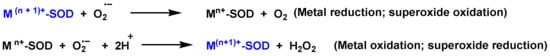2. Function and Features of Immunity and Innate Immune System
The immune system involves cells, organs, proteins, and tissues throughout the body, and it comprises components such as leukocytes, spleen, bone marrow, lymphatic system, thymus, tonsils, adenoids, and appendix [
31]. There are three types of immunity in humans: innate, adaptive, and passive.
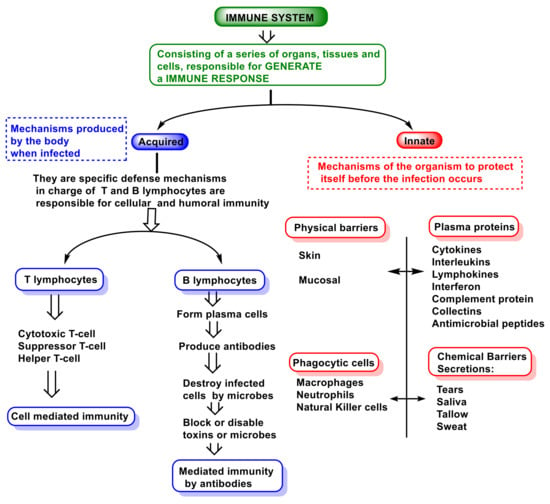
Figure 2 is a representation of the immune system.
Figure 2. Innate and adaptive immune responses.
Innate immunity is the immunity that people are born with and provides a certain level of immunity that attacks invaders from day one. This innate immunity is the first line of defense against pathogens and includes the skin and mucous membranes of the throat and gut. Innate is non-specific immunity and is an ancient evolutionary defense strategy found in plants, fungi, animals, and primitive multicellular organisms [
32].
Adaptive immunity involves specific immune cells and antibodies, and it can prevent disease in the future by remembering what those substances look like and mounting a new immune response, and is carried out by lymphocytes.
Vertebrates, exclusively, have adaptive immunity, which can recognize and destroy specific substances [
33]. The adaptive immune response provides vertebrates with the ability to recognize and remember specific pathogens, generating immunity and delivering increasingly potent responses to the re-encountered pathogen. Adaptative immunity includes two parts: one is called humoral and involves a variety of substances found in the body’s humors or fluids, which interfere with the growth of pathogens or clump them together so that they can be eliminated from the body.
The antibody and cell-mediated immune responses are carried out by different classes of lymphocytes, called B cells and T cells, respectively. B cells are activated to secrete antibodies, a type of protein called immunoglobulins, which circulate through the bloodstream and penetrate other body fluids, eventually binding specifically to the foreign antigen that stimulated their production. Antibody binding inactivates viruses and microbial toxins by blocking their ability to bind to receptors on host cells. Antibody binding also marks invading pathogens for destruction by making it easier for the phagocytes of the innate immune system to ingest them [
34]. The cell-mediated response is carried out by phagocytes, which ingest and degrade pathogens, as well as by natural killer cells that destroy certain cancer cells [
35].
In contrast, adaptive immunity is also responsible for allergic reactions and the rejection of transplanted tissues, which it recognizes as a foreign invader [
36].
Passive immunity is a type of temporary immunity that is derived from another person. For example, a newborn receives antibodies from the mother through the placenta before birth and in breast milk after birth. This passive immunity protects the baby from some infections during the first years of life.
Innate and adaptive systems work together to provide vertebrates with increased resistance to micro-organisms, parasites, and potential intruders that may cause harm.
In innate immunity, invaders are identified by pattern recognition receptors that distinguish molecules expressed on microbial surfaces, called pathogen-associated molecular patterns (PAMPs). A second trigger is molecules released from broken or damaged cells, called damage-associated molecular patterns (DAMPs) [
37].
Polymorphonuclear leukocytes PMNs recognize secreted molecules produced by bacteria, including peptidoglycan, lipoproteins, lipoteichoic acid, lipopolysaccharide (LPS), CpG-containing DNA, and flagellin. Peptidoglycan recognition protein (PGRP) plays a role in the neutrophil killing of Gram-positive bacteria [
38], inhibiting their growth.
A class of pathogen recognition receptors is toll-like receptors (TLRS), a family of at least 10 different receptors found on the surface or in the cytoplasm of cells such as macrophages, intestinal epithelial cells, and mast cells, and which are located on the surface or the membrane of endosomes [
39]. Toll receptors bind to PAMPs on extracellular bacteria, such as lipopolysaccharides, flagellin, and lipoproteins [
40]. Cytoplasmic TLRs bind to the nucleic acids of intracellular viruses [
41]. Once bound to these ligands, TLRs trigger the production of inflammatory cytokines such as interleukin IL-β1 or the tumor necrosis factor TNF-α, triggering what is termed acute inflammation [
42].
The presence of inflammatory chemokines controls the recruitment of effector leukocytes in infections, inflammation, tissue injury, and tumors, and has a broad cellular selectivity, acting on cells of both the innate and adaptive immune systems [
43]. In this process participates integrins and transmembrane cell adhesion molecules, which regulate cellular growth, proliferation, migration, cellular signaling, cytokine activation, and its release. Therefore, they play important roles in cell proliferation and migration, apoptosis, and tissue repair, as well as in all processes critical to inflammation, infection, and angiogenesis [
43,
44].
Acute inflammation is the central feature of innate immunity and it is the subsequent step in the early detection of invading organisms or damaged tissues [
45]. The inflammatory response is characterized by several features: reddening of the skin (due to increased blood circulation), warmth or increased temperature (sensation of heat around a local infection or systemic fever), swelling of affected tissues (in the throat during the common cold or in joints affected by rheumatoid arthritis), mucus production (runny nose or cough), pain (in sore joints or in the throat) and even possible dysfunction of affected organs and tissues [
1]. Inflammation guarantees that leukocytes converge in large numbers towards the site of microbial invasion, attracting these cells from the bloodstream and inducing them to migrate through the tissues to the invasion site [
46].
The key to an effective innate response is the rapid recognition of the invasion, for which there are several types of sentinel cells. The most important are macrophages, dendritic cells, and mast and innate lymphoid cells [
47]. The first three possess pattern recognition receptors and can detect the presence of PAMPs and DAMPs, so they send a signal through the nuclear factor NF-κβ, to produce cytokines such as IL-1, interferon IFN-α and TNF-α [
48]. Molecules such as histamine, leukotrienes, prostaglandin, and specialized peptides are released to initiate the inflammatory process. Three main populations of leukocytes can eliminate invaders: (i) neutrophils, which are particularly effective at killing invading bacteria by engulfing them, activating the respiratory burst, and generating lethal oxidative molecules such as hydrogen peroxide and hypochlorite ions, which kill most invading bacteria; (ii) eosinophils, specialized killers of invading parasites, which for example contain enzymes optimized to kill helminth larvae; and (iii) M1-like macrophages, capable of migrating to areas of microbial invasion more slowly than granulocytes, but able to maintain sustained and effective phagocytosis [
49]. They contain the lethal antimicrobial
•NO and can kill neutrophil-resistant organisms. If inflammation activates macrophages, they secrete a cytokine called IL-23, which acts on the Th17 cell subset, secreting IL-17, which attracts neutrophil granulocytes to sites of inflammation, infection, and tissue damage [
50].
Mammals possess at least four populations of innate lymphoid cells ILCs that participate in innate immunity: natural killer (NK) cells, ILC1s, ILC2s, ILC3s, and lymphoid tissue inducer cells [
51]. NK natural killer cells are innate lymphoid cells optimized to kill virus-infected cells, and can even kill abnormal cells, which do not express MHC class I major histocompatibility complex molecules [
52]. Group I of innate lymphoid cells are found in large numbers in the intestinal wall and secrete macrophage-activating cytokines, playing a key role in antiviral immunity [
53]. Group II of innate lymphoid cells are distributed throughout the body and secrete cytokines important for anti-parasitic immunity [
54]. Group III of innate lymphoid cells act as Th17 cells and promote inflammation by releasing IL-17 [
55]. Lymphoid tissue-inducing cells are hematopoietic and have critical roles in the immune system, both in the embryonic and adult stages [
56]. These cells fulfill the following four functions: defense against pathogens, surveillance of tumorigenesis, regulation of homeostasis, and tissue remodeling [
57].
When neutrophils arrive at the site of invasion, they bind to invading bacteria and ingest them by phagocytosis, a process mediated by a metabolic pathway called a respiratory burst, which generates oxidative species such as H
2O
2 and hypochlorous acid HOCl [
58]. In contrast, the energy reserves of neutrophils are minimal, and they can only perform a few phagocytic events before they decay. Once the invading microbes are successfully eliminated, the body must repair the damage and eliminate cellular debris and dying cells through the work of macrophages, which originate from monocytes in the blood [
59]. Macrophages are attracted to sites of microbial invasion and tissue damage by chemokines, DAMPs, and PAMPs, help kill invaders, remove toxic waste produced in tissues and destroy remaining neutrophils. M1-like macrophages complete the destructive process and are optimized for microbial destruction, while M2-like macrophages are optimized for the removal and repair of damaged tissues [
60].
Besides the role played by neutrophils, there is also a parallel mechanism called NETosis, related to the formation of neutrophil extracellular traps (NETs). Various pathogens, antibodies and immune complexes, cytokines, and other physiological stimuli can trigger NETosis. Its induction depends on ROS, the main source being NOx [
61]. NOx activation depends on increased Ca
2+ concentration in the cytoplasm and, in some cases, on the generation of mitochondrial ROS. NETosis results in the release of granule components into the cytosol, histone modification leading to chromatin decondensation, destruction of the nuclear envelope, as well as the formation of pores in the plasma membrane. Two forms of NETosis have now been described: classical or suicidal NETosis (leading to cell death), and vital NETosis, where the cell retains its viability and many of its effector functions [
62].
Classical NETosis is a special form of programmed cell death (PCD), characterized by the release of granule components into the cytosol. Several features of apoptosis, necroptosis, pyroptosis, autophagy, and secondary necrosis are inherent to this form of NETosis. Mitochondrial ROS are involved in NOx activation and in the induction of classical NETosis by various stimuli [
63,
64].
Vital NETosis helps contain local infections by allowing PMNs to rapidly release NETs and continue to phagocytose live bacteria. In addition, live PMNs that release NETs manage to maintain their membrane integrity, thereby imprisoning the captured bacteria [
65,
66].
Alongside ROS production, macrophages also employ several directly antimicrobial mechanisms, such as the generation of RNS in the phagosome, and the delivery of cathepsins and other hydrolases into maturing phagosomes [
67]. Other indirect antimicrobial mechanisms include: (i) activation of inflammasomes and (ii) secretion of cytokines and chemokines [
68]. These mechanisms help orchestrate subsequent innate and adaptive immune responses, as well as major histocompatibility complex MHC-dependent presentation of pathogen-derived antigens [
69].
In parallel with acute inflammation, the body has other innate defenses as tissues contain a variety of antimicrobial peptides. These include antimicrobial peptides, such as defensins or cathelicidins, enzymes such as lysozyme that kill many Gram-positive bacteria, and iron-binding proteins such as hepcidin or haptoglobin that prevent the growth of bacteria by depriving them of vital iron [
70]. The most important of these defenses is the complement system, a group of about 30 proteins that work together to eliminate invading microbes by covalently and irreversibly binding two proteins called C3 and C4 to microbial surfaces. Once bound, they can lyse microbes via the C5–C8 complex formation and the polymerization of C9 protein forming a membrane attack complex MAC, or participates as opsonins, promoting a quickly and efficiently phagocytosis by leukocytes [
71].
This system can be activated in three ways:
- (i)
-
The so-called alternative pathway is activated by the presence of bacterial surfaces that can bind complement protein C3. C3-coated bacteria are rapidly and efficiently phagocytosed and destroyed. C3 can activate other complement components by inducing a protein called C9 to insert itself into the cell walls of bacteria, causing them to rupture;
- (ii)
-
A second pathway of complement activation is triggered when bacterial surface carbohydrates bind to a mannose-binding lectin (MBL), collectin 11 (CL-K1), and ficolins (Ficolin-1, Ficolin-2, and Ficolin-3). Its activation leads to C4 and C2 activation by their serine-proteases; or
- (iii)
-
The classical complement pathway is initiated by antigen-antibody complexes with the antibody isotypes IgG and IgM. Upon activation, several proteins are recruited to generate C3 convertase, which cleaves the C3 protein. The C3b component of cleaved C3 binds to the C3 convertase to generate the C5 convertase, which cleaves the C5 protein. The cleaved products attract phagocytes to the site of infection and mark target cells for elimination by phagocytosis. C5 convertase initiates the terminal phase of the complement system, resulting in the assembly of the MAC membrane attack complex, creating a pore in the target cell membrane, and inducing its lysis [
72].
Because of its potential to cause severe tissue damage, the activation of the complement system is carefully controlled through multiple complex regulatory pathways [
73].
The complement system plays an important role in mediating tissue injury following the triggering of oxidative stress. Collard et al., 2000, investigated the role of mannose-binding lectin (MBL) and the lectin complement pathway (LCP) in mediating complement activation following endothelial oxidative stress and observed that the LCP lectin complement pathway mediates complement activation following tissue oxidative stress. Thus, they suggest that inhibition of MBL may represent a novel therapeutic strategy for ischaemia/reperfusion injury and other complement-mediated disease states [
74].
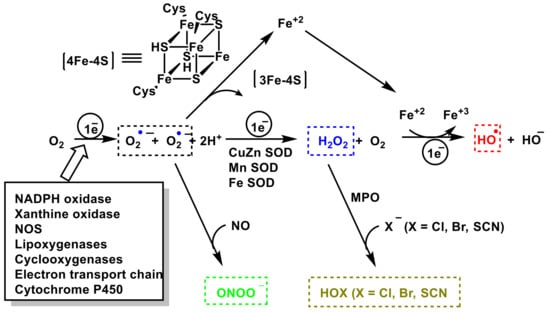
3. Role of Superoxide Anion •O2− and Hydrogen Peroxide H2O2 on Innate Immunity
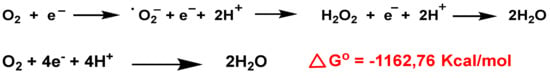
O
2− anion is a by-product of mitochondrial respiration and a crucial element of the innate immune defense system. Biochemically,
•O
2− is generated from two main sources: in the respiratory chain in the mitochondrial matrix and via nicotinamide adenine dinucleotide phosphate. In the electron transport chain, protons introduced by ATP synthase reduce molecular O
2 to
•O
2− anion, H
2O
2, and H
2O [
75],
Figure 3. Consecutive reduction of O
2 with H
+ and e
— have a negative Gibbs energy, so it occurs spontaneously, with a ∆Go ≤ 0. The Gibbs free energy is used to calculate the maximum amount of work that can be done by a thermodynamically closed system, with temperature and pressure being constant, and is a necessary condition in processes such as chemical reactions.
Figure 3. O2 reduction chain to •O2−, H2O2 and H2O.
In the cytosolic SOD-Cu/Zn (it contains Cu and Zn in the catalytic site), SOD transforms the •O2− to O2 reducing Cu(II) to Cu(I). Another •O2− molecule causes the oxidation of Cu(I) to Cu(II), producing an H2O2 molecule. Zn is monovalent and it only stabilizes the enzyme. The catalytic cycle of Mn SOD is similar, with Mn in the oxidation-reduction reactions, transiting between Mn(III)) and (Mn(II).
Operative roles of H
2O
2 during inflammation have been observed, modulating protein function by reversible chemical modification of protein thiols [
76]. H
2O
2 induces activation of factor nuclear NF-κβ (the factor that controls DNA transcription), including tyrosine phosphorylation of IkB and activation of IKK [
77]. H
2O
2 can trigger the release of high mobility group 1 protein from macrophages, follow-on increase of proinflammatory stimuli [
78].
Polymorphonuclear neutrophils PMN are a critical constituent of the innate immune system. In case of infection, neutrophils are rapidly recruited from the circulation and bone marrow stores by the host- and pathogen-derived components, priming these cells for enhanced antimicrobial activity [
79]. One of the most potent biochemical attractants is the interleukin IL-8, produced by cells during the inflammatory process associated with infection [
80]. Cells that produce interleukin IL-8 include monocytes, macrophages, mast cells, epithelial cells, keratinocytes, fibroblasts, endothelial cells, and even neutrophils themselves [
81]. Bacteria also produce molecules that can directly attract neutrophils, e.g., N-formyl peptides [
82]. Neutrophil “priming” is the ability to increase superoxide anion. In fact, this capacity is not limited to
•O
2− production, but also to improved adhesion, phagocytosis, cytokine secretion, leukotriene synthesis, degranulation, and, ultimately, bactericidal activity. In this “priming” effect, neutrophils respond to the release of cytokines, chemokines, growth factors, and lipid-derived signaling molecules. In summary, neutrophils react increasing the release of
•O
2− and inducing the expression of, among others, TNF-α, IFN-γ and -α, several interleukins, C2-ceramide, peroxynitrite, or diamide (thiol oxidizer) [
83].
The combination of ROS from neutrophils and granule components is usually effective in killing most bacteria and fungi. PMNs are the most abundant leukocyte in humans and contain a battery of non-specific cytotoxic compounds, so their homeostasis is highly structured. Once neutrophil apoptosis occurs, these cells are eliminated by macrophages, and their apoptosis is accelerated following phagocytosis of bacteria, completing the termination of the infection and associated inflammation [
84].
Ultimately, neutrophils use both O
2-dependent and O
2-independent mechanisms to kill micro-organisms [
85]. Phagocytosis triggers the generation of
•O
2− and other ROS and reactive species, such as hydrogen peroxide H
2O
2, hypochlorous acid HOCl, hydroxyl radical
•OH and chloramines, as potent microbicidal agents [
86]. In parallel, cytoplasmic granules fuse with phagosomes containing bacteria in a process known as degranulation, thereby enriching the vacuole lumen with antimicrobial peptides and proteases [
87].
NOx catalyzes the reduction reaction of O
2 to
•O
2− and/or H
2O
2 using NADPH as an electron donor and it is located extracellularly [
88]. NOx is involved in pathogen clearance and the regulation of associated inflammation plays an important role in physiological and pathological conditions, such as acute lung injury and bacterial or fungal infections. NOx is electrogenic and allows electron transport across the plasma membrane (altering ionic currents) [
89], induces apoptosis (mediating in physiological and pathological processes) [
90], regulates cytokine production and T cell death [
91], influences gene expression and promotes the formation of extracellular traps [
92,
93].
The significance of NOx and ROS production is exemplified by a rare inherited disorder known as chronic granulomatous disease CGD. Individuals with CGD have persistent bacterial and fungal infections due to defects in NOx [
94].
Following NOx activation, there is a rapid expenditure of O
2 in neutrophils, and this mechanism is called the “respiratory burst”. NOx activation by neutrophils occurs in response to stimuli such as formylated peptides, opsonized particles, integrin-dependent adhesion, and the binding of specific pathogen recognition receptors (e.g., dectin-1). SYK tyrosine kinase is a critical component of integrin signaling in neutrophils, mediating NOx activation [
95]. SYK tyrosine kinase is a non-receptor kinase that was long considered to exclusively mediate receptor signaling in the adaptive immune response. However, recent studies indicate that it is also involved in innate immunity and non-immune functions. SYK mediates integrin signaling in neutrophils, macrophages, and platelets, signaling by P-selectin glycoprotein ligand 1 (PSGL1), as well as the development of osteoclasts [
96]. SYK participates in the innate recognition of fungal and other microbial pathogens, as well as of tissue damage, by C-type lectins. SYK activation by C-type lectins activates the caspase-recruitment domain 9–B cell lymphoma and it is also required for NLR family, pyrin domain-containing 3 (NLRP3) inflammasome activation following fungal infection [
97].
It is known that H
2O
2 forms naturally in living organisms and its attributed physiological role is the capability to induce bacterial killing. It has been estimated that, in lymphocytes, the half-life of the H
2O
2 is 1 ms while that of the anion
•O
2− is 1 µs [
98]. It is not a free radical, but it is a very important reactive form, generating the
•OH radical in the presence of metals such as iron (Fenton reaction). The hydroxyl radical
•OH, the most powerful ROS oxidant, is formed during the Haber–Weiss reaction, by the Fenton reaction or by decomposition of peroxynitrite, and has a very short half-life (10
−9 s) and high reactivity.
The central source of H
2O
2 is enzymatically catalyzed by superoxide dismutation through the enzyme superoxide dismutase SOD. SOD is the only enzyme that can clear
•O
2− and it is present at the mitochondrial level as well, in the cytoplasm and extracellular space [
99]. It is composed of three isoforms, SOD1 (Cu/Zn-SOD is the predominant
•O
2− scavenger and is localized in the cytoplasm), SOD2 (Mn-SOD, in the mitochondrial intermembrane space, nucleus, and lysosomes) and SOD3 (Cu/Zn-SOD, is localized in the mitochondrion and extracellular matrix) [
100].
SOD-catalyzed dismutation of the superoxide radical can be characterized as the next half-reactions, Figure 4.
Figure 4. SOD-catalyzed dismutation of the superoxide radical. M = [Cu (n = 1); Mn and Fe (n = 2)]. The oxidation state of the metal cation varies between n and n + 1.
Oxidative burst is the rapid release of ROS from different cell types, macrophages and neutrophils are especially implicated, and it requires a 10-to-20-fold increase in oxygen consumption through NOx activity. The oxidative burst in phagocytes is commonly associated with bacterial killing, but in the case of alveolar macrophages, they typically produce lower levels of ROS than neutrophils and may require their activation to exhibit their bactericidal properties. Instead, their transient oxidative burst regulates the inflammatory response by inducing cytokine synthesis for redox signaling, resulting in an influx of activated neutrophils and macrophages [
101].
In adaptive immunity, ROS-mediated T-cell activation has been suggested to have an immunosuppressive role. T cell activation also requires the help of accessory cells, induction of regulatory T cells Treg by macrophage-derived ROS suppresses other T cells also via ROS. Additionally, localized ROS production drives Treg lineage commitment, while their removal decreases the balance of Treg/T effector cells [
102].