Emerging studies suggested that the S-(-)-mitotane is more potent than the R-(+)-mitotane for AdrenoCortical Carcinoma (ACC) treatment. Currently, mitotane is the only FDA-approved drug . Therefore, we suggest that the production of pure and active S-(-)-mitotane might offer synergic or additive benefits for ACC patients, and even better if combined to solid lipid-based nanocarriers, and smart/advanced nanocarriers.
- Mitotane
- Enantiomers
- Adrenocortical Carcinoma
- Nanocarriers
- Nanomedicine
- Drug Targets
- Solid Cancers
- Cancer Therapy
Please note: Below is an entry draft based on your previous paper, which is wrirren tightly around the entry title. Since it may not be very comprehensive, we kindly invite you to modify it (both title and content can be replaced) according to your extensive expertise. We believe this entry would be beneficial to generate more views for your work. In addition, no worry about the entry format, we will correct it and add references after the entry is online (you can also send a word file to us, and we will help you with submitting).
Definition
Adrenocortical carcinoma (ACC) is a rare but aggressive malignancy with a poor prognosis. Treatment options for advanced ACC are limited. Indeed, radical tumor resection can lead to local or metastatic recurrence, and mitotane (Lysodren (R)), the only recognized adrenolytic drug, offers modest response rates, notably due to some of its physico-chemical and pharmacological properties (i.e. hydrophobicity, low bioavailability). Meantime, high cumulative doses of Lysodren (R) usually cause systemic toxicities. To reduce adverse health effects, the search of safe and efficient mitotane nano-formulations as well as the full characterization and testing of its enantiomers can represent valuable therapeutic options. Interestingly, recent investigations showed that solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) could considerably improve the efficacy of mitotane (i.e. enhanced solubility and bioavailability, progressive release of the loaded drug into blood and targeted tissues) as well as its safety (i.e. lower toxicity, higher biocompatibility). These two nano-carriers for mitotane delivery and targeting are of particular interest over other polymeric particles (i.e. low-cost, efficient and simple scaling to an industrial production level following green methods). Besides, emerging studies suggested that the S-(-)-mitotane is more potent than the R-(+)-mitotane for ACC treatment. Therefore, the production of pure and active S-(-)-mitotane might offer synergic or additive benefits for ACC patients when combined to solid lipid-based nanocarriers.
1. Adreno-Cortical Carcinoma: an Update
1.1. Epidemiology and Etiology
In contrast to benign adreno-cortical adenomas (ACA) that occur in at least 3% of the population aged over 50 years, adrenocortical carcinoma (ACC) is a rare endocrine malignancy [1-3]. Indeed, the worldwide incidence of this orphan disease is estimated to be around 0.5-2 cases per million/year and its prevalence about 4-12 cases per million population [1-3]. Exceptionally, in some regions of the world such as southern Brazil, the annual incidence in the children population - under 15 years old - is about 3.4-4.2 cases per million, which is considerable comparatively to the worldwide incidence [4]. A bimodal age distribution has been observed in ACC patients with peaks in childhood, before the age of 5, and in the fourth to fifth decades of life [5, 6]. Moreover, women are slightly more predisposed to the disease than men (ratio 1.5) [2, 7, 8].
The majority of ACCs are sporadic neoplasms of undetermined etiology while familial predisposition can occur. Interestingly, somatic mutations in genes predisposing to some syndromes associated with the increased susceptibility of cancer (e.g. Li-Fraumeni, Beckwith-Wiedemann), have also been identified in either benign or malignant sporadic adreno-cortical tumors (ACTs) [9]. Thereby, inactivating mutations at the 17p13 locus including the TP53 - protein considered as the “guardian of the genome” - [10], as well as alterations of the 11p15 locus leading to IGF-II (type II Insulin Growth Factor) over-expression and adrenal cancer cell proliferation, were frequently observed [11].
1.2. Prognosis, Diagnosis and Therapy
ACC is characterized by a poor prognosis and a high risk of recurrence post-therapy. Indeed, the recurrence is about 49% after adjuvant chemotherapy and up to 85% after surgery without adjuvant treatment [12, 13]. In about 40% of the cases, the disease relapse is manifested by the development of metastatic disease to the lungs, liver or bone at the diagnosis or within 6-24 months of surgical resection [7, 8, 12-14]. Moreover, the unsatisfactory overall 5-year survival ranges between 23% and 60% [2, 6, 8, 15]. Three main prognostic parameters are significantly associated with a shorter patient´s survival: (i) older age at diagnosis; (ii) tumor stage at presentation (i.e. stage III aka involvement of local lymph nodes and, stage IV aka local organ invasion or distant metastases), which can slightly differs according to the staging system used (i.e. MacFarlane-Sullivan [16] or European Network for the Study of Adrenal Tumors (ENSAT) [17]); (iii) hypersecretion of cortisol, a major adrenal steroid hormone [18].
At present, early diagnosis of this aggressive malignancy is mainly monitored by cortisol levels, which are too high in 60% of patients - especially in children (about 90%) - frequently leading to Cushing´s syndrome with or without virilization [1, 2, 5].
Complete surgical resection of the tumor (i.e. ipsilateral adrenalectomy with or without nephrectomy and/or splenectomy) is the only therapy that has consistently shown to prolong patient´s survival, particularly if disease is detected at early stages (I and II) but this, usually concerns less than 60% of the patients [1, 19, 20]. Indeed, median survival in patients with unresectable tumors or incomplete tumor resection is usually less than 12 months (about 3 to 9 months) [14, 21, 22] whereas, after complete resection, the median survival is generally improved (13 to 28 months) as shown in retrospective studies [19, 23-25]. Radiotherapy is only indicated as palliative treatment for patients with bone (and brain) metastases [1], and international prospective randomized studies are still insufficient to evaluate its benefit in the treatment of unresectable disease [26]. Finally, chemotherapy mainly consists of using mitotane, the only Food and Drug Administration (FDA)-approved drug against ACC, and can be administrated as follows: (i) alone; (ii) as combined regimens (e.g. mitotane plus etoposide-doxorubicincisplatin (EDP/M)) preferably in patients with incomplete, not possible or not successful tumor resection; (iii) as adjuvant in patients with high risk of recurrence at presentation or at relapse [1, 20, 27].
Interestingly, adjuvant treatment (i.e. chemotherapy and/or radiotherapy after surgery) might significantly decrease the disease recurrence after surgery and, increase the overall survival of the patients [12, 13, 18, 28-31]. Nevertheless, the potential benefits of adjuvant treatment have not been confirmed in some other studies [32-34], most likely because of incomplete surgery and variable drug metabolism [1, 35, 36]. Besides, current available systemic therapies provided incomplete efficient responses (<50%) in cases of advanced ACC and, remain severely limited mainly because of the rarity of ACC disease that had hampered the ability to undertake international randomized clinical studies to identify the most effective first- and second-line cytotoxic regimens [37]. Consequently, in spite of its relative efficacy, mitotane drug therapy remains the cornerstone, mainly in metastatic stage [38, 39].
Hopefully, the two most recent international randomized clinical studies, FIRM-ACT (First International Randomized trial in locally advanced and Metastatic Adrenocortical Carcinoma Treatment) and, ADIUVO (an international prospective, randomized, open-label, and controlled phase III trial for patients with ACC after radical resection), endorsed by the ENSAT [40], will show interesting data. Briefly, FIRM-ACT aims to assess the efficacy of mitotane combined to other drugs (e.g. EDP/M) as first line treatment versus Streptozotocin plus mitotane (Sz/M), while ADIUVO consist to evaluate the efficacy of mitotane as adjuvant treatment versus observation in patients with ACC at low-intermediate risk of recurrence after radical resection [27, 37]. However, until results from all randomized clinical trials become available, healthcare professionals will be challenged by an uncertainty.
Alternatively, the better understanding of the molecular pathogenesis of ACC, such as IGF signaling pathway, might allow the design of promising therapeutic targets [20, 41].
Eventually, the rapid emergence of the nanotechnology and state-of-art chromatography systems, shall significantly contribute to the development of mitotane chiral nano-formulations, which might present greater therapeutic features than the free mitotane drug formulation (i.e. lower toxicity, significant efficacy to lower the disease progression, capability to enhance patient´s survival and patient´s quality of life). However, the development of generic mitotane formulations shall be avoided due to the Narrow Therapeutic Index (NTI), and inadequate glucocorticoids administration must be prevented in order to limit the risk of adverse effects.
2. Mitotane: Synthesis, Structure and Drug Properties
2.1. Synthesis Route
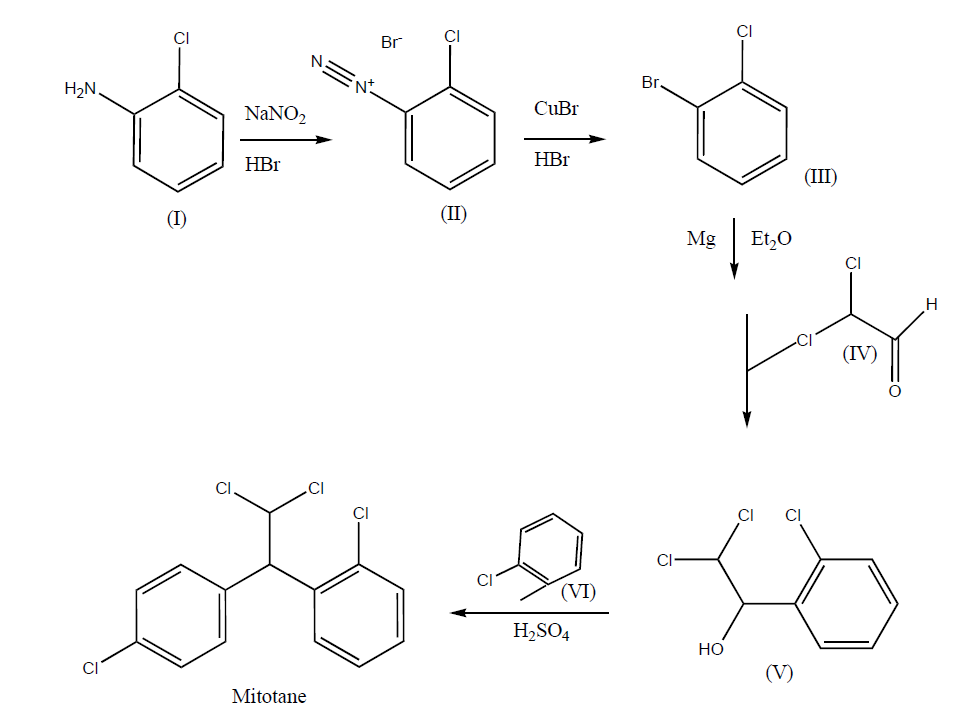
The synthesis route of mitotane (o,p´-DDD aka 1-(2-chlorophenyl)-1-(4-chlorophenyl)-2,2-dichloroethane, or 1-chloro-
2-[2,2-dichloro-1(4-chlorophenyl)ethyl]benzene, or 1,1-(o,p'-dichlorodiphenyl)-2,2-dichoroethane, or 1,1-dichloro-2-[o-chlorophenyl]-2-[p-chlorophenyl]ethane) has been more recently reviewed [42]. The manufacturing process is simply carried out in 5 steps Fig. 1 - three of them include chemical reactions and the other two correspond to recrystallizations -, and gas chromatography coupled to mass spectroscopy (GC-MS) is generally sufficient to characterize mitotane during this process or in the final product. Thereby, the classical synthesis route starts with the diazotisation of 2-chloroaniline (I) with NaNO2 and HBr in H2O that furnishes 2-chlorobenzenediazonium bromide (II), which is then brominated with CuBr and HBr to afford 1-bromo-2-chlorobenzene (III) [43]. 1-bromo-2-chlorobenzene (III) is treated with Mg in ether, and the obtained Grignard reagent is condensed with dichloroacetaldehyde (IV) in ether, yielding 2,2-dichloro-1-(2-chlorophenyl)ethanol (V), which finally is condensed with chlorobenzene (VI) in the presence of H2SO4 to provide mitotane [44].
Figure 1. Synthesis route of mitotane (Lysodren®; C14H10Cl4). Diazotisation of 2-chloroaniline (I) with Sodium nitrite (NaNO2) and hydrogen bromide (HBr) in water (H2O) furnishes 2-chlorobenzenediazonium bromide (II), which is then brominated with copper bromide (CuBr) and HBr to afford 1-bromo-2-chlorobenzene (III). Treatment of 1-bromo-2-chlorobenzene (III) with magnesium (Mg) in ether (ET2O), and condensing the obtained Grignard reagent with dichloroacetaldehyde (IV) in ether yielding 2,2-dichloro-1-(2-chlorophenyl)ethanol (V), which finally condenses with chlorobenzene (VI) in the presence of sulfuric acid (H2SO4) to provide mitotane. Adapted with permission from [42].
2.2. Structure and Physical-Chemical Properties
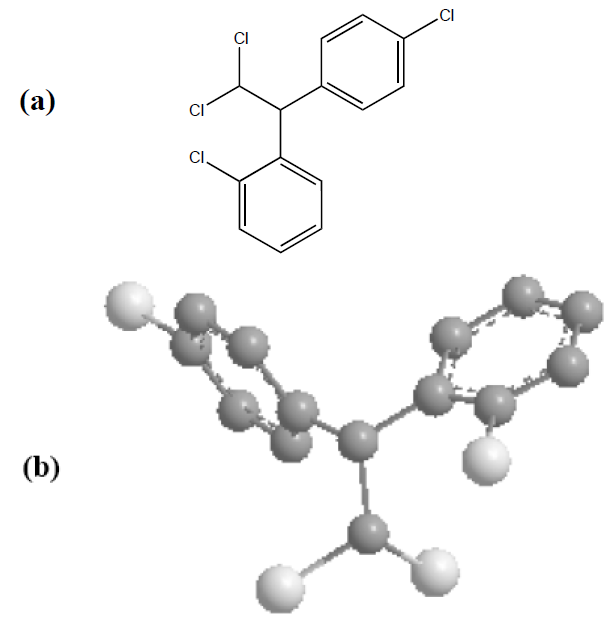
This oral antineoplastic agent is best known by its trivial name, o,p'-DDD. Its systematic chemical name, according to IUPAC (International Union of Pure and Applied Chemistry) nomenclature, is 1-chloro-2-[2,2-dichloro-1(4chlorophenyl)ethyl]benzene [45]. The 2D and 3D chemical structures of mitotane (C14H10Cl4) are shown in Fig. 2.
Figure 2. Chemical structures of mitotane (C14H10Cl4). (a) 2D structural view; (b) 3D structural view, where carbons from aromatic rings are in grey and chlorine atoms in white. In both cases (a) and (b), the hydrogen bonds have been omitted for clarity.
According to the US Pharmacopeia (USP) [46], Lysodren® must present the major following chemical-physical and pharmaceutical features: (i) a monoisotopic mass /molecular weight (MW) of about 318-320 Da; (ii) a melting point ranging between 75-81ºC; (iii) a dosing of 500 mg of the active substance mitotane; (iv) a white granular solid composed of clear colorless crystals; (v) a tasteless and slight pleasant aromatic odor; (vi) a low solubility in water, an acceptable solubility in either ethanol, ether, hexane, isooctane, carbon tetrachloride, fixed oils or fats; (vii) inactive ingredients represented by Avicel (matrix of microcrystalline cellulose), Polyethylene Glycol (PEG) 3350, colloidal silicon dioxide, and corn starch; (viii) an optimal stability when stored at 25°C (77°F), with excursions permitted to 15°C-30°C (59°F-86°F); (ix) absence of chromophores that absorb at wavelengths >290 nm, to avoid direct photolysis by sunlight; (x) an estimated usual half-life of 90 days, taking into consideration the possible atmosphere-degradation by a reaction involving photochemically-produced hydroxyl radicals.
These properties underlining the effect of the particle size and physical form on the dissolution, hence the bioavailability of the active substance – consequently need to be tightly controlled to ensure the clinical safety and efficacy of the medicinal product [46].
2.3. Biological Effects and Pharmacological Properties
Mitotane (Lysodren®), developed in 1960, is an isomer of DDD (dichloro-diphenyl-dichloro-ethane), a derivative of the pesticide DDT (dichloro-diphenyl-trichloro-ethane) which was shown to produce adrenal atrophy in dogs in 1948 and, represents up-to-date the only Food and Drug Administration (FDA)-approved drug for the treatment of ACC [47, 48]. Mitotane acts both as an inhibitor of steroidogenesis and an adrenolytic agent. Mechanistically, it inhibits directly 11β-hydroxylase, and cholesterol side-chain cleavage (SCC) in the mitochondria of steroidogenic cells together with antagonizing chemotherapy drug efflux, therefore blocking cortisol synthesis and reducing multidrug resistance (MDR), respectively [49]. Interestingly, mitotane has been shown to induce a p53-independent irreversible G2-arrested in cultured adrenocortical cell lines when combined to radiotherapy, as well as an increase of the radiotherapy cell growth inhibitory effect [50]. Furthermore, recent findings established a critical role of IGF signaling in ACC pathophysiology and provide rationale for use of targeted IGF-1R (type I Insulin-like Growth Factor Receptor) antagonists - especially when combined with mitotane - to treat ACC in future clinical trials [51].
Pharmacological analysis of oral Lysodren® in humans showed that about 40% only is absorbed, and approximately 10% of the administered dose is recovered in the urine as a water-soluble metabolite [52]. A variable amount of metabolite (1%-17%) is excreted in the bile within 24 hours and, because of its lipophilicity, the balance is apparently stored in the tissues (e.g. mainly adipose, liver, brain and adrenal tissues) [52]. Peak plasma Lysodren® concentrations occur 3-5 hours after a single oral dose of the drug and distribution of the drug between plasma and tissues is completed within 12 hours [52]. Consequently, cumulative high dose administration of mitotane is often required (up to 4-6 g/day during 3-5 months) which, subsequently, can lead to higher toxicity events [52, 53]. Following discontinuation of Lysodren®, the plasma terminal half-life has ranged from 18 to 159 days, but can even last longer in some tissues (e.g. storing tissues such as fat ones) [52]. It is not known whether Lysodren® or its metabolites are able to cross the placenta or distribute into milk. The NTI of mitotane anti-tumor activity is achieved at the plasma concentration of 14 mg/L [52, 54-56], and significant side effects have been noticed in more than 80% of all patients particularly when systemic levels of mitotane were greater than 20 mg/L [1, 57]. The adverse effects include the gastro-intestinal system (e.g. nausea, vomiting, diarrhea) or the central nervous system (e.g. lethargy, ataxia, depression), which can be reversible after cessation of mitotane [1, 57-60]. According to the World Health Organization (WHO) criteria, the overall response rate in 72 patients was 49%, including five patients with complete response [27]. This inter-patient variability, low response and considerable drug toxicity might be explained by many factors and mechanisms (e.g. genetics, epigenetics, ability of human tumor cells to efflux the drug or to metabolize it) and, underlines the importance of personalized medicine for mitotane dose titration as well as close clinical supervision.
In fact, mitotane metabolism is being studied for almost four decades to better understand its pharmacokinetics and pharmacodynamics and so, its molecular activity, which would help in carrying out the treatment [61-70]. Several approaches, using chromatography and/or spectrometry, have been developed to quantitatively determine mitotane and its metabolites in body fluids (e.g. serum, plasma, and urine) as well as in feces, and associate them with clinical outcomes [61, 66-70]. Among the major mitotane metabolites, we can cite o,p'-DDE (i.e. 1,1-(o,p'-dichlorodiphenyl)-2,2 dichloroethane aka 1,1-dichloro-2-[p-chlorophenyl]-2-[o-chloro phenyl]ethane) [61], and o,p'-DDA (1,1-(o,p'-dichlorodiphenyl) acetic acid) [62] which has been identified through a proposed route involving the adrenal mitochondrial cytochrome P450-catalyzed hydroxylation of mitotane at the β-carbon [63, 64]. Subsequent dehydrochlorination of the hydroxylated product forms the corresponding acyl chloride that, in the presence of water, formed the acidic metabolite, o,p'-DDA, although it could alternatively bind to tissue nucleophiles [65]. Interestingly, the synthesis route of β-3H-mitotane has been reported few years ago for use in a assay for mitotane metabolism [65], and consisted in the reduction of 1-(2-chlorophenyl)-1-(4- chlorophenyl)-2,2,2-trichloroethane (o,p´-DDT) by an aluminium-Hg2Cl2 couple in the presence of tritied water (3H20). Thereby, the determination of the 3H+, released to the aqueous media after organic solvent extraction, constituted a specific, faster (about 2-3 hours), and more sensitive assay for mitotane metabolic activation mediated by β-hydroxylation than 14Cmitotane- high-performance liquid chromatography (HPLC) [65]. Initial experiments in rats using 14C-labeled mitotane along with thin-layer chromatography (TLC), gas-liquid chromatography (GLC) and MS, reported that most of metabolites (87.8%) was concentrated in the feces (e.g. o,p'-DDA and its hydroxy-derivates such as 4-hydroxy-, 3-hydroxy-, and 3,4-dihydroxy-substituted o,p'-DDA, as well as o,p'-DDE) [66]. Interestingly, it was shown using GC-MS/Selected Ion Monitoring (SIM) that o,p'-DDA is present in the plasma at a concentration about 10 times higher than the levels of o,p'-DDD (mitotane) and o,p'-DDE [67]. This finding was later confirmed by other approaches using HPLC separation [61, 68]. The clinical significance of such high plasmatic o,p'-DDA levels is not established yet. Nevertheless, a relatively recent study that explored the relationship between the plasma levels of mitotane and its metabolites, o,p'-DDA and o,p'-DDE determined by HPLC, with the efficacy of mitotane therapy during a long-term follow-up of pediatric and adult patients with adrenal cancer, suggested that plasmatic o,p'-DDE concentrations could be more closely related to clinical improvement or remission than the mitotane levels [69]. Indeed, higher o,p’-DDE and o,p’-DDE/o,p’-DDD seemed to be associated with a good/favorable prognosis during the prolonged mitotane therapy [69], and so might consitute interesting/important factors in clinical practice.
3. Conclusions and Perspectives
Adrenocortical carcinoma (ACC) is a rare but aggressive malignancy with a poor prognosis. Owing to some of its physicalchemical and pharmacological properties (e.g. hydrophobicity, lability, and subsequent very low systemic bioavailability), mitotane (Lysodren®), the only FDA-approved adrenolytic drug offers modest response rates in ACC cancer patients while causing significant health side-effects when chronic doses are employed. Thus, the management of ACC patients, particularly those with advanced ACC, requires multidisciplinary and innovative approaches to overcome these current therapeutic limitations. In this manuscript, we highlighted a possible “two-in-one” solution to efficiently treat patients with ACC, based on the recent and emerging investigations that suggest a favorable use of: (i) nanostructured lipid carriers (NLC) to load mitotane with greater features compared to other polymeric particles (e.g. in terms of safety, efficacy and cost of production), and (ii) S-(-)-mitotane, considered to be more potent than R-(+)-mitotane (Lysodren®) in cancer patients. Therefore, pure active S-(-)-mitotane loaded into NLC might offer better clinical results than S-(-)-mitotane as a free drug and, synergic or additive beneficial health effects for a larg number of ACC patients comparatively to the use of R-(+)-mitotane loaded in the same experimental conditions into NLC. Eventually, more studies (e.g. in vivo, clinical, epidemiological ones) are needed to assess the pharmacological, physical and chemical properties as well as the risk/benefits ratio of such possible novel mitotane drug formulations (e.g. triggered release, long-term stability, safety/toxicity, efficacy) for the patients before tempting a large scale production that can be greatly conducted using “green” methods.
This entry is adapted from the peer-reviewed paper 10.2174/092986712804143376