1. Vermicompost and Vermiwash
The VC is an organic fertilizer that is prepared through the conversion of organic wastes by worms [
40], and is rich in different enzymes including humic and fulvic acids [
41]. It contains a number of plant growth regulating substances (micro and macro elements, vitamins, enzymes, and hormones) and has anti-stress effects [
42,
43]. Earthworms in VC have beneficial effects on soil qualities such as physical, chemical, and biological properties [
44], as well as increasing plant development and production by making nutrients available to the plant [
45].
The VW and vermicompost leachate (VCL) are two important derivatives prepared from vermicompost. The VW is a clear, translucent, pale-yellow fluid obtained by passing water through a column of the vermi-worms’ excreta, which contains mucus secretions as well as micronutrients from decomposed organic sources [
46]. Khan et al. [
47] reported that VW has been utilized as an organic fertilizer for plants, and is a rich source of amino acids, vitamins, N, P, Mg, Zn, Fe, Cu, auxins and cytokinins. The VCL is a liquid that is collected when water drains over decomposing solids [
48]. This liquid may drain out from a traditional compost bin or a worm bin. Leachate is used as soil drench after dilution. VCL appears to be an effective and environmental friendly VC derivative for lowering salt’s harmful impact on bean seedlings [
49]. It was demonstrated that vermicompost promoted seed germination, root and shoot growth, proline accumulation, and oxidative stress management (
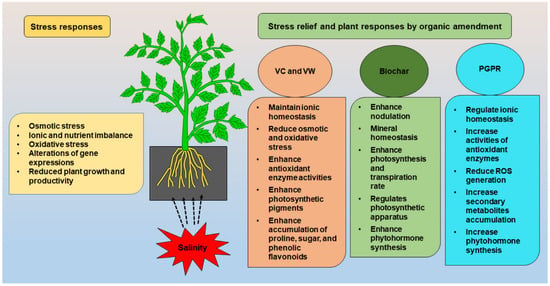
Figure 1, [
50,
51,
52]). In addition, VCL alleviates salt stress by enhancing photosynthetic efficiency, promoting antioxidant enzyme activity, and reducing electrolyte leakage [
42]. Among the various organic amendments practices, VC and VW are low-cost techniques to reduce the detrimental impact of salts on plants. The VC has been shown to reduce salt toxicity and enhance the emergence rate and seedling growth of different plants [
54,
55]. The VC enhances soil organic matter in salt soils by decreasing electrical conductivity (EC), bulk density and increasing field capacity, saturated hydraulic conductivity, and cation exchange capacity [
56].
Figure 1. Stress relief and possible plant responses by VC, VW, BC, and PGPR.
A number of studies show that VC enhanced salinity tolerance and improved the morphological characteristics such as stem and root length, fresh and dry weight of root-shoot, vigor index, leaf area, and dry weight per plant [
54,
57,
58,
59]. It has been reported that VC boosted Na
+ exclusion and K
+ accumulation, alleviated stomatal restriction, raised leaf pigment concentrations, improved root activity, decreased oxidative damage in Fountain Grass, and improved salt tolerance [
60]. In addition, VC application in maize and tomato plants under salt stress improved Chl a, Chl b, Total Chl, carotenoids, CAT, POD, and SOD and lowered H
2O
2 and MDA [
27,
61]. The addition of VC and VW to potato improved growth metrics, plant height, stem diameter, and tuber features, reducing the impact of salt stress [
35]. Liu et al. [
62] found that in coastal saline soil, VC application in maize increased nutrient availability and soil macro-aggregates by up to 91.02 percent. The soil amendment of VC increased exchangeable K
+, Ca
2+, and Mg
2+, plant height, total dry matter content, and decreased exchangeable Na
+ in the saline soil [
63,
64,
65]. In addition, VC application reduced salt-induced injuries of plants grown in saline soil by increasing relative water content, stomatal conductance, chlorophyll-a, superoxide dismutase (SOD), ascorbate peroxidase (APX), and catalase (CAT) activities and decreased electrolyte leakage and malondialdehyde (MDA) levels [
55,
66,
67,
68].
2. Biochar
BC is a carbon-rich organic substance with a porous structure, a wide surface area, and a high ion exchange ability that improves the physical qualities of agricultural soil [
83,
84]. A number of studies found that BC application improves the different physio-biochemical processes such as photosynthesis, hormonal and enzymatic activity in plants and decreases the harmful effects of salt stress on plants.
Morphological attributes such as seedling emergence, plant height, shoot biomass, root biomass, leaf area, dry matter, and yield of plants under salinity stress have been shown to be improved by BC incorporation [
32,
86,
87]. Moreover, BC application boosted photosynthetic rate, stomatal conductance, and transcription rate under salinity stress conditions in wheat [
84], sorghum [
87], quinoa [
83], and eggplant [
32]. On the other hand, the availability and uptake of different nutrients such as N, P, K in maize [
88] and P, K, Fe, Mn, Zn, and Cu in tomato [
89] improved by the utilization of BC as amendment to saline soil.
Furthermore, in saline conditions, BC traps excess Na
+ in soil, releasing mineral nutrients and decreasing osmotic stress [
86]. Studies showed that the use of BC lowered the concentration of Na
+ and decreased the Na
+/K
+ ratio in a variety of plants, assisting in the reduction of the negative effects of salt on plants [
84,
90]. Moreover, under salinity stress, BC application improves osmotic balance by increasing water holding capacity and CO
2 assimilation, which ultimately results in a better photosynthetic rate, stomatal conductance, and transcription rate [
32,
83,
86]. It has been reported that the leaf photosynthesis and net assimilation rate of the rice population was greatly aided by biochar’s potential positive effects on chlorophyll content, leaf N content, leaf area index, photosynthetic potential, stomatal conductance, and transpiration rates [
91,
92]. Additionally, the biochar treatment greatly enhanced the salt tolerance of cabbage seedlings and dramatically raised LRWC, Chl a, Chl b, and total Chl while reducing sucrose, proline, H
2O
2, and MDA [
29]. In addition, BC application reduced the effects of salt by lowering the levels of phytohormones such ABA, ACC, and JA, as well as increasing the amount of IAA in beans [
93]. Similarly, Nikpour-Rashidabad et al. [
94] reported that BC improved the vascular cylinder, parenchyma, IAA/ABA and IAA/ACC ratios to ameliorate the effects of salinity. Furthermore, under salt stress, the treatment with BC enhanced nodulation, nitrogen content, rubisco activity, GDH, GS, GOGAT, and NR activities in various parts of the soybean plant [
85]. Given the findings of the preceding investigations, BC appears to be a promising strategy for reducing salt stress and increasing plant growth and biomass in a variety of plants.
3. Bio-Fertilizer
BFs are one kind of fertilizer that contains living cells from various microorganisms and can transform via biological mechanisms; nutrients are converted from the inaccessible to the accessible form [
99,
100]. Recently, many studies have described the potential of BF in salt tolerance enhancement. The application of BF in wheat seedlings lessened the negative effects of salinity by increasing chlorophyll content and decreasing proline content, and improved plant growth and yield [
28,
37]. Under salt stress, amaranth enhanced plant height and biomass production [
101]. It has also been reported that the application of BF to lavender enhanced its capacity to withstand salt stress by increasing morphological attributes and RWC, Chl a, Chl b, and total Chl content as well as its essential oil output [
102]. Similarly, BF application on wheat (
Triticum aestivum L.), okra (
Abelmoschus esculentus L.), yellow passion fruit (
Passiflora edulis), cowpea (
Vigna unguiculata L.), corn (
Zea mays L.), and olive plants (
Olea europaea L.) enhanced growth and yield metrics, micro and macronutrient content, and relieved salinity-related detrimental effects [
28,
37,
38,
103,
104,
105,
106]. Souza et al. [
104] showed that in yellow passion fruit, BF application reduced the salt stress and enhanced the absolute growth rate, side branches, and yield. In addition, BF application to olive and papaya plants increased growth and plant biomass, improved osmotic adjustments between root and soil, increased microbial activity in the rhizosphere zone, and reduced the toxic effects of salts [
106,
107].
BF application increased antioxidant activity through the up-regulating of POX, SOD, and CAT, and reduced MDA and H
2O
2 production in lettuce (
Lactuca sativa L.), safflower (
Carthamus tinctorius L.) and cowpea (
Vigna unguiculata L.) [
105,
108,
109]. Al-Taey and Majid [
108] found that the functions of POD, CAT, SOD, and MDA were increased as a result of the increased salinity stress in lettuce (
Lactuca sativa L.). It has been reported that BF ameliorates the effects of salt stress via the production of phytohormones (IAA, CK, and ABA) and secondary metabolites (proline) in plants [
109,
110,
111].
Overall, the discussion concluded that BFs increased plant growth and production while also inducing salt tolerance by enhancing antioxidant enzyme activities, secondary metabolite accumulation and phytohormone synthesis.
4. PGPR
Plant-growth-promoting rhizobacteria (PGPR) are microorganisms that colonize plant roots and are used as chemical alternatives in agricultural fields for crop production and protection [
6,
118]. PGPR, which are resistant to salinity, help the plants to endure salty conditions. These plant-associated rhizobacteria can synthesize a variety of substances, including extracellular polymeric substance, 1-aminocyclopropane-1-carboxylate deaminase, phytohormones, antioxidants and volatile chemical compounds [
6,
30]. Gao et al. [
119] reported that rhizosphere bacteria reduce salt stress while promoting plant development by supplying nitrogen, phosphate, potassium, auxin, cytokinin, and abscisic acid to plants. During several field tests, crops grown under saline soil conditions responded favorably to the utilization of PGPR in terms of growth and yield. Kumawat et al. [
120] in his study revealed that PGPR increased seed germination, height of the plant, biomass, and chlorophyll contents under salt stress that ameliorate the negative effects of soil salinity. Water potential and stomatal opening is a crucial plant physiological activity for their survival which even salinity-stressed condition were found to be modified by PGPR to compensate salt stress [
121,
122]. For example,
Enterobacter cloacae, Pseudomonas fluorescence, Bacillus pumilus, and Exiguobacterium aurantiacum were found to greatly alleviate the toxic effect of salt stress in
Triticum aestivum plants [
122,
123]. Moreover, Ali et al. [
124] reported that under salt stress circumstances,
Enterobacter cloacae PM23 boosted maize growth, biomass, photosynthetic pigment contents, carotenoids, and relative water content compared to control treatment. Similar effects were observed when co-inoculation of
Rhizobium sp. and
Enterococcus mundtii in
Vigna radiata were carried out and obtained the grain production was improved under saline stress by regulating ion homeostasis [
120]. Additionally, when infected with
B. megaterium,
Solanum lycopersicum and
Arabidopsis thaliana both grew roots, shoots, and more leaves under salt stress [
125,
126]. Furthermore,
S. marcescens inoculation enhanced
Triticum aestivum shoot length, fresh weight, and chlorophyll (Chl) content [
127]. Under saline stress conditions, the
Enterobacter cloacae in
Brassica napus enhanced seedling development [
128]. Inoculating
Triticum aestivum with
Pseudomonas fluorescens led to similar outcomes, as did inoculating
Oryza sativa with
Alcaligenes faecalis,
B. pumilus, and
Ochrobactrum sp. [
129]. In addition, the application of some PGPR has been shown to improve nodule formation and fix nitrogen in plants under salt stress [
130]. For example,
Rhizobium sp. and
Bradyrhizobium japonicum’s co-inoculation improved root nodule formation in
Glycine max compared to control conditions, resulting in increased stress tolerance, plant growth, and higher yield [
130]. Likewise,
Bacillus aryabhattai and
Azotobacter vinelandii inoculation enhanced root nodule numbers and N-contents in
Trifolium repens compared with the non-inoculated plants [
131]. However, PGPR not only increased nodule numbers but also increased plant dry weight, shoot dry weight, the extent of nitrogen yield and protein content in some applications [
132].
Many studies described that PGPR can alleviate the salt-induced growth inhibition of plants by positively regulating ion homeostasis and antioxidant enzyme activity, improving photosynthetic attributes, secondary metabolite accumulation, and oxidative stress reduction. For instance, the use of PGPR reduced the negative effects of salinity in pea (
Pisum sativum) by enhancing the plants’ proline and soluble sugar contents while lowering sodium (Na
+) contents, which in turn reduced the amount of electrolyte leakage and H
2O
2 content [
135,
136]. In addition, the harmful effects of salinity are reduced by PGPR via declining lipid peroxidation and ROS in wheat plants [
137]. Singh et al. [
125] and Kumawat et al. [
120] reported that PGPR alters the selectivity of Na
+, K
+, and Ca
2+ under salt stress and thus maintains ionic balance due to ion homeostasis. Moreover, inoculating
Pseudomonas sp. or
Glutamicibacter sp. with the halophte
Suaeda fruticosa led to noticeably greater shoot dry weight and decreased buildup of Na
+ and Cl
− in shoots of salt-treated plants [
138]. Similarly, the
Piriformospora indica inoculation in
Zea mays decreased K
+ flow from roots while increasing K
+ concentration in shoots under saline condition; this effect may be linked to a high-affinity K
+ transporter where PGPR produced a proton-driven force through H
+-ATPase [
139]. Moreover,
Azotobacter isolates under salinity stress had greater K
+/Na
+ proportions in shoots and decreased Na
+ and Cl
− amounts in maize leaves [
140]. Additionally, some PGPR lowered Cl
2 and NO
2 concentrations and increased the K
+/Na
+ ratio, which contributed to enhanced stomatal conductance, and maintained hormonal balance and photosynthesis under salt stress [
6,
141]. It has also been reported that PGPR incorporation enhances the synthesis of the phytohormone that improves salt stress tolerance [
6]. Such as, Some PGPR i.e.,
Thalassobacillus denorans,
Oceanobacillus kapialis,
Pseudomonas strains,
Bacillus tequilensis and
Bacillus aryabhattai synthesized more auxin and ABA, accumulated osmolytes in cell cytoplasm that sustain their cell turgor to make ensure plant growth under osmotic stress in
Oryza sativa to endure high saline conditions [
142]. In addition to the mechanisms mentioned above, in order to survive under salt stress, PGPR may alter salt tolerant gene expressions. The expression of TaABARE, TaOPR1, TaMYB, TaWRKY, TaST, SOS1, SOS4, TaNHX1, TaHAK, and TaHKT1 genes were up-regulated in PGPR inoculated plants leading to the expression of stress related genes [
143,
144]. According to the findings, salinity tolerance genes ZmNHX1, ZmNHX2, ZmN HX3, ZmWRKY58, and ZmDREB2A were up-regulated, as well as the antioxidants ZmGR1 (Zea mays glutathione reductase) and ZmAPX1’s (Zea mays ascorbate peroxidase) transcript levels [
145,
146]. Moreover, salinity tolerance was increased when PGPR enhanced antioxidant enzymes’ gene expression such as CAT, POD, APX, MnSOD, GR and GPX in inoculated plants [
143]. Furthermore, according to Ali et al. [
124], the inoculation of maize with
Enterobacter cloacae PM23 increased APX, SOD, POD, total soluble sugars, and proteins while decreasing flavonoids and phenolic contents under salt stress. Additionally, in
Suaeda fruticosa under high salinity,
Glutamicibacter sp. inoculation dramatically decreased MDA levels while enhancing the activities of SOD, CAT, APX, and GR. Habib et al. [
147] reported that salinity circumstances in the okra plant led to greater synthesis of APX and CAT by
B. megaterium and
Enterobacter sp. It has also been found that the treatment of
Arabidopsis seedlings with
Enterobacter sp. increased APX function and boosted salt tolerance [
148]. Thus, from the reports of the studies it is apparent that the exogenous application PGPR could bring positive growth and yield results within the plants under saline condition and can be considered as a promising modern agronomic tactic to develop the plants survival under a saline environment. In future, dealing with extensive molecular research may reveal the efficacy of PGPR isolates and mechanisms to improve its stress responsive capability within the short duration in a wide area for sustainable agricultural production.
This entry is adapted from the peer-reviewed paper 10.3390/life12101632