
Video Upload Options
Millions of years’ evolution has imparted life on earth with excellent environment adaptability. Of particular interest to scientists are some plants capable of macroscopically and reversibly altering their morphological and mechanical properties in response to external stimuli from the surrounding environment. These intriguing natural phenomena and underlying actuation mechanisms have provided important design guidance and principles for man-made soft robotic systems. Constructing bio-inspired soft robotic systems with effective actuation requires the efficient supply of mechanical energy generated from external inputs, such as temperature, light, and electricity. By combining bio-inspired designs with stimuli-responsive materials, various intelligent soft robotic systems that demonstrate promising and exciting results have been developed. As one of the building materials for soft robotics, hydrogels are gaining increasing attention owing to their advantageous properties, such as ultra-tunable modulus, high compliance, varying stimuli-responsiveness, good biocompatibility, and high transparency. In this review article, we summarize the recent progress on plant-inspired soft robotics assembled by stimuli-responsive hydrogels with a particular focus on their actuation mechanisms, fabrication, and application. Meanwhile, some critical challenges and problems associated with current hydrogel-based soft robotics are briefly introduced, and possible solutions are proposed. We expect that this review would provide elementary tutorial guidelines to audiences who are interested in the study on nature-inspired soft robotics, especially hydrogel-based intelligent soft robotic systems.
1. Introduction
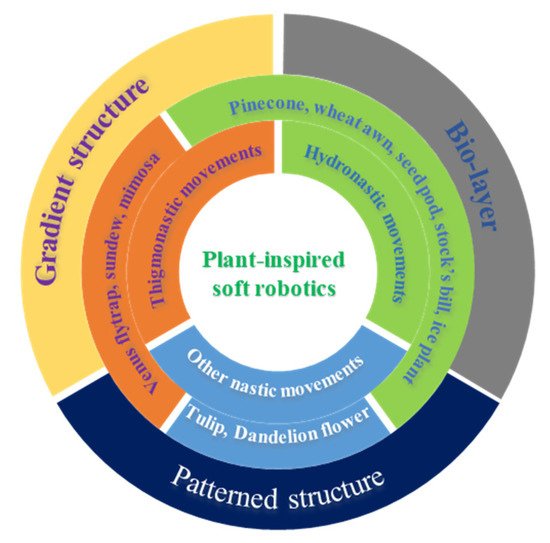
2. Nastic Movements of Plants
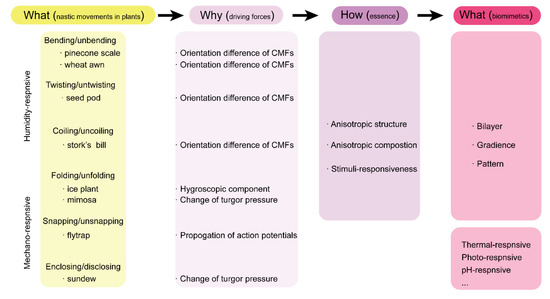
2.1. Hydronastic Movements and Actuation Mechanisms
2.1.1. Pinecone
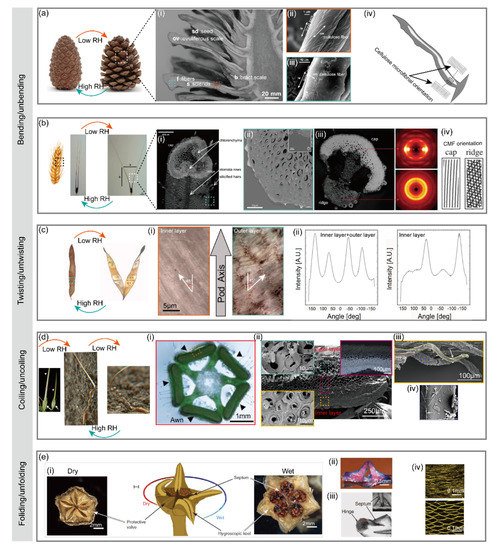
2.1.2. Wheat Awn
2.1.3. Seed Pod
2.1.4. Stork’s Bill
2.1.5. Ice Plant
2.2. Thigmonastic Movements and Actuation Mechanisms
2.2.1. Venus Flytrap

2.2.2. Sundew
2.2.3. Mimosa
3. Plant-Inspired, Hydrogel-Based Soft Robotics
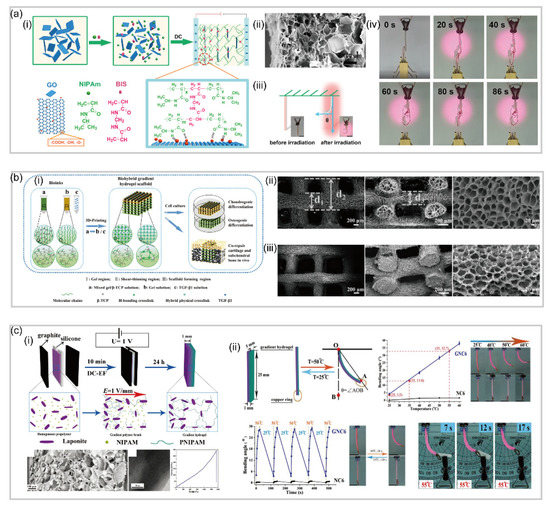
3.1. Bilayer Hydrogel-Based Soft Robotics
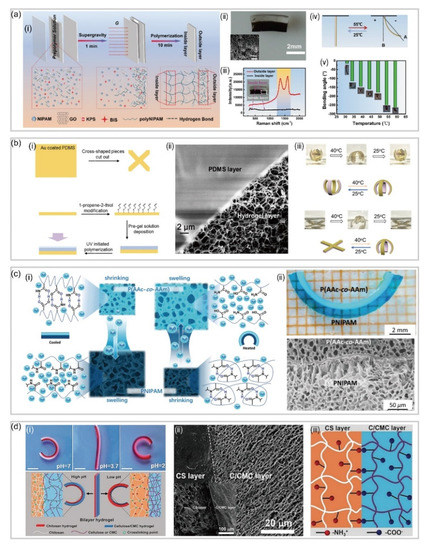
3.2. Gradient Hydrogel-Based Soft Robotics
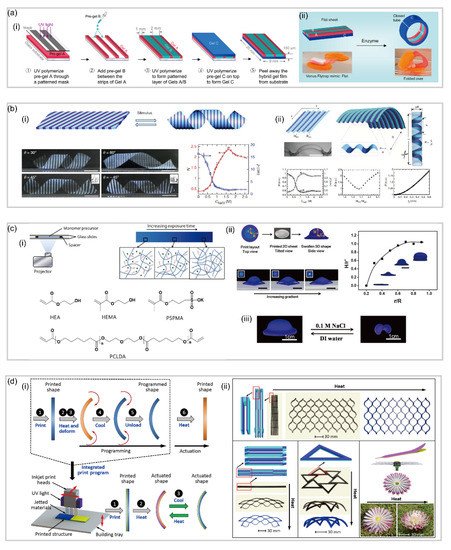
3.3. Patterned Hydrogel-Based Soft Robotics
References
- Ospovat, D. The Development of Darwin’s Theory: Natural History, Natural Theology, and Natural Selection, 1838–1859; Cambridge University Press: Cambridge, UK, 1995.
- Somveille, M.; Rodrigues, A.S.; Manica, A. Why do birds migrate? A macroecological perspective. Glob. Ecol. Biogeogr. 2015, 24, 664–674.
- Hansen, L.P.; Jonsson, N.; Jonsson, B. Oceanic migration in homing Atlantic salmon. Anim. Behav. 1993, 45, 927–941.
- Skotheim, J.M.; Mahadevan, L. Physical limits and design principles for plant and fungal movements. Science 2005, 308, 1308–1310.
- Koller, D.; Van Volkenburgh, E. The Restless Plant; Harvard University Press: Cambridge, MA, USA, 2011.
- Forterre, Y. Slow, fast and furious: Understanding the physics of plant movements. J. Exp. Bot. 2013, 64, 4745–4760.
- Brauner, L. Tropisms and nastic movements. Annu. Rev. Plant Biol. 1954, 5, 163–182.
- Kiss, J.Z. Up, down, and all around: How plants sense and respond to environmental stimuli. Proc. Natl. Acad. Sci. USA 2006, 103, 829–830.
- Dawson, C.; Vincent, J.F.; Rocca, A.-M. How pine cones open. Nature 1997, 390, 668.
- Elbaum, R.; Zaltzman, L.; Burgert, I.; Fratzl, P. The role of wheat awns in the seed dispersal unit. Science 2007, 316, 884–886.
- Harrington, M.J.; Razghandi, K.; Ditsch, F.; Guiducci, L.; Rueggeberg, M.; Dunlop, J.W.C.; Fratzl, P.; Neinhuis, C.; Burgert, I. Origami-like unfolding of hydro-actuated ice plant seed capsules. Nat. Commun. 2011, 2, 337.
- Forterre, Y.; Skotheim, J.M.; Dumais, J.; Mahadevan, L. How the Venus flytrap snaps. Nature 2005, 433, 421–425.
- Nakamura, Y.; Reichelt, M.; Mayer, V.E.; Mithöfer, A. Jasmonates trigger prey-induced formation of ‘outer stomach’in carnivorous sundew plants. Proc. Royal Soc. B 2013, 280, 20130228.
- Ahmad, H.; Sehgal, S.; Mishra, A.; Gupta, R. Mimosa pudica L. (Laajvanti): An overview. Pharmacogn. Rev. 2012, 6, 115.
- Van Doorn, W.G.; Van Meeteren, U. Flower opening and closure: A review. J. Exp. Bot. 2003, 54, 1801–1812.
- Kim, S.; Laschi, C.; Trimmer, B. Soft robotics: A bioinspired evolution in robotics. Trends Biotechnol. 2013, 31, 287–294.
- Fratzl, P.; Barth, F.G. Biomaterial systems for mechanosensing and actuation. Nature 2009, 462, 442–448.
- Coyle, S.; Majidi, C.; LeDuc, P.; Hsia, K.J. Bio-inspired soft robotics: Material selection, actuation, and design. Extreme Mech. Lett. 2018, 22, 51–59.
- Lee, Y.; Song, W.J.; Sun, J.Y. Hydrogel soft robotics. Mater. Today Phys. 2020, 15, 100258.
- Gladman, A.S.; Matsumoto, E.A.; Nuzzo, R.G.; Mahadevan, L.; Lewis, J.A. Biomimetic 4D printing. Nat. Mater. 2016, 15, 413–418.
- Stano, G.; Percoco, G. Additive manufacturing aimed to soft robots fabrication: A review. Extreme Mech. Lett. 2021, 42, 101079.
- Ionov, L. Biomimetic hydrogel-based actuating systems. Adv. Funct. Mater. 2013, 23, 4555–4570.
- Van der Pijl, L. Principles of Dispersal; Springer: Berlin, Germany, 1982.
- Cousens, R.; Dytham, C.; Law, R. Dispersal in Plants: A Population Perspective; Oxford University Press: Oxford, UK, 2008.
- Burgert, I.; Fratzl, P. Actuation systems in plants as prototypes for bioinspired devices. Philos. Trans. R. Soc. A 2009, 367, 1541–1557.
- Harlow, W.M.; Côté, W.A.; Day, A.C. The opening mechanism of pine cone scales. J. For. Res. 1964, 62, 538–540.
- Erb, R.M.; Sander, J.S.; Grisch, R.; Studart, A.R. Self-shaping composites with programmable bioinspired microstructures. Nat. Commun. 2013, 4, 1–8.
- Reyssat, E.; Mahadevan, L. Hygromorphs: From pine cones to biomimetic bilayers. J. R. Soc. Interface 2009, 6, 951–957.
- Emons, A.M.C.; Mulder, B.M. How the deposition of cellulose microfibrils builds cell wall architecture. Trends Plant Sci. 2000, 5, 35–40.
- Burgert, I.; Fratzl, P. Plants control the properties and actuation of their organs through the orientation of cellulose fibrils in their cell walls. Integr. Comp. Biol. 2009, 49, 69–79.
- Abraham, Y.; Tamburu, C.; Klein, E.; Dunlop, J.W.; Fratzl, P.; Raviv, U.; Elbaum, R. Tilted cellulose arrangement as a novel mechanism for hygroscopic coiling in the stork’s bill awn. J. R. Soc. Interface 2012, 9, 640–647.
- Armon, S.; Efrati, E.; Kupferman, R.; Sharon, E. Geometry and mechanics in the opening of chiral seed pods. Science 2011, 333, 1726–1730.
- Forterre, Y.; Dumais, J. Generating Helices in Nature. Science 2011, 333, 1715–1716.
- Aßhoff, S.J.; Lancia, F.; Iamsaard, S.; Matt, B.; Kudernac, T.; Fletcher, S.P.; Katsonis, N. High-Power Actuation from Molecular Photoswitches in Enantiomerically Paired Soft Springs. Angew. Chem. Int. Ed. 2017, 56, 3261–3265.
- Ghafouri, R.; Bruinsma, R. Helicoid to spiral ribbon transition. Phys. Rev. Lett. 2005, 94, 138101.
- Abraham, Y.; Elbaum, R. Hygroscopic movements in Geraniaceae: The structural variations that are responsible for coiling or bending. New Phytol. 2013, 199, 584–594.
- Evangelista, D.; Hotton, S.; Dumais, J. The mechanics of explosive dispersal and self-burial in the seeds of the filaree, Erodium cicutarium (Geraniaceae). J. Exp. Biol. 2011, 214, 521–529.
- Abraham, Y.; Dong, Y.; Aharoni, A.; Elbaum, R. Mapping of cell wall aromatic moieties and their effect on hygroscopic movement in the awns of stork’s bill. Cellulose 2018, 25, 3827–3841.
- Almeida, A.P.C.; Querciagrossa, L.; Silva, P.E.S.; Gonçalves, F.; Canejo, J.P.; Almeida, P.L.; Godinho, M.H.; Zannoni, C. Reversible water driven chirality inversion in cellulose-based helices isolated from Erodium awns. Soft Matter 2019, 15, 2838–2847.
- Klak, C.; Reeves, G.; Hedderson, T. Unmatched tempo of evolution in Southern African semi-desert ice plants. Nature 2004, 427, 63–65.
- Parolin, P. Ombrohydrochory: Rain-operated seed dispersal in plants–With special regard to jet-action dispersal in Aizoaceae. Flora 2006, 201, 511–518.
- Elbaum, R.; Abraham, Y. Insights into the microstructures of hygroscopic movement in plant seed dispersal. Plant Sci. 2014, 223, 124–133.
- Hedrich, R.; Neher, E. Venus flytrap: How an excitable, carnivorous plant works. Trends Plant Sci. 2018, 23, 220–234.
- Volkov, A.G.; Vilfranc, C.L.; Murphy, V.A.; Mitchell, C.M.; Volkova, M.I.; O’Neal, L.; Markin, V.S. Electrotonic and action potentials in the Venus flytrap. J. Plant Physiol. 2013, 170, 838–846.
- Scherzer, S.; Shabala, L.; Hedrich, B.; Fromm, J.; Bauer, H.; Munz, E.; Jakob, P.; Al-Rascheid, K.A.; Kreuzer, I.; Becker, D. Insect haptoelectrical stimulation of Venus flytrap triggers exocytosis in gland cells. Proc. Natl. Acad. Sci. USA. 2017, 114, 4822–4827.
- Volkov, A.G.; Adesina, T.; Jovanov, E. Closing of Venus flytrap by electrical stimulation of motor cells. Plant Signal. Behav. 2007, 2, 139–145.
- Fabricant, A.; Iwata, G.Z.; Scherzer, S.; Bougas, L.; Rolfs, K.; Jodko-Władzińska, A.; Voigt, J.; Hedrich, R.; Budker, D. Action potentials induce biomagnetic fields in carnivorous Venus flytrap plants. Sci. Rep. 2021, 11, 1438.
- Sachse, R.; Westermeier, A.; Mylo, M.; Nadasdi, J.; Bischoff, M.; Speck, T.; Poppinga, S. Snapping mechanics of the Venus flytrap (Dionaea muscipula). Proc. Natl. Acad. Sci. USA 2020, 117, 16035–16042.
- Adlassnig, W.; Lendl, T.; Peroutka, M.; Lang, I. Deadly glue—Adhesive traps of carnivorous plants. In Biological Adhesive Systems; Springer: Vienna, Austria, 2010; pp. 15–28.
- Krausko, M.; Perutka, Z.; Šebela, M.; Šamajová, O.; Šamaj, J.; Novák, O.; Pavlovič, A. The role of electrical and jasmonate signalling in the recognition of captured prey in the carnivorous sundew plant Drosera capensis. New Phytol. 2017, 213, 1818–1835.
- Pavlovič, A.; Krausko, M.; Adamec, L. A carnivorous sundew plant prefers protein over chitin as a source of nitrogen from its traps. Plant Physiol. Biochem. 2016, 104, 11–16.
- Poppinga, S.; Hartmeyer, S.R.H.; Seidel, R.; Masselter, T.; Hartmeyer, I.; Speck, T. Catapulting tentacles in a sticky carnivorous plant. PLoS ONE 2012, 7, e45735.
- Guo, Q.; Dai, E.; Han, X.; Xie, S.; Chao, E.; Chen, Z. Fast nastic motion of plants and bioinspired structures. J. R. Soc. Interface 2015, 12, 20150598.
- Volkov, A.G.; Foster, J.C.; Baker, K.D.; Markin, V.S. Mechanical and electrical anisotropy in Mimosa pudica pulvini. Plant Signal. Behav. 2010, 5, 1211–1221.
- Scorza, L.C.; Dornelas, M.C. Plants on the move: Towards common mechanisms governing mechanically-induced plant movements. Plant Signal. Behav. 2011, 6, 1979–1986.
- Le, X.; Lu, W.; Zhang, J.; Chen, T. Recent progress in biomimetic anisotropic hydrogel actuators. Adv. Sci. 2019, 6, 1801584.
- Kharlampieva, E.; Kozlovskaya, V.; Sukhishvili, S.A. Layer-by-layer hydrogen-bonded polymer films: From fundamentals to applications. Adv. Mater. 2009, 21, 3053–3065.
- He, X.; Sun, Y.; Wu, J.; Wang, Y.; Chen, F.; Fan, P.; Zhong, M.; Xiao, S.; Zhang, D.; Yang, J. Dual-stimulus bilayer hydrogel actuators with rapid, reversible, bidirectional bending behaviors. J. Mater. Chem. C 2019, 7, 4970–4980.
- Li, X.; Cai, X.; Gao, Y.; Serpe, M.J. Reversible bidirectional bending of hydrogel-based bilayer actuators. J. Mater. Chem. B 2017, 5, 2804–2812.
- Zheng, J.; Xiao, P.; Le, X.; Lu, W.; Théato, P.; Ma, C.; Du, B.; Zhang, J.; Huang, Y.; Chen, T. Mimosa inspired bilayer hydrogel actuator functioning in multi-environments. J. Mater. Chem. C 2018, 6, 1320–1327.
- Duan, J.; Liang, X.; Zhu, K.; Guo, J.; Zhang, L. Bilayer hydrogel actuators with tight interfacial adhesion fully constructed from natural polysaccharides. Soft Matter 2017, 13, 345–354.
- Myung, D.; Waters, D.; Wiseman, M.; Duhamel, P.E.; Noolandi, J.; Ta, C.N.; Frank, C.W. Progress in the development of interpenetrating polymer network hydrogels. Polym. Adv. Technol. 2008, 19, 647–657.
- Kozhunova, E.Y.; Makhaeva, E.E.; Khokhlov, A.R. Collapse of thermosensitive polyelectrolyte semi-interpenetrating networks. Polymer 2012, 53, 2379–2384.
- Tominaga, T.; Tirumala, V.R.; Lee, S.; Lin, E.K.; Gong, J.P.; Wu, W.-l. Thermodynamic interactions in double-network hydrogels. J. Phys. Chem. B 2008, 112, 3903–3909.
- Tan, Y.; Wu, R.; Li, H.; Ren, W.; Du, J.; Xu, S.; Wang, J. Electric field-induced gradient strength in nanocomposite hydrogel through gradient crosslinking of clay. J. Mater. Chem. B 2015, 3, 4426–4430.
- Yang, Y.; Tan, Y.; Wang, X.; An, W.; Xu, S.; Liao, W.; Wang, Y. Photothermal nanocomposite hydrogel actuator with electric-field-induced gradient and oriented structure. ACS Appl. Mater. Interfaces 2018, 10, 7688–7692.
- Maeda, S.; Hara, Y.; Sakai, T.; Yoshida, R.; Hashimoto, S. Self-walking gel. Adv. Mater. 2007, 19, 3480–3484.
- Gao, F.; Xu, Z.; Liang, Q.; Liu, B.; Li, H.; Wu, Y.; Zhang, Y.; Lin, Z.; Wu, M.; Ruan, C.; et al. Direct 3D Printing of High Strength Biohybrid Gradient Hydrogel Scaffolds for Efficient Repair of Osteochondral Defect. Adv. Funct. Mater. 2018, 28, 1706644.
- Tan, Y.; Wang, D.; Xu, H.; Yang, Y.; An, W.; Yu, L.; Xiao, Z.; Xu, S. A Fast, Reversible, and Robust Gradient Nanocomposite Hydrogel Actuator with Water-Promoted Thermal Response. Macromol. Rapid. Commun. 2018, 39, 1700863.
- Wang, C.; Huang, W.; Zhou, Y.; He, L.; He, Z.; Chen, Z.; He, X.; Tian, S.; Liao, J.; Lu, B. 3D printing of bone tissue engineering scaffolds. Bioact. Mater. 2020, 5, 82–91.
- Gao, Q.; Niu, X.; Shao, L.; Zhou, L.; Lin, Z.; Sun, A.; Fu, J.; Chen, Z.; Hu, J.; Liu, Y. 3D printing of complex GelMA-based scaffolds with nanoclay. Biofabrication 2019, 11, 035006.
- Athas, J.C.; Nguyen, C.P.; Zarket, B.C.; Gargava, A.; Nie, Z.; Raghavan, S.R. Enzyme-Triggered Folding of Hydrogels: Toward a Mimic of the Venus Flytrap. ACS Appl. Mater. Interfaces 2016, 8, 19066–19074.
- Wu, Z.L.; Moshe, M.; Greener, J.; Therien-Aubin, H.; Nie, Z.; Sharon, E.; Kumacheva, E. Three-dimensional shape transformations of hydrogel sheets induced by small-scale modulation of internal stresses. Nat. Commun. 2013, 4, 1–7.
- Peng, X.; Liu, T.; Zhang, Q.; Shang, C.; Bai, Q.W.; Wang, H. Surface patterning of hydrogels for programmable and complex shape deformations by ion inkjet printing. Adv. Funct. Mater. 2017, 27, 1701962.
- Wang, J.; Li, T.; Chen, F.; Zhou, D.; Li, B.; Zhou, X.; Gan, T.; Handschuh-Wang, S.; Zhou, X. Softening and Shape Morphing of Stiff Tough Hydrogels by Localized Unlocking of the Trivalent Ionically Cross-Linked Centers. Macromol. Rapid. Commun. 2018, 39, 1800143.
- Huang, L.; Jiang, R.; Wu, J.; Song, J.; Bai, H.; Li, B.; Zhao, Q.; Xie, T. Ultrafast digital printing toward 4D shape changing materials. Adv. Funct. Mater. 2017, 29, 1605390.
- Ding, Z.; Yuan, C.; Peng, X.; Wang, T.; Qi, H.J.; Dunn, M.L. Direct 4D printing via active composite materials. Sci. Adv. 2017, 3, e1602890.




