1000/1000
Hot
Most Recent

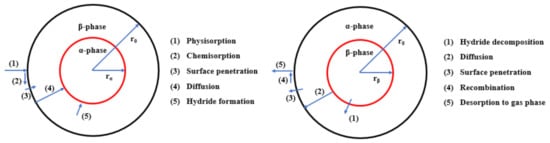
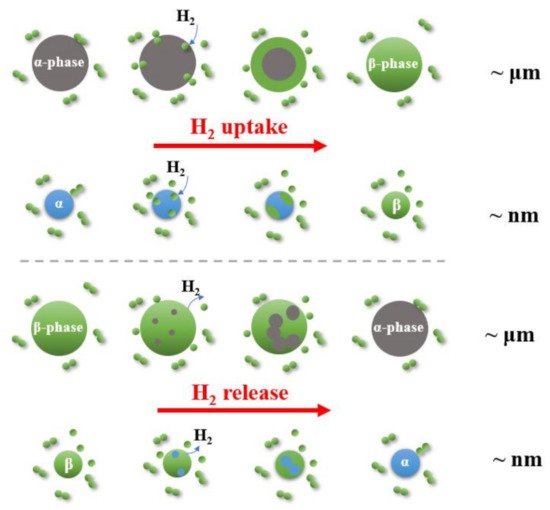
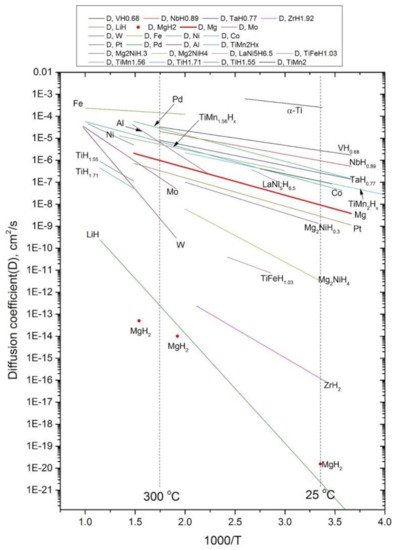
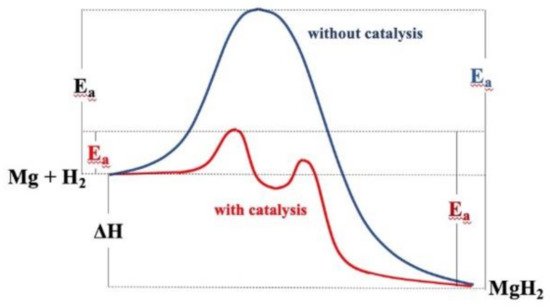
Magnesium-based hydrides are considered as promising candidates for solid-state hydrogen storage and thermal energy storage, due to their high hydrogen capacity, reversibility, and elemental abundance of Mg. To improve the sluggish kinetics of MgH2, catalytic doping using Ti-based catalysts is regarded as an effective approach to enhance Mg-based materials.
|
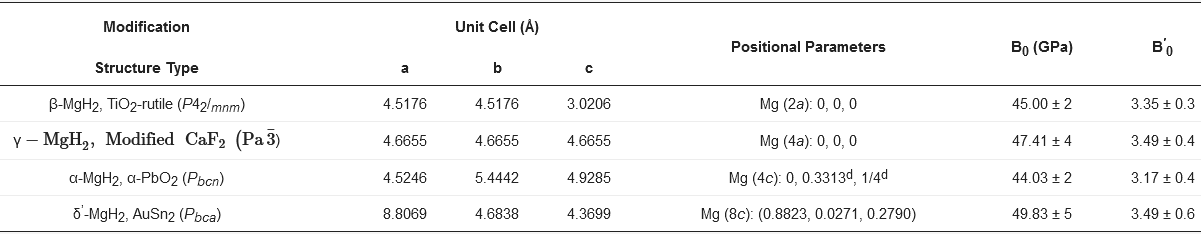
Thermodynamic Parameters |
Values |
|---|---|
|
Formation enthalpy, kJ/(mol·H2) |
−74.5 |
|
Formation entropy, J/(mol·H2·K) |
−135 |
|
Hydrogen Storage Capacity (Theoretical) |
|
|
Gravimetric capacity, wt% |
7.6 |
|
Volumetric capacity, g/(L·H2) |
110 |
|
Thermal Energy Storage Capacity (Theoretical) |
|
|
Gravimetric capacity, kJ/kg |
2204 |
|
Volumetric capacity, kJ/dm3 |
1763 |
|
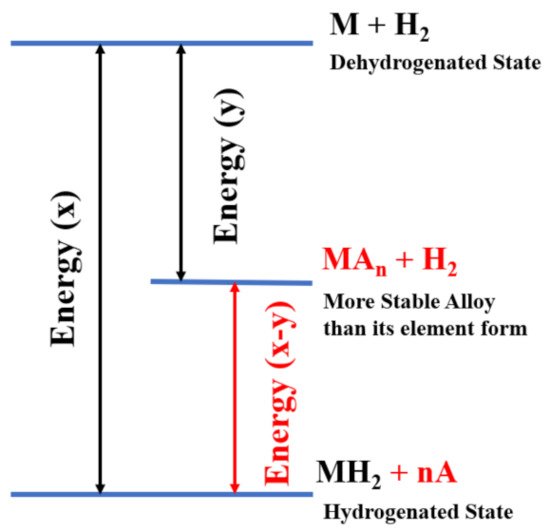
Metal |
Dissociation Energy (eV) |
|---|---|
|
Pure Mg |
0.87, 0.40, 0.50, 1.15, 1.05, 0.95, 1.00 |
|
Ti-doped Mg |
Null, negligible |
|
Ni-doped Mg |
0.06 |
|
V-doped Mg |
Null |
|
Cu-doped Mg |
0.56 |
|
Pd-doped Mg |
0.39 |
|
Fe-doped Mg |
0.03 |
|
Ag-doped Mg |
1.18 |
|
Materials |
Synthetic Methods |
Hydrogen Storage Properties |
Reference |
|||
|---|---|---|---|---|---|---|
|
Desorption Kinetics |
Eades (kJ/mol) |
Absorption Kinetics |
Eaabs (kJ/mol) |
|||
|
Titanium/Titanium Hydrides |
||||||
|
Mg-2%Ti |
Inert gas condensation |
Des: 4.50%/320 °C/0.2 bar/25 min |
Abs: 4.80%/320 °C/8 bar/21 min |
[53] |
||
|
MgH2 + 2 at% Ti |
Ball milling (argon) |
Des: 6.32 wt%/623 K/35 kPa/0.5 h |
Abs: 6.32 wt%/623 K/2000 kPa/4 min |
[54] |
||
|
Cold rolling (5 times, air) |
Des: 6.00 wt%/623 K/35 kPa/0.5 h |
Abs: 5.70 wt%/623 K/2000 kPa/4 min |
||||
|
MgH2-4 mol% Ti |
Ball milling |
Des: 1.10%/573 K/2 MPa/5 min |
Abs: 6.40%/573 K/2 MPa/5 min |
[55] |
||
|
MgH2-5 at% Ti |
Ball milling |
Des Temperature: 235.6 °C |
70.11 |
[56] |
||
|
MgH2-5 at% Ti |
Ball milling |
Des: 5.50%/523 K/0.015 MPa/20 min |
71.1 |
Abs: 4.20%/373 K/1.0 MPa/15 min |
[57] |
|
|
MgH2-5 at% Ti |
Ball milling |
Des: 5.20%/573 K/0.03 MPa/15 min |
Abs: 6.70%/ 573 K/0.8 MPa/15 min |
[58] |
||
|
Mg-5% Ti |
Chemical vapor synthesis |
104 |
[59] |
|||
|
Mg-14 at% Ti |
Gas phase condensation |
35 |
52 |
[60] |
||
|
Mg-22 at% Ti |
31 |
47 |
||||
|
MgH2-15% Ti |
Ball milling |
Des: 0.12%/573 K/1 bar/60 min |
Abs: 3.48%/573 K/12 bar/60 min |
[61] |
||
|
Mg0.9Ti0.1 |
Ball milling |
76 |
Abs: 6.62% (after milling) |
[62] |
||
|
Mg0.75Ti0.25 |
Ball milling |
88 |
Abs: 6.18% (after milling) |
|||
|
Mg0.5Ti0.5 |
Ball milling |
91 |
Abs: 5.21% (after milling) |
|||
|
MgH2-20% Ti |
Ball milling |
72 ± 3 |
[63] |
|||
|
MgH2-coated Ti |
Ball milling |
Des: 5.00%/250 °C/15 min (TPD) Des Temperature: 175 °C |
[64] |
|||
|
Mg83.5Ti16.5 |
Inert gas condensation |
Des: 2.50%/300 °C/0.15 bar/2 min |
Abs: 2.20%/300 °C/9 bar/1 min |
[65] |
||
|
15Mg-Ti |
Chemical method |
72.2 |
[66] |
|||
|
MgH2-4 mol% TiH2 |
Ball milling |
Des: 0.70%/573 K/2 MPa/5 min |
Abs: 6.10%/573 K/2 MPa/5 min |
[55] |
||
|
MgH2-5 at% TiH2 |
Ball milling |
Des: 5.80%/270 °C/0.12 bar/10 min Des Temperature: 235.5 °C |
67.24 |
Abs: 2.70%/25 °C/1 bar/250 min |
[56] |
|
|
10MgH2-TiH2 |
Ball milling |
73 |
[67] |
|||
|
7MgH2-TiH2 |
Ball milling |
71 |
[68] |
|||
|
4MgH2-TiH2 |
Ball milling |
68 |
[68] |
|||
|
MgH2-10 mol% TiH2 |
Ball milling |
Abs: 5.70%/240 °C/2 MPa/200 s |
16.4 |
[69] |
||
|
MgH2-10% TiH2 |
Ball milling |
24.2 |
[70] |
|||
|
MgH2-10% TiH2 |
Ball milling |
17.9 |
[71] |
|||
|
Mg-9.2% TiH1.971-3.7% TiH1.5 |
Ball milling |
Des: 4.10%/573 K/100 Pa/20 min |
46.2 |
Abs: 4.30%/298 K/4 MPa/10 min |
12.5 |
[72] |
|
Mg0.65Ti0.35D1.2 |
Ball milling |
17 |
[73] |
|||
|
Titanium Oxides |
||||||
|
MgH2-10% TiO2 |
Ball milling |
Des: 6.00%/300 °C/vacuum/20 min |
Abs: 6.00%/300 °C/0.84 MPa/5 min |
[74] |
||
|
Mg-20% TiO2 |
Reactive ball milling |
Des: 4.40%/350 °C/1 bar/8.5 min |
Abs: 3.80%/350 °C/20 bar/2 min |
[75] |
||
|
MgH2-6% TiO2 |
Ball milling |
145.8 ± 14.2 |
[76] |
|||
|
MgH2 + 10% TiO2 |
Ball milling |
Des Temperature: 200 °C |
75.50 |
[77] |
||
|
Titanium Halides |
||||||
|
MgH2-10% TiF4 |
Ball milling |
Des Temperature: 216.7 °C |
71 |
(Des: 6.6%) |
[78] |
|
|
MgH2-10% TiF4 |
Ball milling (2 h, argon) |
Des Temperature: 154 °C |
70 |
[79] |
||
|
MgH2 + 10% TiF4 |
Ball milling |
Des Temperature: 150 °C |
70 |
[77] |
||
|
MgH2-4 mol% TiF3 |
Ball milling |
Des: 4.50%/573 K/2 MPa/5 min |
Abs: 5.10%/573 K/2 MPa/5 min |
[55] |
||
|
MgH2-4 mol% TiCl3 |
Ball milling |
Des: 3.70%/573 K/2 MPa/5 min |
Abs: 5.30%/573 K/2 MPa/5 min |
[55] |
||
|
MgH2-7% TiCl3 |
Ball milling |
Des temperature: 274 °C |
85 |
[80] |
||
|
Titanium Alloys |
||||||
|
MgH2-5a% TiAl |
Ball milling |
Des: 4.90%/270 °C/0.12 bar/10 min Des Temperature: 219.6 °C |
65.08 |
Abs: 2.50%/25 °C/1 bar/250 min |
[56] |
|
|
MgH2-5 a% Ti3Al |
Ball milling |
Des Temperature: 232.3 °C |
70.61 |
[56] |
||
|
Mg85Al7.5Ti7.5 |
DC-magnetron co-sputtering |
Des: 5.30%/200 °C/vacuum/20 min |
Abs: 5.60%/200 °C/3 bar/0.5 min |
[81] |
||
|
Mg0.63Ti0.27Si0.10D1.1 |
Ball milling |
27 |
[73] |
|||
|
MgH2-5 at%TiNi |
Ball milling |
Des Temperature: 242.4 °C |
73.09 |
[56] |
||
|
15Mg-Ti-0.75Ni |
Chemical method |
63.7 |
[66] |
|||
|
Mg0.63Ti0.27Ni0.10D1.3 |
Ball milling |
21 |
[73] |
|||
|
MgH2-5at%TiNb |
Ball milling |
Des: 5.90%/27 °C/0.12 bar/10 min Des Temperature: 231.3 °C |
71.72 |
Abs: 2.80%/25 °C/1 bar/250 min |
[56] |
|
|
MgH2-5at% Cr-5a% Ti |
Film |
Des: 6.00%/200 °C/5 mbar/25 min |
Abs: 6.20%/200 °C/3 bar/10 min |
[82] |
||
|
MgH2-7 at% Cr-13 at% Ti |
Film |
Des: 5.00%/200 °C/5 mbar/25 min |
Abs: 5.60%/200 °C/3 bar/10 min |
|||
|
MgH2-5 at% TiFe |
Ball milling |
Des: 5.20%/270 °C/0.12 bar/10 min Des Temperature: 237.7 °C |
72.63 |
Abs: 3.00%/25 °C/1 bar/250 min |
[56] |
|
|
MgH2-5% FeTi |
Ball milling |
Abs: 2.30%/150 °C/2 MPa/5 min |
21 |
[83] |
||
|
MgH2-5 at% TiMn2 |
Ball milling |
Des: 4.80%/270 °C/0.12 bar/10 min Des Temperature: 219.7 °C |
74.22 |
Abs: 3.20%/25 °C/1 bar/250 min |
[56] |
|
|
MgH2-10% TiMn2 |
Ball milling |
22.6 |
[70] |
|||
|
MgH2-5% VTi |
Ball milling |
Abs: 3.30%/150 °C/2 MPa/5 min |
10.4 |
[83] |
||
|
Mg87.5Ti9.6V2.9 |
Hydrogen plasma metal reaction |
Des: 4.00%/300 °C/1 mbar/5 min |
73.8 |
Abs: 4.80%/200 °C/40 bar/5 min |
29.2 |
[84] |
|
MgH2-5 at% TiVMn |
Ball milling |
Des: 5.70%/270 °C/0.12 bar/10 min Des Temperature: 216.7 °C |
85.20 |
Abs: 3.00%/25 °C/1 bar/250 min |
[56] |
|
|
Multiple Catalysts |
||||||
|
Mg-10% Ti-10% Pd |
Ball milling |
114 ± 4 |
[85] |
|||
|
Mg-TiH1.971-TiH1.5-ZrH1.66 |
Arc melting |
36.6 |
21.2 |
[86] |
||
|
Mg0.9Ti0.1 + 5% C |
Ball milling |
88 |
Abs: 6.43% (after milling) |
[62] |
||
|
MgH2-6% NiTiO3 |
Ball milling |
74 ± 4 |
[87] |
|||
|
MgH2-6% CoTiO3 |
Ball milling |
100 ± 2 |
||||
|
MgH2-10 mol% TiH2-6 mol% TiO2 |
Ball milling |
118 |
[88] |
|||
|
MgH2-5% VTi-CNTs |
Ball milling |
Abs: 5.10%/150 °C/2 MPa/5 min |
10.2 |
[83] |
||
|
MgH2-5% FeTi-CNTs |
Ball milling |
Abs: 0.60%/150 °C/2 MPa/5 min |
65.5 |
[83] |
||
|
MgH2-10% Ni-TiO2 |
Ball milling |
Des: 6.50%/265 °C/0.02 bar/7 min |
43.7 ± 1.5 |
Abs: 5.00%/100 °C/60 bar/7 min |
[76] |
|
|
MgH2-4% Ni-6% TiO2 |
Ball milling |
91.6 ± 8.5 |
[76] |
|||
|
MgH2-10% Co-TiO2 |
Ball milling |
Des: 6.20%/250 °C/0.02 bar/15 min |
77 |
Abs: 4.24%/100 °C/60 bar/10 min |
[89] |
|