1000/1000
Hot
Most Recent

Recent discoveries in the “omics” field and the growing focus on preventive health have opened new avenues for personalized nutrition (PN), which is becoming an important theme in the strategic plans of organizations that are active in healthcare, food, and nutrition research. PN holds great potential for individual health optimization, disease management, public health interventions, and product innovation. However, there are still multiple challenges to overcome before PN can be truly embraced by the public and healthcare stakeholders. The diagnosis and management of lactose intolerance (LI), a common condition with a strong inter-individual component, is explored as an interesting example for the potential role of these technologies and the challenges of PN. From the development of genetic and metabolomic LI diagnostic tests that can be carried out in the home, to advances in the understanding of LI pathology and individualized treatment optimization, PN in LI care has shown substantial progress. However, there are still many research gaps to address, including the understanding of epigenetic regulation of lactase expression and how lactose is metabolized by the gut microbiota, in order to achieve better LI detection and effective therapeutic interventions to reverse the potential health consequences of LI.
| Term | Definition |
|---|---|
| Lactose malabsorption | Failure to digest/absorb lactose due to primary or secondary lactase deficiency. |
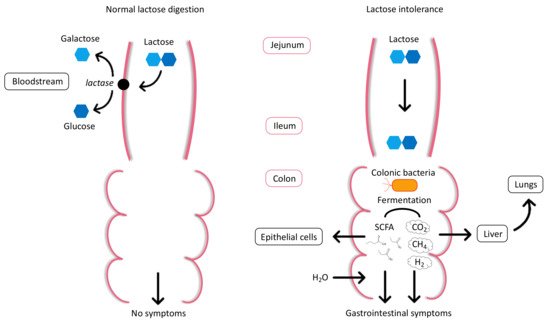
| Lactose intolerance (LI) | Clinical syndrome in which the ingestion of lactose causes typical gastrointestinal symptoms such as diarrhea, bloating, flatulence, nausea, abdominal pain, cramps. |
| Self-reported LI | Individuals who perceive themselves as being LI without medical diagnosis. |
| Lactase deficiency | Lack or absence of intestinal lactase enzyme activity. |
| Congenital lactase deficiency | Rare genetic disorder in which lactase is already absent at birth. |
| Primary lactase deficiency | Progressive decline of lactase enzyme activity with age. |
| Secondary lactase deficiency | Reversible condition caused by illness or injury of the small intestine and resulting in deficiency of intestinal lactase enzyme activity. |
| Lactase non-persistence (LNP) | Most common phenotype associated with lactase gene expression worldwide. Characterized by lactase activity decline during early childhood. |
| Lactase persistence (LP) | Phenotype expressed by the continued activity of the lactase enzyme throughout adulthood. |