
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Aránzazu Bocanegra | + 4261 word(s) | 4261 | 2021-05-14 11:03:20 | | | |
| 2 | Peter Tang | Meta information modification | 4261 | 2021-05-19 05:04:59 | | |
Video Upload Options
Type-2 diabetes mellitus (T2DM) is a major systemic disease which involves impaired pancreatic function and currently affects half a billion people worldwide. Diet is considered the cornerstone to reduce incidence and prevalence of this disease. Algae contains fiber, polyphenols, ω-3 PUFAs, and bioactive molecules with potential antidiabetic activity.
1. Introduction
2. Type-2 Diabetes Mellitus
3. Algae
3.1. Algae Definition and Classification
|
Division/Common Name |
Specie |
Pigments |
Storage Product |
Cell Structural Wall |
Intercellular Mucilage |
|---|---|---|---|---|---|
|
Phaeophyta/Brown algae |
Sargassum polycystum |
Chlorophyll a, c Fucoxanthins β-carotenes Xanthophylls |
Laminarans Mannitol |
Cellulose, Chitin |
Alginic acid/Alginates, Sulfated polysaccharides (Fucoidans) |
|
Himanthalia elongata |
|||||
|
Undaria pinnatifida |
|||||
|
Laminaria spp. |
|||||
|
Laminaria japónica, L digitata |
|||||
|
Hizikia fusiforme |
|||||
|
Chlorophyta/Green algae |
Ulva spp. |
Chlorophyll a, b Xanthophylls |
Starch |
Cellulose, Xylans, Mannans |
Sulfated polysaccharides |
|
Ulva lactuta |
|||||
|
Ulva pertusa |
|||||
|
Enteromorpha spp. (E. compresa) |
|||||
|
Rhodophyta/Red algae |
Pyropia spp. |
Phycoerythrin Phycocyanin Chlorophyll a β-carotene Xanthophylls |
Florideans Starch |
Cellulose, Xylans, Mannans |
Sulfated polysaccharides (Agar, Carrageenans, Porphyrans) |
|
Pyropia tenera |
|||||
|
Pyropia yezoensis |
|||||
|
Chondrus crispu |
|||||
|
Gracilaria verrucosa |
3.2. Algal Consumption and Commercial Importance
3.3. Algae as Functional Foods or as a Potential Raw Material for Bioactive Ingredients
4. Algal Composition, Structure, and Beneficial Effects on Type-2 Diabetes Mellitus
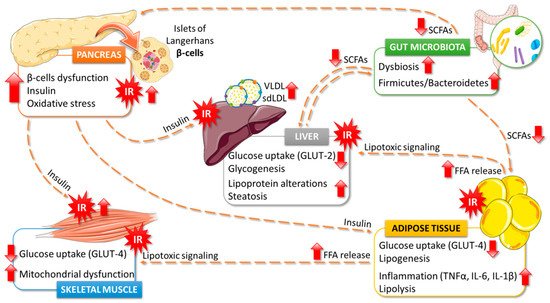
5. Effects of Algae Consumption on T2DM Pathophysiology
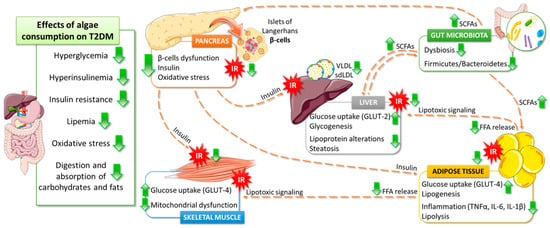
5.1. Glucose Homeostasis
5.2. Lipids Metabolism
5.3. Gut Microbiota
5.4. Antioxidant Properties
References
- World Health Organization. Global Report on Diabetes. 2016. Available online: (accessed on 10 January 2021).
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 1–11.
- Laakso, M. Biomarkers for type 2 diabetes. Mol. Metab. 2019, 27, S139–S146.
- Stančáková, A.; Laakso, M. Genetics of type 2 diabetes. Endocr. Dev. 2016, 31, 203–220.
- Sharma, S.; Tripathi, P. Gut microbiome and type 2 diabetes: Where we are and where to go? J. Nutr. Biochem. 2019, 63, 101–108.
- PR Newswire. Transparency Market Research. Available online: (accessed on 12 January 2021).
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24.
- Hannon, B.A.; Fairfield, W.D.; Adams, B.; Kyle, T.; Crow, M.; Thomas, D.M. Use and abuse of dietary supplements in persons with diabetes. Nutr. Diabetes 2020, 10, 14.
- Jiménez-Escrig, A.; Goñi, I. Nutritional evaluation and physiological effects of edible seaweeds. Arch. Latinoam. Nutr. 1999, 49, 114–120.
- Bocanegra, A.; Bastida, S.; Benedí, J.; Ródenas, S.; Sánchez-Muniz, F.J. Characteristics and nutritional and cardiovascular-health properties of seaweeds. J. Med. Food 2009, 12, 236–258.
- Brown, E.M.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.R.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216.
- Xu, S.-Y.; Huang, X.; Cheong, K.-L. Recent advances in marine algae polysaccharides: Isolation, structure, and activities. Mar. Drugs 2017, 15, 388.
- Peña-Romero, A.C.; Navas-Carrillo, D.; Marín, F.; Orenes-Piñero, E. The future of nutrition: Nutrigenomics and nutrigenetics in obesity and cardiovascular diseases. Crit. Rev. Food Sci. Nutr. 2018, 58, 3030–3041.
- Mathers, J.C. Nutrigenomics in the modern era. Proc. Nutr. Soc. 2017, 76, 265–275.
- Tiffon, C. The impact of nutrition and environmental epigenetics on human health and disease. Int. J. Mol. Sci. 2018, 19, 3425.
- Amar, J.; Chabo, C.; Waget, A.; Klopp, P.; Vachoux, C.; Bermúdez-Humarán, L.G.; Smirnova, N.; Bergé, M.; Sulpice, T.; Lahtinen, S.; et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: Molecular mechanisms and probiotic treatment. EMBO Mol. Med. 2011, 3, 559–572.
- Kolodziejczyk, A.A.; Zheng, D.; Shibolet, O.; Elinav, E. The role of the microbiome in NAFLD and NASH. EMBO Mol. Med. 2019, 11, e9302.
- Barbier, M.; Charrier, B.; Araujo, R.; Holdt, S.L.; Jacquemin, B.; Rebours, C. PEGASUS—Phycomorph European Guidelines for a Sustainable Aquaculture of Seaweeds; Barbier, M., Charrier, B., Eds.; COST Action FA1406: Roscoff, France, 2019.
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597.
- Macho-González, A.; Bastida, S.; Garcimartín, A.; López-Oliva, M.E.; González, P.; Benedí, J.; González-Muñoz, M.J.; Sánchez-Muniz, F.J. Functional meat products as oxidative stress modulators: A review. Adv. Nutr. 2021, nmaa182.
- Mirmiran, P.; Bahadoran, Z.; Azizi, F. Functional foods-based diet as a novel dietary approach for management of type 2 diabetes and its complications: A review. World J. Diabetes 2014, 5, 267–281.
- Schultz, A.R.; Garcimartín, A.; Bastida, S.; Jiménez-Escrig, A.; Rupérez, P.; Green, B.D.; Rafferty, E.; Sánchez-Muniz, F.J.; Benedí, J. Effects of Undaria pinnatifida, Himanthalia elongata and Porphyra umbilicalis extracts on in vitro α-glucosidase activity and glucose diffusion. Nutr. Hosp. 2014, 29, 1434–1446.
- Garcimartín, A.; Benedi, J.; Bastida, S.; Sánchez-Muniz, F.J. Aqueous extracts and suspensions of restructured pork formulated with Undaria pinnatifida, Himanthalia elongata and Porphyra umbilicalis distinctly affect the in vitro α-glucosidase activity and glucose diffusion. LWT Food Sci. Technol. 2015, 64, 720–726.
- Ródenas de la Rocha, S.; Sánchez-Muniz, F.; Gómez-Juaristi, M.; Larrea-Marín, M. Trace elements determination in edible seaweeds by an optimized and validated ICP-MS method. J. Food Compos. Anal. 2009, 22, 330–336.
- Larrea-Marín, M.; Pomares-Alfonso, M.; Gómez-Juaristi, M.; Sánchez-Muniz, F.; de la Rocha, S.R. Validation of an ICP-OES method for macro and trace element determination in Laminaria and Porphyra seaweeds from four different countries. J. Food Compos. Anal. 2010, 23, 814–820.
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329.
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013, 37, S81–S90.
- Schlienger, J.-L. Complications du diabète de type 2. Presse Med. 2013, 42, 839–848.
- Macho-González, A.; López-Oliva, M.E.; Merino, J.J.; García-Fernández, R.A.; Garcimartín, A.; Redondo-Castillejo, R.; Bastida, S.; Sánchez-Muniz, F.J.; Benedí, J. Carob fruit extract-enriched meat improves pancreatic beta-cell dysfunction, hepatic insulin signaling and lipogenesis in late-stage type 2 diabetes mellitus model. J. Nutr. Biochem. 2020, 84, 108461.
- Beale, E.G. Insulin signaling and insulin resistance. J. Investig. Med. 2013, 61, 11–14.
- Sánchez-Muniz, F.J.; García-Quismondo, A.; del Cañizo, F.J.; Dorado, J. Classical and emergent cardiovascular disease risk factors in type 2 diabetics from the Vallecas area (DICARIVA study). Nutr. Hosp. 2017, 34, 1432–1441.
- Bays, H.E. “Sick Fat,” metabolic disease, and atherosclerosis. Am. J. Med. 2009, 122, S26–S37.
- Carmena, R. Dyslipemia in type 2 diabetes mellitus. In Type 2 Diabetes Mellitus; Serrano, M., Gutiérrez, J.A., Eds.; Elsevier España S.L.: Barcelona, Spain, 2010; pp. 219–230.
- Chehade, J.M.; Gladysz, M.; Mooradian, A.D. Dyslipidemia in type 2 diabetes: Prevalence, pathophysiology, and management. Drugs 2013, 73, 327–339.
- Athyros, V.G.; Doumas, M.; Imprialos, K.P.; Stavropoulos, K.; Georgianou, E.; Katsimardou, A.; Karagiannis, A. Diabetes and lipid metabolism. Hormones 2018, 17, 61–67.
- Tangvarasittichai, S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J Diabetes. 2015, 6, 456–480.
- Qin, H.; Chen, Z.; Zhang, Y.; Wang, L.; Ouyang, P.; Cheng, L.; Zhang, Y. Triglyceride to high-density lipoprotein cholesterol ratio is associated with incident diabetes in men: A retrospective study of Chinese individuals. J. Diabetes Investig. 2020, 11, 192–198.
- Chen, Z.; Hu, H.; Chen, M.; Luo, X.; Yao, W.; Liang, Q.; Yang, F.; Wang, X. Association of Triglyceride to high-density lipoprotein cholesterol ratio and incident of diabetes mellitus: A secondary retrospective analysis based on a Chinese cohort study. Lipids Health Dis. 2020, 19, 1–11.
- Ojo, O. Dietary intake and type 2 diabetes. Nutrients 2019, 11, 2177.
- Burgio, E.; Lopomo, A.; Migliore, L. Obesity and diabetes: From genetics to epigenetics. Mol. Biol. Rep. 2014, 42, 799–818.
- Gesteiro, E.; Bastida, S.; Vázquez-Velasco, M.; Corella, D.; Guillén, M.; Ordovas, J.M.; Sánchez-Muniz, F.J. Effects of APOA5 S19W polymorphism on growth, insulin sensitivity and lipoproteins in normoweight neonates. Eur. J. Pediatr. 2011, 170, 1551–1558.
- Gesteiro, E.; Sánchez-Muniz, F.J.; Ortega-Azorín, C.; Guillén, M.; Corella, D.; Bastida, S. Maternal and neonatal FTO rs9939609 polymorphism affect insulin sensitivity markers and lipoprotein profile at birth in appropriate-for-gestational-age term neonates. J. Physiol. Biochem. 2016, 72, 169–181.
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional Foods and Lifestyle Approaches for Diabetes Prevention and Management. Nutrients 2017, 9, 1310.
- Wehr, J.D.; Sheath, R.G.; Kociolek, J.P. Freshwater Algae of North America. Ecology and Classification, 2nd ed.; Academia Press: San Diego, CA, USA; Elsevier: Oxford, UK, 2015; A volume in Aquatic Ecology; ISBN 9780123858771.
- Ścieszka, S.; Klewicka, E. Algae in food: A general review. Crit. Rev. Food Sci. Nutr. 2019, 59, 3538–3547.
- Tiwari, B.K.; Troy, D.J. Seaweed Sustainability. Food and Non-Food Applications, 1st ed.; Academic Press: Waltham, MA, USA; Elsevier: Oxford, UK, 2015; ISBN 978-0-12-418697-2.
- Van den Hoek, C.; Mann, D.G.; Jahns, H.M. Algae: An Introduction to Phycology; Cambridge University Press: Cambridge, UK, 1995.
- Ferdouse, F.; Løvstad-Holdt, S.; Smith, R.; Murúa, P.; Yang, Z. Food and Agriculture Organization of the United Nations. Available online: (accessed on 25 January 2021).
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. Appl. Phycol. 2017, 29, 949–982.
- EUMOFA. Observatorio Europeo del Mercado de los Productos de la Pesca y de la Acuicultura. Available online: (accessed on 25 January 2021).
- Tseng, C.K. Commercial cultivation. In The Biology of Seaweeds; Lobban, C.S., Wynne, M.J., Eds.; University of California Press: Berkeley, CA, USA; Blackwell Science Publications: Oxford, UK, 1981; Volume 17, pp. 680–725.
- Afonso, N.C.; Catarino, M.D.; Silva, A.M.S.; Cardoso, S.M. Brown Macroalgae as Valuable Food Ingredients. Antioxidants 2019, 8, 365.
- Food and Agriculture Organization of the United Nations (FAO). The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018; ISBN 978-92-5-130562-1.
- Hafting, J.T.; Critchley, A.T.; Cornish, M.L.; Hubley, S.A.; Archibald, A.F. On-land cultivation of functional seaweed products for human usage. J. Appl. Phycol. 2012, 24, 385–392.
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemodiversity and bioactivity: Sources of natural variability and implications for commercial application. Biotechnol. Adv. 2011, 29, 483–501.
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98.
- Villanueva-Peñacarrillo, M.L.; Martín-Duce, A.; Ramos-Álvarez, I.; Gutiérrez-Rojas, I.; Moreno, P.; Nuche-Berenguer, B.; Acitores, A.; Sancho, V.; Valverde, I.; González, N. Characteristic of GLP-1 effects on glucose metabolism in human skeletal muscle from obese patients. Regul. Pept. 2011, 168, 39–44.
- Cofrades, S.; Benedí, J.; Garcimartin, A.; Sánchez-Muniz, F.; Jimenez-Colmenero, F. A comprehensive approach to formulation of seaweed-enriched meat products: From technological development to assessment of healthy properties. Food Res. Int. 2017, 99, 1084–1094.
- Shang, Q.; Song, G.; Zhang, M.; Shi, J.; Xu, C.; Hao, J.; Li, G.; Yu, G. Dietary fucoidan improves metabolic syndrome in association with increased Akkermansia population in the gut microbiota of high-fat diet-fed mice. J. Funct. Foods 2017, 28, 138–146.
- Granado-Lorencio, F.; Hernández-Alvarez, E. Functional foods and health effects: A nutritional biochemistry perspective. Curr. Med. Chem. 2016, 23, 2929–2957.
- Imamura, F.; Micha, R.; Wu, J.H.; de Oliveira Otto, M.C.; Otite, F.O.; Abioye, A.I.; Mozaffarian, D. Effects of saturated fat, polyunsaturated fat, monounsaturated fat, and carbohydrate on glucose-insulin homeostasis: A systematic review and meta-analysis of randomised controlled feeding trials. PLoS Med. 2016, 13, e1002087.
- Shah, M.; Garg, A. The relationships between macronutrient and micronutrient intakes and type 2 diabetes mellitus in South Asians: A review. J. Diabetes Complicat. 2019, 33, 500–507.
- Galbete, C.; Schwingshackl, L.; Schwedhelm, C.; Boeing, H.; Schulze, M.B. Evaluating Mediterranean diet and risk of chronic disease in cohort studies: An umbrella review of meta-analyses. Eur. J. Epidemiol. 2018, 33, 909–931.
- The InterAct Consortium. Dietary fibre and incidence of type 2 diabetes in eight European countries: The EPIC-InterAct Study and a meta-analysis of prospective studies. Diabetologia 2015, 58, 1394–1408.
- Malik, V.S.; Li, Y.; Tobias, D.K.; Pan, A.; Hu, F.B. Dietary Protein Intake and Risk of Type 2 Diabetes in US Men and Women. Am. J. Epidemiol. 2016, 183, 715–728.
- Lee, K.W.; Woo, H.D.; Cho, M.J.; Park, J.K.; Kim, S.S. Identification of dietary patterns associated with incidence of hyperglycemia in middle-aged and older Korean adults. Nutrients 2019, 11, 1801.
- Lee, H.J.; Kim, H.C.; Vitek, L.; Nam, C.M. Algae consumption and risk of type 2 diabetes: Korean National Health and Nutrition Examination Survey in 2005. J. Nutr. Sci. Vitaminol. 2010, 56, 13–18.
- Oba, S.; Nanri, A.; Kurotani, K.; Goto, A.; Kato, M.; Mizoue, T.; Noda, M.; Inoue, M.; Tsugane, S.; Japan Public Health Center-based Prospective Study Group. Dietary glycemic index, glycemic load and incidence of type 2 diabetes in Japanese men and women: The Japan public health center-based prospective study. Nutr. J. 2013, 12, 165.
- Takahashi, K.; Kamada, C.; Yoshimura, H.; Okumura, R.; Iimuro, S.; Ohashi, Y.; Araki, A.; Umegaki, H.; Sakurai, T.; Yoshimura, Y.; et al. Effects of total and green vegetable intakes on glycated hemoglobin A1c and triglycerides in elderly patients with type 2 diabetes mellitus: The Japanese Elderly Intervention Trial. Geriatr. Gerontol. Int. 2012, 12, 50–58.
- Paradis, M.-E.; Couture, P.; Lamarche, B. A randomised crossover placebo-controlled trial investigating the effect of brown seaweed (Ascophyllum nodosum and Fucus vesiculosus) on postchallenge plasma glucose and insulin levels in men and women. Appl. Physiol. Nutr. Metab. 2011, 36, 913–919.
- Yoshinaga, K.; Mitamura, R. Effects of Undaria pinnatifida (Wakame) on postprandial glycemia and insulin levels in humans: A randomized crossover trial. Plant Foods Hum. Nutr. 2019, 74, 461–467.
- Jensen, M.G.; Pedersen, C.; Kristensen, M.; Frost, G.; Astrup, A. Review: Efficacy of alginate supplementation in relation to appetite regulation and metabolic risk factors: Evidence from animal and human studies. Obes. Rev. 2013, 14, 129–144.
- Tanemura, Y.; Yamanaka-Okumura, H.; Sakuma, M.; Nii, Y.; Taketani, Y.; Takeda, E. Effects of the intake of Undaria pinnatifida (Wakame) and its sporophylls (Mekabu) on postprandial glucose and insulin metabolism. J. Med Investig. 2014, 61, 291–297.
- Kim, K.-T.; Rioux, L.-E.; Turgeon, S.L. Alpha-amylase and alpha-glucosidase inhibition is differentially modulated by fucoidan obtained from Fucus vesiculosus and Ascophyllum nodosum. Phytochemistry 2014, 98, 27–33.
- Murray, M.; Dordevic, A.L.; Bonham, M.P.; Ryan, L. Do marine algal polyphenols have antidiabetic, antihyperlipidemic or anti-inflammatory effects in humans? A systematic review. Crit. Rev. Food Sci. Nutr. 2017, 58, 2039–2054.
- Seung-Hong, L.; You-Jin, J. Anti-diabetic effects of brown algae derived phlorotannins, marine polyphenols through diverse mechanisms. Fitoterapia 2013, 86, 129–136.
- Hall, A.; Fairclough, A.; Mahadevan, K.; Paxman, J. Ascophyllum nodosum enriched bread reduces subsequent energy intake with no effect on post-prandial glucose and cholesterol in healthy, overweight males. A pilot study. Appetite 2012, 58, 379–386.
- Nesto, R.W. Beyond low-density lipoprotein: Addressing the atherogenic lipid triad in type 2 diabetes mellitus and the metabolic syndrome. Am. J. Cardiovasc. Drugs 2005, 5, 379–387.
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 122.
- Sánchez-Muniz, F.J. IV y V Curso Avanzado en Obesidad y Síndrome Metabólico. Monografía 46; Instituto de España; Real Academia Nacional de Farmacia: Madrid, Spain, 2018.
- Bocanegra, A.; Bastida, S.; Benedí, J.; Nus, M.; Sánchez-Montero, J.M.; Sánchez-Muniz, F.J. Effect of seaweed and cholesterol-enriched diets on postprandial lipoproteinaemia in rats. Br. J. Nutr. 2009, 102, 1728–1739.
- Bermano, G.; Stoyanova, T.; Hennequart, F.; Wainwright, C.L. Seaweed-derived bioactives as potential energy regulators in obesity and type 2 diabetes. Adv. Pharmacol. 2020, 87, 205–256.
- Cherry, P.; Yadav, S.; Strain, C.R.; Allsopp, P.J.; McSorley, E.M.; Ross, R.P.; Stanton, C. Prebiotics from seaweeds: An ocean of opportunity? Mar. Drugs 2019, 17, 327.
- O’Sullivan, L.; Murphy, B.; McLoughlin, P.; Duggan, P.; Lawlor, P.G.; Hughes, H.; Gardiner, G.E. Prebiotics from marine macroalgae for human and animal health applications. Mar. Drugs 2010, 8, 2038–2064.
- Lopez-Santamarina, A.; Miranda, J.M.; Mondragon, A.D.C.; Lamas, A.; Cardelle-Cobas, A.; Franco, C.M.; Cepeda, A. Potential use of marine seaweeds as prebiotics: A review. Molecules 2020, 25, 1004.
- Faure, P. Protective effects of antioxidant micronutrients (vitamin E, Zinc and Selenium) in type 2 diabetes mellitus. Clin. Chem. Lab. Med. 2003, 41, 995–998.
- Sánchez-Muniz, F.J.; Bocanegra, A.; Bastida, S.; Benedí, J. Algae and cardiovascular health. In Functional Ingredients from Algae for Foods and Nutraceuticals, 1st ed.; Domínguez, H., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2013; pp. 369–415.




