High consumption of polyunsaturated fatty acids (PU FAs), specifically omega-3 FAs (Ω3FAs) such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), results in low plasma cholesterol levels and minimal coronary heart disease (CHD). Furthermore, as elevated triglycerides (TGs) appear to be a causal factor for atherosclerotic CVD (ASCVD) and possibly for premature all-cause mortality, more so when they are associated with genetic variants, PUFAs can reduce TG levels by decreasing lipoproteins with high amounts of TGs, such as very-low-density lipoproteins, intermediate-density lipoproteins, chylomicrons, and their remnants.
- omega-3 FA
- fatty acids
- cardiovascular disease
1. Dietary Sources and Guidelines
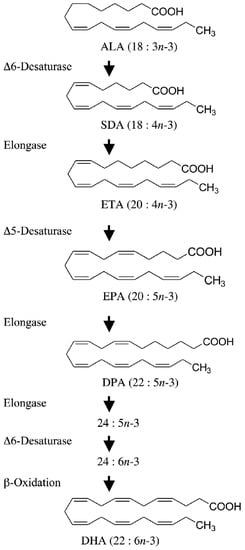
2. Molecular Mechanisms
3. Controversies Surrounding Ω3
4. Index
This entry is adapted from the peer-reviewed paper 10.3390/nu14235146
References
- U.S. Department of Agriculture, Agricultural Research Service. Food Data Central. 2019. Available online: Fdc.nal.usda.gov (accessed on 17 November 2022).
- Lavie, C.J.; Milani, R.V.; Mehra, M.R.; Ventura, H.O. Omega-3 polyunsaturated fatty acids and cardiovascular diseases. J. Am. Coll. Cardiol. 2009, 54, 585–594.
- O’Keefe, E.L.; Harris, W.S.; DiNicolantonio, J.J.; Elagizi, A.; Milani, R.V.; Lavie, C.J.; O’Keefe, J.H. Sea Change for Marine Omega-3s: Randomized Trials Show Fish Oil Reduces Cardiovascular Events. Mayo Clin. Proc. 2019, 94, 2524–2533.
- Fielding, B.A. Omega-3 index as a prognosis tool in cardiovascular disease. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 360–365.
- Thies, F.; Garry, J.M.C.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485.
- Erkkila, A.T.; Lehto, S.; Pyorala, K.; Uusitupa, M.I. n-3 Fatty acids and 5-y risks of death and cardiovascular disease events in patients with coronary artery disease. Am. J. Clin. Nutr. 2003, 78, 65–71.
- Rimm, E.B.; Appel, L.J.; Chiuve, S.E.; Djousse, L.; Engler, M.B.; Kris-Etherton, P.M.; Mozaffarian, D.; Siscovick, D.S.; Lichtenstein, A.H.; American Heart Association Nutrition Committee of the Council on Lifestyle and Cardiometabolic Health; et al. Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and Cardiovascular Disease: A Science Advisory from the American Heart Association. Circulation 2018, 138, e35–e47.
- Terry, A.L.; Herrick, K.A.; Afful, J.; Ahluwalia, N. Seafood Consumption in the United States, 2013–2016. NCHS Data Brief 2018, 1–8.
- Love, D.C.; Thorne-Lyman, A.L.; Conrad, Z.; Gephart, J.A.; Asche, F.; Godo-Solo, D.; McDowell, A.; Nussbaumer, E.M.; Bloem, M.W. Affordability influences nutritional quality of seafood consumption among income and race/ethnicity groups in the United States. Am. J. Clin. Nutr. 2022, 116, 415–425.
- Marmot, M.G.; Stansfeld, S.; Patel, C.; North, F.; Head, J.; White, I.; Brunner, E.; Feeney, A. Health inequalities among British civil servants: The Whitehall II study. Lancet 1991, 337, 1387–1393.
- Mackenbach, J.P.; Cavelaars, A.E.; Kunst, A.E.; Groenhof, F. Socioeconomic inequalities in cardiovascular disease mortality; an international study. Eur. Heart J. 2000, 21, 1141–1151.
- Manrique-Garcia, E.; Sidorchuk, A.; Hallqvist, J.; Moradi, T. Socioeconomic position and incidence of acute myocardial infarction: A meta-analysis. J. Epidemiol. Community Health 2011, 65, 301–309.
- Khaing, W.; Vallibhakara, S.A.; Attia, J.; McEvoy, M.; Thakkinstian, A. Effects of education and income on cardiovascular outcomes: A systematic review and meta-analysis. Eur. J. Prev. Cardiol. 2017, 24, 1032–1042.
- Addo, J.; Ayerbe, L.; Mohan, K.M.; Crichton, S.; Sheldenkar, A.; Chen, R.; Wolfe, C.D.; McKevitt, C. Socioeconomic status and stroke: An updated review. Stroke 2012, 43, 1186–1191.
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Alonso, A.; Beaton, A.Z.; Bittencourt, M.S.; Boehme, A.K.; Buxton, A.E.; Carson, A.P.; Commodore-Mensah, Y.; et al. Heart Disease and Stroke Statistics—2022 Update: A Report from the American Heart Association. Circulation 2022, 145, e153–e639.
- Harris, W.S.; Bulchandani, D. Why do omega-3 fatty acids lower serum triglycerides? Curr. Opin. Lipidol. 2006, 17, 387–393.
- Gaba, P.; Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Juliano, R.A.; Jiao, L.; Doyle, R.T., Jr.; et al. Prevention of Cardiovascular Events and Mortality with Icosapent Ethyl in Patients with Prior Myocardial Infarction. J. Am. Coll. Cardiol. 2022, 79, 1660–1671.
- De Caterina, R.; Massaro, M. Omega-3 fatty acids and the regulation of expression of endothelial pro-atherogenic and pro-inflammatory genes. J. Membr. Biol. 2005, 206, 103–116.
- Welty, F.K.; Alfaddagh, A.; Elajami, T.K. Targeting inflammation in metabolic syndrome. Transl. Res. 2016, 167, 257–280.
- Mason, R.P.; Libby, P.; Bhatt, D.L. Emerging Mechanisms of Cardiovascular Protection for the Omega-3 Fatty Acid Eicosapentaenoic Acid. Arter. Thromb. Vasc. Biol. 2020, 40, 1135–1147.
- Burr, M.L.; Gilbert, J.F.; Holliday, R.M.; Elwood, P.C.; Fehily, A.M.; Rogers, S.; Sweetnam, P.M.; Deadman, N.M. Effects of Changes in Fat, Fish, and Fibre Intakes on Death and Myocardial Reinfarction: Diet and Reinfarction Trial (Dart). Lancet 1989, 334, 757–761.
- Jialal, I.; Devaraj, S.; Huet, B.A.; Traber, M. GISSI-Prevenzione trial. Lancet 1999, 354, 1554.
- Kris-Etherton, P.; Eckel, R.H.; Howard, B.V.; St Jeor, S.; Bazzarre, T.L.; Nutrition Committee Population Science Committee; Clinical Science Committee of the American Heart Association. AHA Science Advisory: Lyon Diet Heart Study. Benefits of a Mediterranean-style, National Cholesterol Education Program/American Heart Association Step I Dietary Pattern on Cardiovascular Disease. Circulation 2001, 103, 1823–1825.
- Bhatt, D.L.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Steg, P.G.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. REDUCE-IT USA: Results From the 3146 Patients Randomized in the United States. Circulation 2020, 141, 367–375.
- Quispe, R.; Alfaddagh, A.; Kazzi, B.; Zghyer, F.; Marvel, F.A.; Blumenthal, R.S.; Sharma, G.; Martin, S.S. Controversies in the Use of Omega-3 Fatty Acids to Prevent Atherosclerosis. Curr. Atheroscler. Rep. 2022, 24, 571–581.
- Jacobson, T.A.; Glickstein, S.B.; Rowe, J.D.; Soni, P.N. Effects of eicosapentaenoic acid and docosahexaenoic acid on low-density lipoprotein cholesterol and other lipids: A review. J. Clin. Lipidol. 2012, 6, 5–18.
- Weintraub, H.S. Overview of prescription omega-3 fatty acid products for hypertriglyceridemia. Postgrad. Med. 2014, 126, 7–18.
- Singh, S.; Arora, R.R.; Singh, M.; Khosla, S. Eicosapentaenoic Acid Versus Docosahexaenoic Acid as Options for Vascular Risk Prevention: A Fish Story. Am. J. Ther. 2016, 23, e905–e910.
- Wei, M.Y.; Jacobson, T.A. Effects of eicosapentaenoic acid versus docosahexaenoic acid on serum lipids: A systematic review and meta-analysis. Curr. Atheroscler. Rep. 2011, 13, 474–483.
- Fialkow, J. Omega-3 Fatty Acid Formulations in Cardiovascular Disease: Dietary Supplements are Not Substitutes for Prescription Products. Am. J. Cardiovasc. Drugs 2016, 16, 229–239.
- Ramadeen, A.; Connelly, K.A.; Leong-Poi, H.; Hu, X.; Fujii, H.; Laurent, G.; Domenichiello, A.F.; Bazinet, R.P.; Dorian, P. Docosahexaenoic acid, but not eicosapentaenoic acid, supplementation reduces vulnerability to atrial fibrillation. Circ. Arrhythmia Electrophysiol. 2012, 5, 978–983.
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22.
- Albert, C.M.; Cook, N.R.; Pester, J.; Moorthy, M.V.; Ridge, C.; Danik, J.S.; Gencer, B.; Siddiqi, H.K.; Ng, C.; Gibson, H.; et al. Effect of Marine Omega-3 Fatty Acid and Vitamin D Supplementation on Incident Atrial Fibrillation: A Randomized Clinical Trial. JAMA 2021, 325, 1061–1073.
- Nicholls, S.J.; Lincoff, A.M.; Garcia, M.; Bash, D.; Ballantyne, C.M.; Barter, P.J.; Davidson, M.H.; Kastelein, J.J.P.; Koenig, W.; McGuire, D.K.; et al. Effect of High-Dose Omega-3 Fatty Acids vs Corn Oil on Major Adverse Cardiovascular Events in Patients at High Cardiovascular Risk: The STRENGTH Randomized Clinical Trial. JAMA 2020, 324, 2268–2280.
- Kalstad, A.A.; Myhre, P.L.; Laake, K.; Tveit, S.H.; Schmidt, E.B.; Smith, P.; Nilsen, D.W.T.; Tveit, A.; Fagerland, M.W.; Solheim, S.; et al. Effects of n-3 Fatty Acid Supplements in Elderly Patients after Myocardial Infarction: A Randomized, Controlled Trial. Circulation 2021, 143, 528–539.
- Kapoor, K.; Alfaddagh, A.; Al Rifai, M.; Bhatt, D.L.; Budoff, M.J.; Nasir, K.; Miller, M.; Welty, F.K.; McEvoy, J.W.; Dardari, Z.; et al. Association Between Omega-3 Fatty Acid Levels and Risk for Incident Major Bleeding Events and Atrial Fibrillation: MESA. J. Am. Heart Assoc. 2021, 10, e021431.
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098.
- Hu, Y.; Hu, F.B.; Manson, J.E. Marine Omega-3 Supplementation and Cardiovascular Disease: An Updated Meta-Analysis of 13 Randomized Controlled Trials Involving 127,477 Participants. J. Am. Heart Assoc. 2019, 8, e013543.
- Bernasconi, A.A.; Lavie, C.J.; Milani, R.V.; Laukkanen, J.A. Omega-3 Benefits Remain Strong Post-STRENGTH. Mayo Clin. Proc. 2021, 96, 1371–1372.
- Stillwell, W.; Wassall, S.R. Docosahexaenoic acid: Membrane properties of a unique fatty acid. Chem. Phys. Lipids 2003, 126, 1–27.
- Calder, P.C. n-3 fatty acids, inflammation and immunity: New mechanisms to explain old actions. Proc. Nutr. Soc. 2013, 72, 326–336.
- Harris, W.S.; Von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220.
