzach, Germany, 2013.
- Brunelli, E.; Minassi, A.; Appendino, G.; Moro, L. 8-Prenylnaringenin, inhibits estrogen receptor-alfa mediated cell growth and induces apoptosis in MCF-7 breast cancer cells. J. Steroid Biochem. Mol. Biol. 2007, 107, 140–148.
- Karabín, M.; Hudcová, T.; Jelínek, L.; Dostálek, P. Biologically Active Compounds from Hops and Prospects for Their Use. Compr. Rev. Food Sci. Food Saf. 2016, 15, 542–567.
- Fresco, P.; Borges, F.; Diniz, C.; Marques, M.P.M. New Insights on the Anticancer Properties of Dietary Polyphenols. Med. Res. Rev. 2006, 26, 747–766.
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747.
- Corrêa, R.C.G.; Peralta, R.M.; Haminiuk, C.W.I.; Maciel, G.M.; Bracht, A.; Ferreira, I.C.F.R. New phytochemicals as potential human anti-aging compounds: Reality, promise, and challenges. Crit. Rev. Food Sci. Nutr. 2018, 58, 942–957.
- Dai, J.; Mumper, J.R. Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties. Molecules 2010, 15, 7313–7352.
- Dresel, M.; Dunkel, A.; Hofmann, T. Sensomics Analysis of Key Bitter Compounds in the Hard Resin of Hops (Humulus lupulus L.) and Their Contribution to the Bitter Profile of Pilsner-Type Beer. J. Agric. Food Chem. 2015, 63, 3402–3418.
- Ghosh, S.; Basak, P.; Duttam, S.; Chowdhury, S.; Sil, P.C. New insights into the ameliorative effects of ferulic acid in pathophysiological conditions. Food Chem. Toxicol. 2017, 103, 41–55.
- Spreng, S.; Hofmann, T. Activity-Guided Identification of in Vitro Antioxidants in Beer. J. Agric. Food Chem. 2018, 66, 720–731.
- Park, E.J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113.
- Brglez Mojzer, E.; Knez-Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901.
- Samaras, T.S.; Camburn, P.A.; Chandra, S.X.; Gordon, M.H.; Ames, J.M. Antioxidant properties of kilned and roasted malts. J. Agric. Food Chem. 2005, 53, 8068–8074.
- Leitao, C.; Marchioni, E.; Bergaentzlé, M.; Zhao, M.; Didierjean, L.; Miesch, L.; Holder, E.; Miesch, M.; Ennahar, S. Fate of polyphenols and antioxidant activity of barley throughout malting and brewing. J. Cereal Sci. 2012, 55, 318–322.
- Inns, E.L.; Buggey, L.A.; Booer, C.; Nursten, H.E.; Ames, J.M. Effect of modification of the kilning regimen on levels of free ferulic acid and antioxidant activity in malt. J. Agric. Food Chem. 2011, 59, 9335–9343.
- Inns, E.L.; Buggey, L.A.; Booer, C.; Nursten, H.E.; Ames, J.M. Effect of heat treatment on the antioxidant activity, color, and free phenolic acid profile of malt. J. Agric. Food Chem. 2007, 55, 6539–6546.
- Buiatti, S. Beer composition: An overview. In Beer in Health and Disease Prevention; Academic Press: London, UK, 2009; pp. 213–225.
- Hanke, S.; Kern, M.; Herrmann, M.; Back, W.; Becker, T.; Krottenthaler, M. Suppression of gushing by hop constituents. Mon. für Brauwiss. 2009, 62, 181–186.
- Srečec, S.; Zechner-Krpan, V.; Petravić-Tominac, V.; Košir, I.J.; Čerenak, A. Importance of Medical Effects of Xanthohumol, Hop (Humulus lupulus L.) Bioflavonoid in Restructuring of World Hop Industry. Agric. Conspec. Sci. 2012, 77, 61–67.
- The Role of Polyphenols in Beer Haze Formation. Available online: (accessed on 1 May 2021).
- Vanbeneden, N.; Van Roey, T.; Willems, F.; Delvaux, F.; Delvaux, F.R. Release of phenolic flavour precursors during wort production: Influence of process parameters and grist composition on ferulic acid release during brewing. Food Chem. 2008, 111, 83–91.
- Schwarz, K.J.; Boitz, L.I.; Methner, F.-J. Release of phenolic acids and amino acids during mashing dependent on temperature, pH, time and raw materials. J. Am. Soc. Brew. Chem. 2012, 70, 290–295.
- Jurková, M.; Horák, T.; Hašková, D.; Čulík, J.; Čejka, P.; Kellner, V. Control of antioxidant beer activity by the mashing process. J. Inst. Brew. 2012, 118, 230–235.
- Fumi, M.D.; Galli, R.; Lambri, M.; Donadini, G.; De Faveri, D.M. Effect of full-scale brewing process on polyphenols in italian all-malt and maize adjunct lager beers. J. Food Comp. Anal. 2011, 24, 568–573.
- Pascoe, H.M.; Ames, J.M.; Sachin, C. Critical stages of the brewing process for changes in antioxidant activity and levels of phenolic compounds in ale. J. Am. Soc. Brew. Chem. 2003, 61, 203–209.
- Forster, A.; Gahr, A. On the fate of certain hop substances during dry hopping. Brew. Sci. 2013, 66, 93–103.
- Kühbeck, F.; Schütz, M.; Thiele, F.; Krottenthaler, M.; Back, W. Influence of lauter turbidity and hot trub on wort composition, fermentation, and beer quality. J. Am. Soc. Brew. Chem. 2006, 64, 16–28.
- Siebert, K.J.; Troukhanova, N.V.; Lynn, P.Y. Nature of polyphenol-protein interactions. J. Agric. Food Chem. 1996, 44, 80–85.
- Stevens, J.F.; Page, J.E. Xanthohumol and related prenylflavonoids from hops and beer: To your good health! Phytochemistry 2004, 65, 1317–1330.
- Stevens, J.F.; Taylor, A.W.; Clawson, J.E.; Deinzer, M.L. Fate of xanthohumol and related prenylflavonoids from hops to beer. J. Agric. Food Chem. 1999, 47, 2421–2428.
- Venturelli, S.; Burkard, M.; Biendl, M.; Lauer, U.M.; Frank, J.; Busch, C. Prenylated chalcones and flavonoids for the prevention and treatment of cancer. Nutrition 2016, 32, 1171–1178.
- Magalháes, P.J.; Dostálek, P.; Cruz, J.M.; Guido, L.F.; Barros, A.A. The impact of a xanthohumol-enriched hop product on the behavior of xanthohumol and isoxanthohumol in pale and dark beers: A pilot scale approach. J. Inst. Brew. 2008, 114, 246–256.
- Magalhães, P.J.; Almeida, S.M.; Carvalho, A.M.; Goncalves, L.M.; Pacheco, J.G.; Cruz, J.M.; Barros, A.A. Influence of malt on the xanthohumol and isoxanthohumol behavior in pale and dark beers: A micro-scale approach. Food Res. Int. 2011, 44, 351–359.
- Wunderlich, S.; Zürcher, A.; Back, W. Enrichment of xanthohumol in the brewing process. Mol. Nutr. Food Res. 2005, 49, 874–881.
- Wunderlich, S.; Zürcher, A.; Back, W. Xanthohumol in brewing—Impact of malt, xanthohumol dosage, wort and storage temperature. Mon. Brauwiss 2012, 65, 7–15.
- Wunderlich, S.; Wurzbacher, M.; Back, W. Roasting of malt and xanthohumol enrichment in beer. Eur. Food Res. Technol. 2013, 237, 137–148.
- McMurrough, I.; Madigan, D.; Smyth, M.R. Adsorption by polyvinylpyrrolidone of catechins and proanthocyanidins from beer. J. Agric. Food Chem. 1995, 43, 2687–2691.
- Gramshaw, J.W. Phenolic constituents of beer and brewing materials. II. The role of polyphenols in the formation of non-biological haze. J. Inst. Brew. 1967, 73, 455–472.
- Moll, M.; Fonknechten, G.; Carnielo, M.; Flayeux, R. Changes in polyphenols from raw materials to finished beer. MBAA Tech. Q. 1984, 21, 79–87.
- McMurrough, I.; Madigan, D.; Kelly, R.J. The role of flavonoid polyphenols in beer stability. J. Am. Soc. Brew. Chem. 1996, 54, 141–148.
- Delcour, J.A.; Dondeyne, P. The reactions between polyphenols and aldehydes and the influence of acetaldehyde on haze formation in beer. J. Inst. Brew. 1982, 88, 234–243.
- Intelmann, D.; Haseleu, G.; Dunkel, A.; Lagemann, A.; Stephan, A.; Hofmann, T. Comprehensive sensomics analysis of hop-derived bitter compounds during storage of beer. J. Agric. Food Chem. 2011, 59, 1939–1953.
- Hufnagel, J.C.; Hofmann, T. Orosensory-directed identification of astringent mouthfeel and bitter-tasting compounds in red wine J. Agric. Food Chem. 2008, 56, 1376–1386.
- Coghe, S.; Benoot, K.; Delvaux, F.; Vanderhaegen, B.; Delvaux, F.R. Ferulic acid release and 4-vinylguaiacol formation during brewing and fermentation: Indications for feruloyl esterase activity in Saccharomyces cerevisiae. J. Agric. Food Chem. 2004, 52, 602–608.
- Lesschaeve, I.; Noble, A.C. Polyphenols: Factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 2005, 81, 330S–335S.
- Caballero, I.; Blanco, C.A.; Porras, M. Iso-a-acids, bitterness and loss of beer quality during storage. Trends Food Sci. Tech. 2012, 26, 21–30.
- Arrieta, Á.A.; Rodríguez-Méndez, M.L.; De Saja, J.A.; Blanco, C.A.; Nimubona, D. Prediction of bitterness and alcoholic strength in beer using an electronic tongue. Food Chem. 2010, 123, 642–646.
- Schönberger, C.; Kostelecky, T. 125th anniversary review: The role of hops in brewing. J. Inst. Brew. 2011, 117, 259–267.
- De Keukeleire, D. Fundamentals of beer and hop chemistry. Quím. Nova 2000, 23, 108–112.
- Drewnowski, A.; Gomez-Carneros, C. Bitter taste, phytonutrients, and the consumer: A review. Am. J. Clin. Nutr, 2000, 72, 1424–1435.
- Guinard, J.-X.; Pangborn, R.-M.; Lewis, M.J. Preliminary studies on acidity-astringency interactions in model solutions and solutions. J. Sci. Food Agric. 1986, 37, 811–817.
- Siebert, K.J.; Chassy, A.W. An alternate mechanism for the astringent sensation of acids. Food Qual. Prefer. 2003, 15, 13–18.
- Callemien, D.; Collin, S. Structure, organoleptic properties, quantification methods, and stability of phenolic compounds in beer—A review. Food Rev. Int. 2009, 26, 1–84.
- Dadic, M.; Belleau, G. Polyphenols and beer flavour. Proc. Am. Soc. Brew. Chem. 1973, 107–114.
- Kageyama, N.; Tada, N.; Inui, T.; Komura, H.; Nakahara, K.; Oka, K.; Isoe, A. Studies on astringent substances in malt. Proc. Congr. Eur. Brew. Conv. 2007, 31, 185–196.
- Tada, N.; Inui, T.; Kageyama, N.; Takaoka, S.; Kawasaki, Y. The influence of malt acrospires on beer taste and foam quality. Tech. Q. Master Brew. Assoc. Am. 2004, 41, 305–309.
- Inui, T.; Tada, N.; Kageyama, N.; Takaoka, S.; Kawasaki, Y. The breakthrough technology in brewing: The capture and retention of fresh beer flavor. Brauwelt 2004, 144, 1488–1498.




