
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Bercis Imge Ucar | + 4580 word(s) | 4580 | 2021-05-24 10:45:43 | | | |
| 2 | Vivi Li | Meta information modification | 4580 | 2021-06-01 11:14:29 | | |
Video Upload Options
Ischemia/reperfusion (I/R) injury is associated with substantial clinical implications, including a wide range of organs such as the brain, kidneys, lungs, heart, and many others. I/R injury (IRI) occurs due to the tissue injury following the reestablishment of blood supply to ischemic tissues, leading to enhanced aseptic inflammation and stimulation of oxidative stress via reactive oxygen and nitrogen species (ROS/RNS). Since ROS causes membrane lipids’ peroxidation, triggers loss of membrane integrity, denaturation of proteins, DNA damage, and cell death, oxidative stress plays a critical part in I/R pathogenesis. Therefore, ROS regulation could be a promising therapeutic strategy for IRI. In this context, Nrf2 (NF-E2-related factor 2) is a transcription factor that regulates the expression of several factors involved in the cellular defense against oxidative stress and inflammation, including heme oxygenase-1 (HO-1).
1. Introduction
1.1. Ischemia/Reperfusion Injury (IRI)
1.2. Oxidative Stress
1.3. Pro-Inflammatory Features of IRI
1.4. Nuclear Factor (Erythroid-Derived 2)-Related Factor 2 (Nrf2)/ Kelch-Like ECH-Associated Protein 1 (Keap1)/ Antioxidant Response Element (ARE)
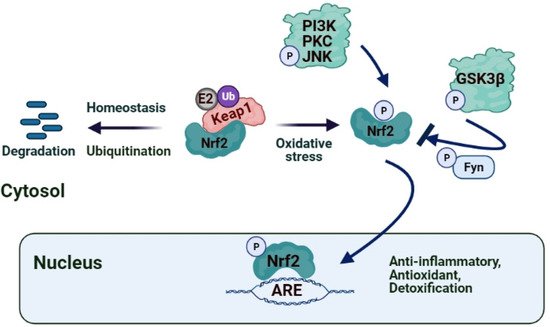
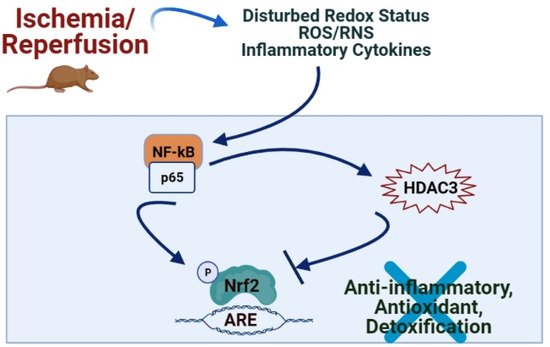
2. Role of Nrf2/Keap1/ARE Pathway in IRI
2.1. Disease-Specific Effects of Nrf2/Keap1/ARE Pathway within the IRI Concept
2.1.1. Central Nervous System (CNS)
2.1.2. Urinary System
2.1.3. Gastrointestinal System
2.1.4. Respiratory System
2.1.5. Hepatobiliary System
2.1.6. Cardiovascular System
3. Nrf2/Keap1/ARE Pathway as a Potential Target in IRI Therapy
3.1. Cardiac IRI
3.2. Hepatic IRI
3.3. Intestinal IRI
3.4. Cerebral IRI
3.5. Renal IRI
References
- Ucar, B.I.; Erikci, A.; Kosemehmetoglu, K.; Ozkul, C.; Iskit, A.B.; Ucar, G.; Zeren, S. Effects of endothelin receptor blockade and COX inhibition on intestinal I/R injury in a rat model: Experimental research. Int. J. Surg. 2020, 83, 89–97.
- Wu, M.-Y.; Yiang, G.-T.; Liao, W.-T.; Tsai, A.P.-Y.; Cheng, Y.-L.; Cheng, P.-W.; Li, C.-Y. Current Mechanistic Concepts in Ischemia and Reperfusion Injury. Cell. Physiol. Biochem. 2018, 46, 1650–1667.
- Padmavathi, G.; Ramkumar, K.M. MicroRNA mediated regulation of the major redox homeostasis switch, Nrf2, and its impact on oxidative stress-induced ischemic/reperfusion injury. Arch. Biochem. Biophys. 2021, 698, 108725.
- Chen, R.; Zhang, Y.-Y.; Lan, J.-N.; Liu, H.-M.; Li, W.; Wu, Y.; Leng, Y.; Tang, L.-H.; Hou, J.-B.; Sun, Q.; et al. Ischemic Postconditioning Alleviates Intestinal Ischemia-Reperfusion Injury by Enhancing Autophagy and Suppressing Oxidative Stress through the Akt/GSK-3β/Nrf2 Pathway in Mice. Oxidative Med. Cell. Longev. 2020, 2020, 1–14.
- Wu, L.; Xiong, X.; Wu, X.; Ye, Y.; Jian, Z.; Zhi, Z.; Gu, L. Targeting Oxidative Stress and Inflammation to Prevent Ischemia-Reperfusion Injury. Front. Mol. Neurosci. 2020, 13, 28.
- Zang, X.; Zhou, J.; Zhang, X.; Han, Y.; Chen, X. Ischemia Reperfusion Injury: Opportunities for Nanoparticles. ACS Biomater. Sci. Eng. 2020, 6, 6528–6539.
- Kalogeris, T.; Bao, Y.; Korthuis, R.J. Mitochondrial reactive oxygen species: A double edged sword in ischemia/reperfusion vs preconditioning. Redox Biol. 2014, 2, 702–714.
- Pi, J.; Zhang, Q.; Fu, J.; Woods, C.G.; Hou, Y.; Corkey, B.E.; Collins, S.; Andersen, M.E. ROS signaling, oxidative stress and Nrf2 in pancreatic beta-cell function. Toxicol. Appl. Pharmacol. 2010, 244, 77–83.
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.; Dinkova-Kostova, A.T.; Hayes, J.D. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015, 88, 108–146.
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Ind. J. Clin. Biochem. 2015, 30, 11–26.
- Vostrikova, S.M.; Grinev, A.B.; Gogvadze, V.G. Reactive Oxygen Species and Antioxidants in Carcinogenesis and Tumor Therapy. Biochemistry (Moscow) 2020, 85, 1254–1266.
- Mariathasan, S.; Weiss, D.S.; Newton, K.; McBride, J.; O’Rourke, K.; Roose-Girma, M.; Lee, W.P.; Weinrauch, Y.; Monack, D.M.; Dixit, V.M. Cryopyrin activates the inflammasome in response to toxins and ATP. Nat. Cell Biol. 2006, 440, 228–232.
- Cabrera-Fuentes, H.A.; Inserte, J.; Saffarzadeh, M.; Galuska, S.; Vijayan, V.; Barba, I.; Barreto, G.; Fischer, S.; Lochnit, G.; Ilinskaya, O.N.; et al. RNase1 prevents the damaging interplay between extracellular RNA and tumour necrosis factor-α in cardiac ischaemia/reperfusion injury. Thromb. Haemost. 2014, 112, 1110–1119.
- Yellon, D.M.; Hausenloy, D.J. Myocardial Reperfusion Injury. N. Engl. J. Med. 2007, 357, 1121–1135.
- Rauf, A.; Shah, M.; Yellon, D.M.; Davidson, S.M. Role of Caspase 1 in Ischemia/Reperfusion Injury of the Myocardium. J. Cardiovasc. Pharmacol. 2019, 74, 194–200.
- Mann, D.L. Innate Immunity and the Failing Heart. Circ. Res. 2015, 116, 1254–1268.
- Nian, M.; Lee, P.; Khaper, N.; Liu, P. Inflammatory Cytokines and Postmyocardial Infarction Remodeling. Circ. Res. 2004, 94, 1543–1553.
- Frangogiannis, N.G. Regulation of the Inflammatory Response in Cardiac Repair. Circ. Res. 2012, 110, 159–173.
- Horckmans, M.; Ring, L.; Duchene, J.; Santovito, D.; Schloss, M.J.; Drechsler, M.; Weber, C.; Soehnlein, O.; Steffens, S. Neutrophils orchestrate post-myocardial infarction healing by polarizing macrophages towards a reparative phenotype. Eur. Heart J. 2016, 38, 187–197.
- Kaspar, J.W.; Niture, S.K.; Jaiswal, A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med. 2009, 47, 1304–1309.
- O’Connell, M.A.; Hayes, J.D. The Keap1/Nrf2 pathway in health and disease: From the bench to the clinic. Biochem. Soc. Trans. 2015, 43, 687–689.
- Kumar, A.; Mittal, R. Nrf2: A potential therapeutic target for diabetic neuropathy. Inflammopharmacology 2017, 25, 393–402.
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim. Biophys. Acta (BBA)-Bioenerg. 2018, 1865, 721–733.
- Sova, M.; Saso, L. Design and development of Nrf2 modulators for cancer chemoprevention and therapy: A review. Drug Des. Dev. Ther. 2018, 12, 3181–3197.
- Zheng, F.; Gonçalves, F.M.; Abiko, Y.; Li, H.; Kumagai, Y.; Aschner, M. Redox toxicology of environmental chemicals causing oxidative stress. Redox Biol. 2020, 34, 101475.
- Cores, Á.; Piquero, M.; Villacampa, M.; León, R.; Menéndez, J.C. NRF2 Regulation Processes as a Source of Potential Drug Targets against Neurodegenerative Diseases. BioMolecules 2020, 10, 904.
- Wakabayashi, N.; Chartoumpekis, D.V.; Kensler, T.W. Crosstalk between Nrf2 and Notch signaling. Free Radic. Biol. Med. 2015, 88, 158–167.
- Wu, S.; Lu, H.; Bai, Y. Nrf2 in cancers: A double-edged sword. Cancer Med. 2019, 8, 2252–2267.
- Mitsuishi, Y.; Taguchi, K.; Kawatani, Y.; Shibata, T.; Nukiwa, T.; Aburatani, H.; Yamamoto, M.; Motohashi, H. Nrf2 Redirects Glucose and Glutamine into Anabolic Pathways in Metabolic Reprogramming. Cancer Cell 2012, 22, 66–79.
- Saha, S.; Buttari, B.; Panieri, E.; Profumo, E.; Saso, L. An Overview of Nrf2 Signaling Pathway and Its Role in Inflammation. Molecules 2020, 25, 5474.
- David, J.A.; Rifkin, W.J.; Rabbani, P.S.; Ceradini, D.J. The Nrf2/Keap1/ARE Pathway and Oxidative Stress as a Therapeutic Target in Type II Diabetes Mellitus. J. Diabetes Res. 2017, 2017, 1–15.
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.-L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317.
- Naidu, S.D.; Dinkova-Kostova, A.T. KEAP1, a cysteine-based sensor and a drug target for the prevention and treatment of chronic disease. Open Biol. 2020, 10, 200105.
- Brigelius-Flohé, R.; Flohé, L. Basic Principles and Emerging Concepts in the Redox Control of Transcription Factors. Antioxid. Redox Signal. 2011, 15, 2335–2381.
- Bellezza, I.; Mierla, A.L.; Minelli, A. Nrf2 and NF-κB and Their Concerted Modulation in Cancer Pathogenesis and Progression. Cancers 2010, 2, 483–497.
- Sandberg, M.; Patil, J.; D’Angelo, B.; Weber, S.G.; Mallard, C. NRF2-regulation in brain health and disease: Implication of cerebral inflammation. Neuropharmacology 2014, 79, 298–306.
- Liu, L.; Locascio, L.M.; Doré, S. Critical Role of Nrf2 in Experimental Ischemic Stroke. Front. Pharmacol. 2019, 10, 153.
- Wang, W.; Kang, J.; Li, H.; Su, J.; Wu, J.; Xu, Y.; Yu, H.; Xiang, X.; Yi, H.; Lu, Y.; et al. Regulation of endoplasmic reticulum stress in rat cortex by p62/ZIP through the Keap1-Nrf2-ARE signalling pathway after transient focal cerebral ischaemia. Brain Inj. 2013, 27, 924–933.
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; León, R. Nrf2–ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104.
- Malek, M.; Nematbakhsh, M. Renal ischemia/reperfusion injury; from pathophysiology to treatment. J. Ren. Inj. Prev. 2015, 4, 20–27.
- Shelton, L.M.; Park, B.K.; Copple, I.M. Role of Nrf2 in protection against acute kidney injury. Kidney Int. 2013, 84, 1090–1095.
- Nezu, M.; Suzuki, N. Roles of Nrf2 in Protecting the Kidney from Oxidative Damage. Int. J. Mol. Sci. 2020, 21, 2951.
- Zheng, Y.; Lu, H.; Huang, H. Desflurane Preconditioning Protects Against Renal Ischemia-Reperfusion Injury and Inhibits Inflammation and Oxidative Stress in Rats Through Regulating the Nrf2-Keap1-ARE Signaling Pathway. Drug Des. Dev. Ther. 2020, 14, 1351–1362.
- Nezu, M.; Souma, T.; Yu, L.; Suzuki, T.; Saigusa, D.; Ito, S.; Suzuki, N.; Yamamoto, M. Transcription factor Nrf2 hyperactivation in early-phase renal ischemia-reperfusion injury prevents tubular damage progression. Kidney Int. 2017, 91, 387–401.
- Hejazian, S.M.; Khatibi, S.M.H.; Barzegari, A.; Pavon-Djavid, G.; Soofiyani, S.R.; Hassannejhad, S.; Ahmadian, E.; Ardalan, M.; Vahed, S.Z. Nrf-2 as a therapeutic target in acute kidney injury. Life Sci. 2021, 264, 118581.
- Park, E.J.; Dusabimana, T.; Je, J.; Jeong, K.; Yun, S.P.; Kim, H.J.; Kim, H.; Park, S.W. Honokiol Protects the Kidney from Renal Ischemia and Reperfusion Injury by Upregulating the Glutathione Biosynthetic Enzymes. Biomedicines 2020, 8, 352.
- Leonard, M.O.; Kieran, N.E.; Howell, K.; Burne, M.J.; Varadarajan, R.; Dhakshinamoorthy, S.; Porter, A.G.; O’Farrelly, C.; Rabb, H.; Taylor, C.T. Reoxygenation-specific activation of the antioxidant transcription factor Nrf2 mediates cytoprotective gene expression in ischemia-reperfusion injury. FASEB J. 2006, 20, 2624–2626.
- Liu, M.; Grigoryev, D.N.; Crow, M.T.; Haas, M.; Yamamoto, M.; Reddy, S.P.; Rabb, H. Transcription factor Nrf2 is protective during ischemic and nephrotoxic acute kidney injury in mice. Kidney Int. 2009, 76, 277–285.
- Noel, S.; Martina, M.N.; Bandapalle, S.; Racusen, L.C.; Potteti, H.R.; A Hamad, A.R.; Reddy, S.P.; Rabb, H. T Lymphocyte–Specific Activation of Nrf2 Protects from AKI. J. Am. Soc. Nephrol. 2015, 26, 2989–3000.
- Hu, Q.; Ren, J.; Li, G.; Wu, J.; Wu, X.; Wang, G.; Gu, G.; Ren, H.; Hong, Z.; Li, J. The mitochondrially targeted antioxidant MitoQ protects the intestinal barrier by ameliorating mitochondrial DNA damage via the Nrf2/ARE signaling pathway. Cell Death Dis. 2018, 9, 1–15.
- Han, X.; Yao, W.; Liu, Z.; Li, H.; Zhang, Z.-J.; Hei, Z.; Xia, Z. Lipoxin A4 Preconditioning Attenuates Intestinal Ischemia Reperfusion Injury through Keap1/Nrf2 Pathway in a Lipoxin A4 Receptor Independent Manner. Oxidative Med. Cell. Longev. 2016, 2016, 1–12.
- Chi, X.; Yao, W.; Xia, H.; Jin, Y.; Li, X.; Cai, J.; Hei, Z. Elevation of HO-1 Expression Mitigates Intestinal Ischemia-Reperfusion Injury and Restores Tight Junction Function in a Rat Liver Transplantation Model. Oxidative Med. Cell. Longev. 2015, 2015, 1–12.
- Fei, L.; Jingyuan, X.; Fangte, L.; Huijun, D.; Liu, Y.; Ren, J.; Jinyuan, L.; Linghui, P. Preconditioning with rHMGB1 ameliorates lung ischemia–reperfusion injury by inhibiting alveolar macrophage pyroptosis via the Keap1/Nrf2/HO-1 signaling pathway. J. Transl. Med. 2020, 18, 1–12.
- Togo, T.; Hoshikawa, Y.; Noda, M.; Yabuki, H.; Hideki, M.; Watanabe, T.; Junichi, F.; Okada, Y. Nrf2 Promotes Recovery from Ische-mia-Reperfusion Injury After Lung Transplantation. In Proceedings of the D109. New Players in Lung Injury and Repair, San Diego, CA, USA, 23 May 2018; American Thoracic Society: New York, NY, USA, 2018; p. A7603.
- Kawamura, T.; Wakabayashi, N.; Shigemura, N.; Huang, C.-S.; Masutani, K.; Tanaka, Y.; Noda, K.; Peng, X.; Takahashi, T.; Billiar, T.R.; et al. Hydrogen gas reduces hyperoxic lung injury via the Nrf2 pathway in vivo. Am. J. Physiol. Cell. Mol. Physiol. 2013, 304, L646–L656.
- Lyu, S.; Xihua Lu Tingkun, L.I.; Sun, Y.; Wang, L.; Wang, D. Role of Nrf2/ARE signaling pathway in propofol-induced reduction of lung ischemia-reperfusion injury in aged rats. Chin. J. Anesthesiol. 2018, 38, 421–424.
- Dong, H.; Qiang, Z.; Chai, D.; Peng, J.; Xia, Y.; Hu, R.; Jiang, H. Nrf2 inhibits ferroptosis and protects against acute lung injury due to intestinal ischemia reperfusion via regulating SLC7A11 and HO-1. Aging 2020, 12, 12943–12959.
- Xu, D.; Chen, L.; Chen, X.; Wen, Y.; Yu, C.; Yao, J.; Wu, H.; Wang, X.; Xia, Q.; Kong, X. The triterpenoid CDDO-imidazolide ameliorates mouse liver ischemia-reperfusion injury through activating the Nrf2/HO-1 pathway enhanced autophagy. Cell Death Dis. 2017, 8, e2983.
- Zhang, Q.; Lai, Y.; Deng, J.; Wang, M.; Wang, Z.; Wang, M.; Zhang, Y.; Yang, X.; Zhou, X.; Jiang, H. Vagus Nerve Stimulation Attenuates Hepatic Ischemia/Reperfusion Injury via the Nrf2/HO-1 Pathway. Oxidative Med. Cell. Longev. 2019, 2019, 9549506.
- Yi, Z.; Deng, M.; Scott, M.J.; Fu, G.; Loughran, P.A.; Lei, Z.; Li, S.; Sun, P.; Yang, C.; Li, W.; et al. Immune-Responsive Gene 1/Itaconate Activates Nuclear Factor Erythroid 2–Related Factor 2 in Hepatocytes to Protect Against Liver Ischemia–Reperfusion Injury. Hepatology 2020, 72, 1394–1411.
- Jia, X.; Zhang, L.; Mao, X. S-propranolol protected H9C2 cells from ischemia/reperfusion-induced apoptosis via downregulation of RACK1 Gene. Int. J. Clin. Exp. Pathol. 2015, 8, 10335.
- Chen, Q.M.; Maltagliati, A.J. Nrf2 at the heart of oxidative stress and cardiac protection. Physiol. Genom. 2018, 50, 77–97.
- Hu, H.; Zhai, C.; Qian, G.; Gu, A.; Liu, J.; Ying, F.; Xu, W.; Jin, D.; Wang, H.; Hu, H.; et al. Protective effects of tanshinone IIA on myocardial ischemia reperfusion injury by reducing oxidative stress, HMGB1 expression, and inflammatory reaction. Pharm. Biol. 2015, 53, 1752–1758.
- Yang, G.; Wang, F.; Wang, Y.; Yu, X.; Yang, S.; Xu, H.; Xing, J. Protective effect of tanshinone IIA on H2O2-induced oxidative stress injury in rat cardiomyocytes by activating Nrf2 pathway. J. Recept. Signal Transduct. 2020, 40, 264–272.
- Shen, Y.; Liu, X.; Shi, J.; Wu, X. Involvement of Nrf2 in myocardial ischemia and reperfusion injury. Int. J. Biol. Macromol. 2019, 125, 496–502.
- Vashi, R.; Patel, B.M. NRF2 in Cardiovascular Diseases: A Ray of Hope! J. Cardiovasc. Transl. Res. 2020, 1–14.
- Lorita, J.; Soley, M.; Ramírez, I. Epidermal growth factor protects the heart against low-flow ischemia-induced injury. J. Physiol. Biochem. 2010, 66, 55–62.
- Ma, J.; Jin, G. Epidermal growth factor protects against myocardial ischaemia reperfusion injury through activating Nrf2 signalling pathway. Free Radic. Res. 2019, 53, 313–323.
- Katsumata, Y.; Shinmura, K.; Sugiura, Y.; Tohyama, S.; Matsuhashi, T.; Ito, H.; Yan, X.; Ito, K.; Yuasa, S.; Ieda, M.; et al. Endogenous Prostaglandin D 2 and Its Metabolites Protect the Heart Against Ischemia–Reperfusion Injury by Activating Nrf2. Hypertension 2014, 63, 80–87.
- Liu, Y.-F.; Chu, Y.-Y.; Zhang, X.-Z.; Zhang, M.; Xie, F.-G.; Zhou, M.; Wen, H.-H.; Shu, A.-H. TGFβ1 protects myocardium from apoptosis and oxidative damage after ischemia reperfusion. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1551–1558.
- Bai, Y.; Chen, Q.; Sun, Y.-P.; Wang, X.; Lv, L.; Zhang, L.-P.; Liu, J.-S.; Zhao, S.; Wang, X.-L. Sulforaphane protection against the development of doxorubicin-induced chronic heart failure is associated with Nrf2 Upregulation. Cardiovasc. Ther. 2017, 35, e12277.
- Li, W.; Wu, M.; Tang, L.; Pan, Y.; Liu, Z.; Zeng, C.; Wang, J.; Wei, T.; Liang, G. Novel curcumin analogue 14p protects against myocardial ischemia reperfusion injury through Nrf2-activating anti-oxidative activity. Toxicol. Appl. Pharmacol. 2015, 282, 175–183.
- Zhang, Y.; Qiao, B.; Gao, F.; Wang, H.; Miao, S.; Zhao, H. Melatonin protects H9c2 cells against ischemia/reperfusion-induced apoptosis and oxidative stress via activation of the Nrf2 signaling pathway. Mol. Med. Rep. 2018, 18, 3497–3505.
- Sun, G.; Li, Y.; Ji, Z. Atorvastatin attenuates inflammation and oxidative stress induced by ischemia/reperfusion in rat heart via the Nrf2 transcription factor. Int. J. Clin. Exp. Med. 2015, 8, 14837–14845.
- Zhao, T.; Chen, S.; Wang, B.; Cai, D. L-Carnitine Reduces Myocardial Oxidative Stress and Alleviates Myocardial Ischemia-Reperfusion Injury by Activating Nuclear Transcription-Related Factor 2 (Nrf2)/Heme Oxygenase-1 (HO-1) Signaling Pathway. Med. Sci. Monit. 2020, 26, e923251.
- Bubb, K.J.; Kok, C.; Tang, O.; Rasko, N.B.; Birgisdottir, A.B.; Hansen, T.; Ritchie, R.; Bhindi, R.; Reisman, S.A.; Meyer, C.; et al. The NRF2 activator DH404 attenuates adverse ventricular remodeling post-myocardial infarction by modifying redox signalling. Free Radic. Biol. Med. 2017, 108, 585–594.
- Enayati, A.; Khori, V.; Saeedi, Y.; Yassa, N. Antioxidant Activity and Cardioprotective Effect of Potentilla reptans L. via Ischemic Preconditioning (IPC). Res. J. Pharmacogn. 2019, 6, 19–27.
- Cheng, L.; Jin, Z.; Zhao, R.; Ren, K.; Deng, C.; Yu, S. Resveratrol attenuates inflammation and oxidative stress induced by myocardial ischemia-reperfusion injury: Role of Nrf2/ARE pathway. Int. J. Clin. Exp. Med. 2015, 8, 10420–10428.
- Xu, G.; Zhao, X.; Fu, J.; Wang, X. Resveratrol increase myocardial Nrf2 expression in type 2 diabetic rats and alleviate myocardial ischemia/reperfusion injury (MIRI). Ann. Palliat. Med. 2019, 8, 565–575.
- Yu, H.; Shi, L.; Zhao, S.; Sun, Y.; Gao, Y.; Sun, Y.; Qi, G. Triptolide Attenuates Myocardial Ischemia/Reperfusion Injuries in Rats by Inducing the Activation of Nrf2/HO-1 Defense Pathway. Cardiovasc. Toxicol. 2015, 16, 325–335.
- Sun, W.; Wang, Z.; Sun, M.; Huang, W.; Wang, Y.; Wang, Y. Aloin antagonizes stimulated ischemia/reperfusion-induced damage and inflammatory response in cardiomyocytes by activating the Nrf2/HO-1 defense pathway. Cell Tissue Res. 2021, 1–10.
- Uddandrao, V.V.S.; Brahmanaidu, P.; Nivedha, P.R.; Vadivukkarasi, S.; Saravanan, G. Beneficial Role of Some Natural Products to Attenuate the Diabetic Cardiomyopathy Through Nrf2 Pathway in Cell Culture and Animal Models. Cardiovasc. Toxicol. 2017, 18, 199–205.
- Xiao, C.; Xia, M.-L.; Wang, J.; Zhou, X.-R.; Lou, Y.-Y.; Tang, L.-H.; Zhang, F.-J.; Yang, J.-T.; Qian, L.-B. Luteolin Attenuates Cardiac Ischemia/Reperfusion Injury in Diabetic Rats by Modulating Nrf2 Antioxidative Function. Oxidative Med. Cell. Longev. 2019, 2019, 1–9.
- Wang, D.; Zhang, X.; Li, D.; Hao, W.; Meng, F.; Wang, B.; Han, J.; Zheng, Q. Kaempferide Protects against Myocardial Ischemia/Reperfusion Injury through Activation of the PI3K/Akt/GSK-3β Pathway. Mediat. Inflamm. 2017, 2017, 1–11.
- Duan, J.; Guan, Y.; Mu, F.; Guo, C.; Zhang, E.; Yin, Y.; Wei, G.; Zhu, Y.; Cui, J.; Cao, J.; et al. Protective effect of butin against ischemia/reperfusion-induced myocardial injury in diabetic mice: Involvement of the AMPK/GSK-3β/Nrf2 signaling pathway. Sci. Rep. 2017, 7, srep41491.
- Wang, X.; Yuan, B.; Cheng, B.; Liu, Y.; Zhang, B.; Wang, X.; Lin, X.; Yang, B.; Gong, G. Crocin Alleviates Myocardial Ischemia/Reperfusion-Induced Endoplasmic Reticulum Stress via Regulation of miR-34a/Sirt1/Nrf2 Pathway. Shock 2019, 51, 123–130.
- Hou, J.-Y.; Liu, Y.; Liu, L.; Li, X.-M. Protective effect of hyperoside on cardiac ischemia reperfusion injury through inhibition of ER stress and activation of Nrf2 signaling. Asian Pac. J. Trop. Med. 2016, 9, 76–80.
- Li, Y.; Zhang, H. Soybean isoflavones ameliorate ischemic cardiomyopathy by activating Nrf2-mediated antioxidant responses. Food Funct. 2017, 8, 2935–2944.
- Xu, D.; Xu, M.; Jeong, S.; Qian, Y.; Wu, H.; Xia, Q.; Kong, X. The Role of Nrf2 in Liver Disease: Novel Molecular Mechanisms and Therapeutic Approaches. Front. Pharmacol. 2019, 9, 1428.
- Ibrahim, S.G.; El-Emam, S.Z.; Mohamed, E.A.; Ellah, M.F.A. Dimethyl fumarate and curcumin attenuate hepatic ischemia/reperfusion injury via Nrf2/HO-1 activation and anti-inflammatory properties. Int. Immunopharmacol. 2020, 80, 106131.
- Chen, K.; Li, J.J.; Li, S.N.; Feng, J.; Liu, T.; Wang, F.; Dai, W.Q.; Xia, Y.J.; Lu, J.; Zhou, Y.Q.; et al. 15-Deoxy-Δ12,14-prostaglandin J2 alleviates hepatic ischemia-reperfusion injury in mice via inducing antioxidant response and inhibiting apoptosis and au-tophagy. Acta Pharmacol. Sinica 2017, 38, 672–687.
- Chi, X.; Zhang, R.; Shen, N.; Jin, Y.; Alina, A.; Yang, S.; Lin, S. Sulforaphane reduces apoptosis and oncosis along with protecting liver injury-induced ischemic reperfusion by activating the Nrf2/ARE pathway. Hepatol. Int. 2015, 9, 321–329.
- Rabie, M.A.; Zaki, H.F.; Sayed, H.M. Telluric acid ameliorates hepatic ischemia reperfusion-induced injury in rats: Involvement of TLR4, Nrf2, and PI3K/Akt signaling pathways. Biochem. Pharmacol. 2019, 168, 404–411.
- Zhuang, L.; Ding, W.; Zhang, Q.; Ding, W.; Xu, X.; Yu, X.; Xi, D. TGR5 Attenuated Liver Ischemia-Reperfusion Injury by Activating the Keap1-Nrf2 Signaling Pathway in Mice. Inflammation 2020, 10, 1–14.
- Ge, M.; Chen, H.; Zhu, Q.; Cai, J.; Chen, C.; Yuan, D.; Chaojin, C.; Yao, W.; Hei, Z. Propofol post-conditioning alleviates hepatic ischaemia reperfusion injuryviaBRG1-mediated Nrf2/HO-1 transcriptional activation in human and mice. J. Cell. Mol. Med. 2017, 21, 3693–3704.
- Kamel, E.O.; Hassanein, E.H.M.; Ahmed, M.A.; Ali, F.E.M. Perindopril Ameliorates Hepatic Ischemia Reperfusion Injury Via Regulation of NF-κB-p65/TLR-4, JAK1/STAT-3, Nrf-2, and PI3K/Akt/mTOR Signaling Pathways. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2020, 303, 1935–1949.
- Cui, R.-S.; Wang, K.; Wang, Z.-L. Sevoflurane anesthesia alters cognitive function by activating inflammation and cell death in rats. Exp. Ther. Med. 2018, 15, 4127–4130.
- Ma, H.; Yang, B.; Yu, L.; Gao, Y.; Ye, X.; Liu, Y.; Li, Z.; Li, H.; Li, E. Sevoflurane protects the liver from ischemia-reperfusion injury by regulating Nrf2/HO-1 pathway. Eur. J. Pharmacol. 2021, 898, 173932.
- Li, Z.; Wang, Y.; Zhang, Y.; Wang, X.; Gao, B.; Li, Y.; Li, R.; Wang, J. Protective Effects of Fisetin on Hepatic Ischemia-reperfusion Injury Through Alleviation of Apoptosis and Oxidative Stress. Arch. Med. Res. 2021, 52, 163–173.
- Yuan, B.; Huang, H.; Qu, S.; Zhang, H.; Lin, J.; Jin, L.; Yang, S.; Zeng, Z. Gastrodin Pretreatment Protects Liver Against Ischemia-Reperfusion Injury via Activation of the Nrf2/HO-1 Pathway. Am. J. Chin. Med. 2020, 48, 1159–1178.
- Lv, H.; An, B.; Yu, Q.; Cao, Y.; Liu, Y.; Li, S. The hepatoprotective effect of myricetin against lipopolysaccharide and D-galactosamine-induced fulminant hepatitis. Int. J. Biol. Macromol. 2020, 155, 1092–1104.
- Jin, C.; Fu, W.-L.; Zhang, D.-D.; Xing, W.-W.; Xia, W.-R.; Wei, Z.; Zou, M.-J.; Zhu, X.-M.; Xu, D.-G. The protective role of IL-1Ra on intestinal ischemia reperfusion injury by anti-oxidative stress via Nrf2/HO-1 pathway in rat. Biomed. J. 2019, 42, 36–45.
- Du, J.; Fan, X.; Yang, B.; Chen, Y.; Liu, K.-X.; Zhou, J. Irisin pretreatment ameliorates intestinal ischemia/reperfusion injury in mice through activation of the Nrf2 pathway. Int. Immunopharmacol. 2019, 73, 225–235.
- Gendy, A.; Soubh, A.; Al-Mokaddem, A.; El-Sayed, M.K. Dimethyl fumarate protects against intestinal ischemia/reperfusion lesion: Participation of Nrf2/HO-1, GSK-3β and Wnt/β-catenin pathway. Biomed. Pharmacother. 2021, 134, 111130.
- Zu, G.; Zhou, T.; Che, N.; Zhang, X. Salvianolic Acid A Protects Against Oxidative Stress and Apoptosis Induced by Intestinal Ischemia-Reperfusion Injury Through Activation of Nrf2/HO-1 Pathways. Cell. Physiol. Biochem. 2018, 49, 2320–2332.
- Chen, S.; Li, X.; Wang, Y.; Mu, P.; Chen, C.; Huang, P.; Liu, D. Ginsenoside Rb1 attenuates intestinal ischemia/reperfusion-induced inflammation and oxidative stress via activation of the PI3K/Akt/Nrf2 signaling pathway. Mol. Med. Rep. 2019, 19, 3633–3641.
- Liu, C.; Zhu, C.; Wang, G.; Xu, R.; Zhu, Y. Higenamine regulates Nrf2-HO-1-Hmgb1 axis and attenuates intestinal ischemia–reperfusion injury in mice. Inflamm. Res. 2015, 64, 395–403.
- Alfieri, A.; Srivastava, S.; Siow, R.C.M.; Cash, D.; Modo, M.; Duchen, M.R.; Fraser, P.A.; Williams, S.C.R.; Mann, G.E. Sulforaphane preconditioning of the Nrf2/HO-1 defense pathway protects the cerebral vasculature against blood–brain barrier disruption and neurological deficits in stroke. Free Radic. Biol. Med. 2013, 65, 1012–1022.
- Ding, Y.; Chen, M.; Wang, M.; Li, Y.; Wen, A. Posttreatment with 11-Keto-β-Boswellic Acid Ameliorates Cerebral Ischemia–Reperfusion Injury: Nrf2/HO-1 Pathway as a Potential Mechanism. Mol. Neurobiol. 2014, 52, 1430–1439.
- Hu, S.; Wu, Y.; Zhao, B.; Hu, H.; Zhu, B.; Sun, Z.; Li, P.; Du, S. Panax notoginseng Saponins Protect Cerebral Microvascular Endothelial Cells against Oxygen-Glucose Deprivation/Reperfusion-Induced Barrier Dysfunction via Activation of PI3K/Akt/Nrf2 Antioxidant Signaling Pathway. Molecules 2018, 23, 2781.
- Peng, B.; Zhao, P.; Lu, Y.-P.; Chen, M.-M.; Sun, H.; Wu, X.-M.; Zhu, L. Z-ligustilide activates the Nrf2/HO-1 pathway and protects against cerebral ischemia–reperfusion injury in vivo and in vitro. Brain Res. 2013, 1520, 168–177.
- Yamauchi, K.; Nakano, Y.; Imai, T.; Takagi, T.; Tsuruma, K.; Shimazawa, M.; Iwama, T.; Hara, H. A novel nuclear factor erythroid 2-related factor 2 (Nrf2) activator RS9 attenuates brain injury after ischemia reperfusion in mice. Neuroscience 2016, 333, 302–310.
- Shi, Y.-S.; Zhang, Y.; Liu, B.; Li, C.-B.; Wu, J.; Li, Y. Nomilin protects against cerebral ischemia–reperfusion induced neurological deficits and blood–brain barrier disruption via the Nrf2 pathway. Food Funct. 2019, 10, 5323–5332.
- Kunze, R.; Urrutia, A.; Hoffmann, A.; Liu, H.; Helluy, X.; Pham, M.; Reischl, S.; Korff, T.; Marti, H.H. Dimethyl fumarate attenuates cerebral edema formation by protecting the blood–brain barrier integrity. Exp. Neurol. 2015, 266, 99–111.
- Singh, D.; Reeta, K.; Sharma, U.; Jagannathan, N.; Dinda, A.; Gupta, Y. Neuro-protective effect of monomethyl fumarate on ischemia reperfusion injury in rats: Role of Nrf2/HO1 pathway in peri-infarct region. Neurochem. Int. 2019, 126, 96–108.
- Liu, X.; Zhu, X.; Chen, M.; Ge, Q.; Shen, Y.; Pan, S. Resveratrol protects PC12 cells against OGD/R-induced apoptosis via the mitochondrial-mediated signaling pathway. Acta Biochim. Biophys. Sin. 2016, 48, 342–353.
- Zenkov, N.K.; Kozhin, P.M.; Chechushkov, A.V.; Martinovich, G.G.; Kandalintseva, N.V.; Menshchikova, E. Mazes of Nrf2 regulation. Biochemistry (Moscow) 2017, 82, 556–564.
- Xu, H.; Deng, R.; Li, E.T.; Shen, J.; Wang, M. Pinosylvin provides neuroprotection against cerebral ischemia and reperfusion injury through enhancing PINK1/Parkin mediated mitophagy and Nrf2 pathway. J. Funct. Foods 2020, 71, 104019.
- Lei, X.; Li, H.; Li, M.; Dong, Q.; Zhao, H.; Zhang, Z.; Sun, B.; Mao, L. The novel Nrf2 activator CDDO-EA attenuates cerebral ischemic injury by promoting microglia/macrophage polarization toward M2 phenotype in mice. CNS Neurosci. Ther. 2021, 27, 82–91.
- Han, P.; Qin, Z.; Tang, J.; Xu, Z.; Li, R.; Jiang, X.; Yang, C.; Xing, Q.; Qi, X.; Tang, M.; et al. RTA-408 Protects Kidney from Ischemia-Reperfusion Injury in Mice via Activating Nrf2 and Downstream GSH Biosynthesis Gene. Oxidative Med. Cell. Longev. 2017, 2017, 1–15.
- Shi, S.; Lei, S.; Tang, C.; Wang, K.; Xia, Z. Melatonin attenuates acute kidney ischemia/reperfusion injury in diabetic rats by activation of the SIRT1/Nrf2/HO-1 signaling pathway. Biosci. Rep. 2019, 39.
- Zúñiga-Toalá, A.; Zatarain-Barrón, Z.L.; Hernández-Pando, R.; Negrette-Guzmán, M.; Huerta-Yepez, S.; Torres, I.; Pinzón, E.; Tapia, E.; Pedraza-Chaverri, J. Nordihydroguaiaretic acid induces Nrf2 nuclear translocation in vivo and attenuates renal damage and apoptosis in the ischemia and reperfusion model. Phytomedicine 2013, 20, 775–779.




