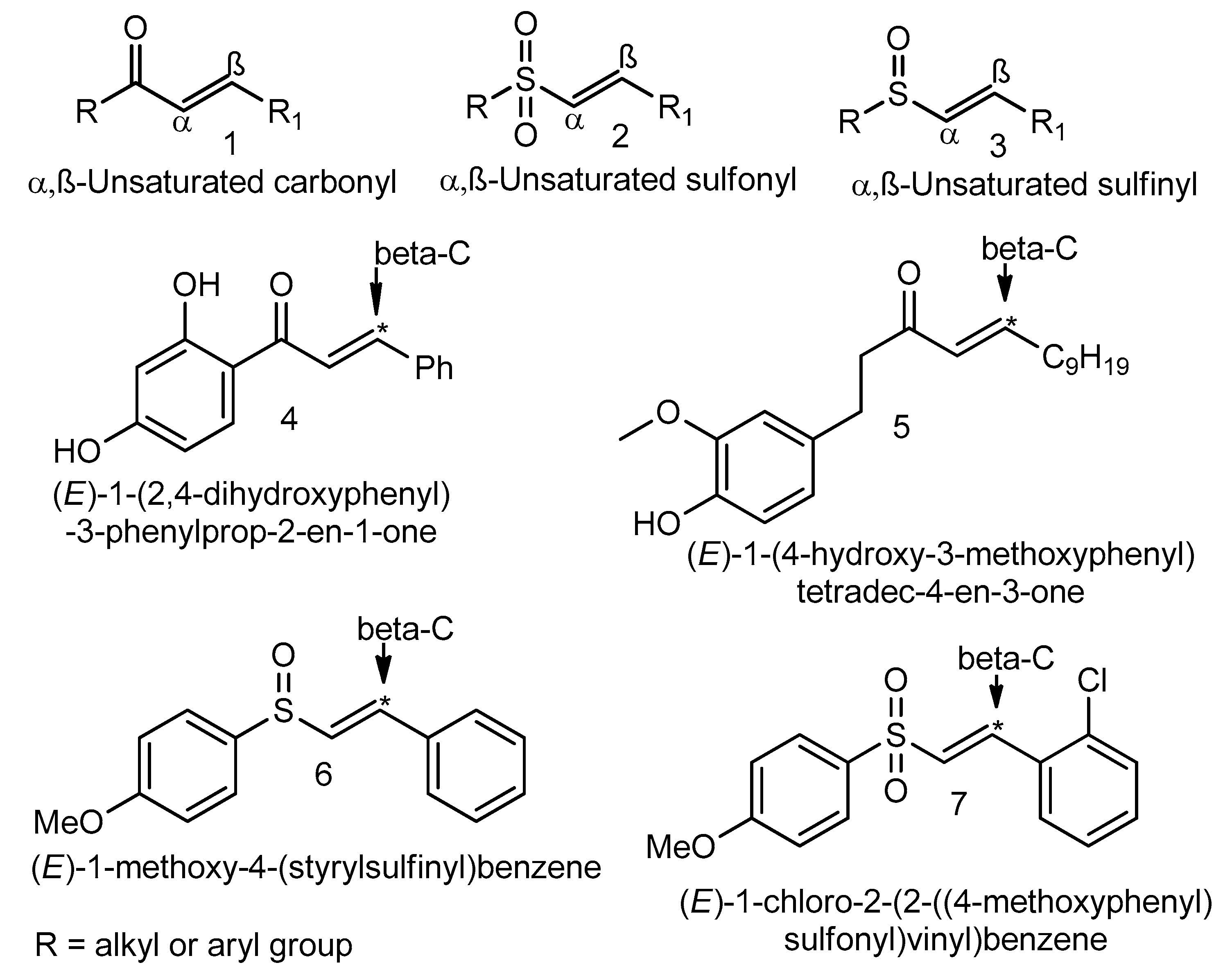
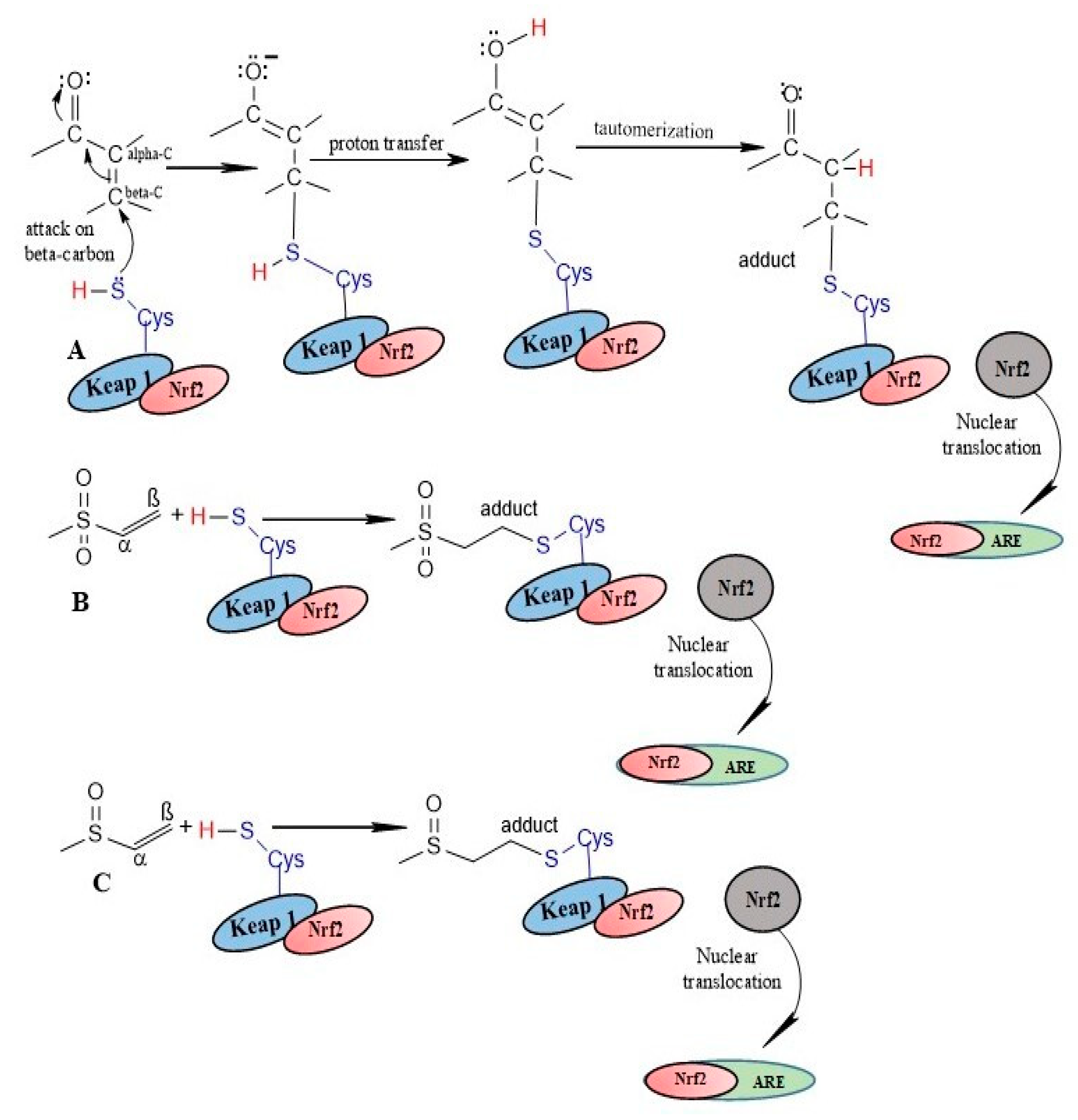
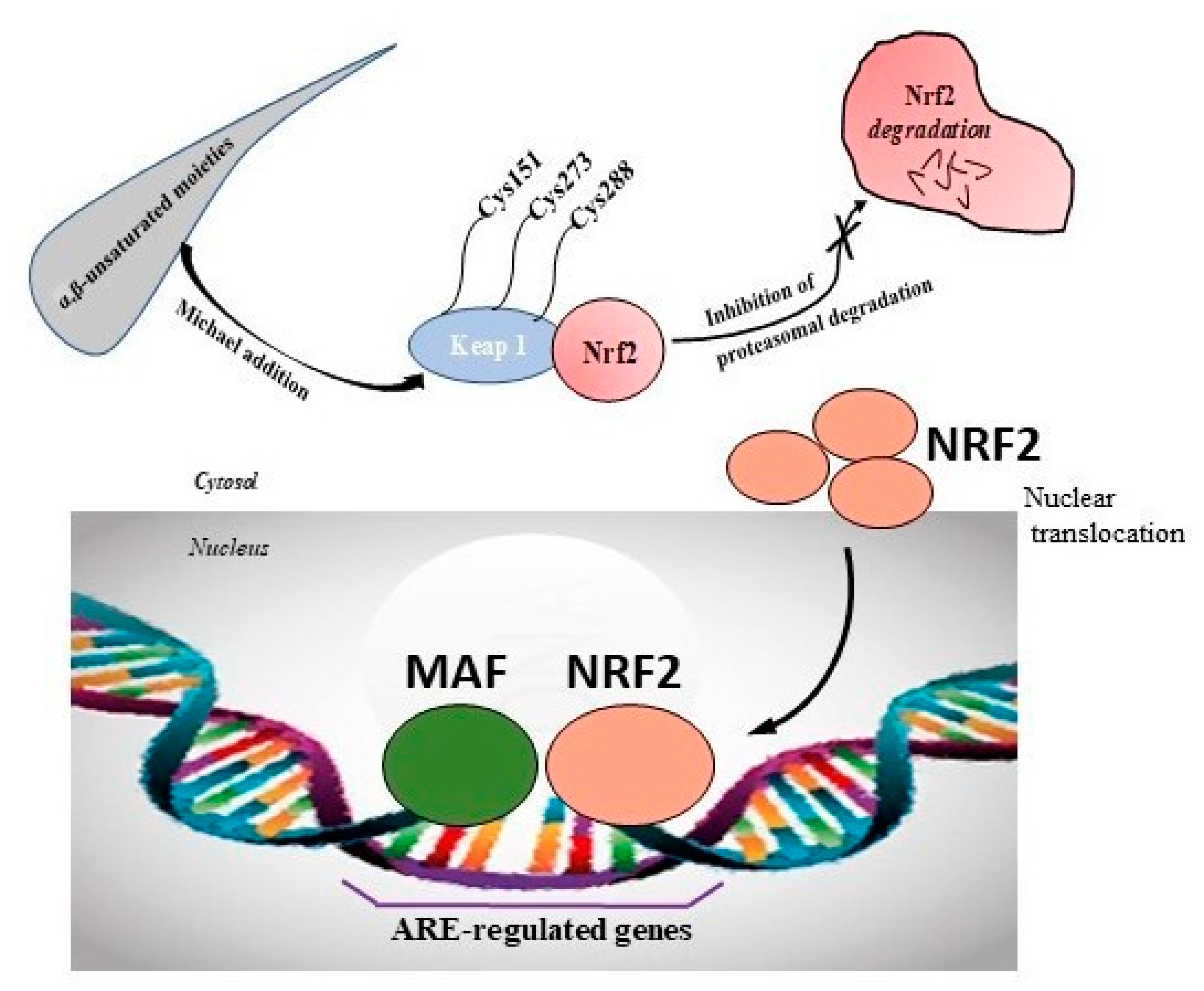
The surge of scientific interest in the discovery of Nuclear Factor Erythroid 2 (NFE2)-Related Factor 2 (NRF2)-activating molecules underscores the importance of NRF2 as a therapeutic target especially for oxidative stress. The chemical reactivity and biological activities of several bioactive compounds have been linked to the presence of α,β-unsaturated structural systems. The α,β-unsaturated carbonyl, sulfonyl and sulfinyl functional groups are reportedly the major α,β-unsaturated moieties involved in the activation of the NRF2 signaling pathway. The carbonyl, sulfonyl and sulfinyl groups are generally electron-withdrawing groups, and the presence of the α,β-unsaturated structure qualifies them as suitable electrophiles for Michael addition reaction with nucleophilic thiols of cysteine residues within the proximal negative regulator of NRF2, Kelch-like ECH-associated protein 1 (KEAP1). The physicochemical property such as good lipophilicity of these moieties is also an advantage because it ensures solubility and membrane permeability required for the activation of the cytosolic NRF2/KEAP1 system.
- NRF2
- KEAP1
- α,β-unsaturated moiety
1. Introduction
2. Modulation of NRF2/KEAP1 Signaling Pathway by α,β-Unsaturated Moiety-Bearing Compounds
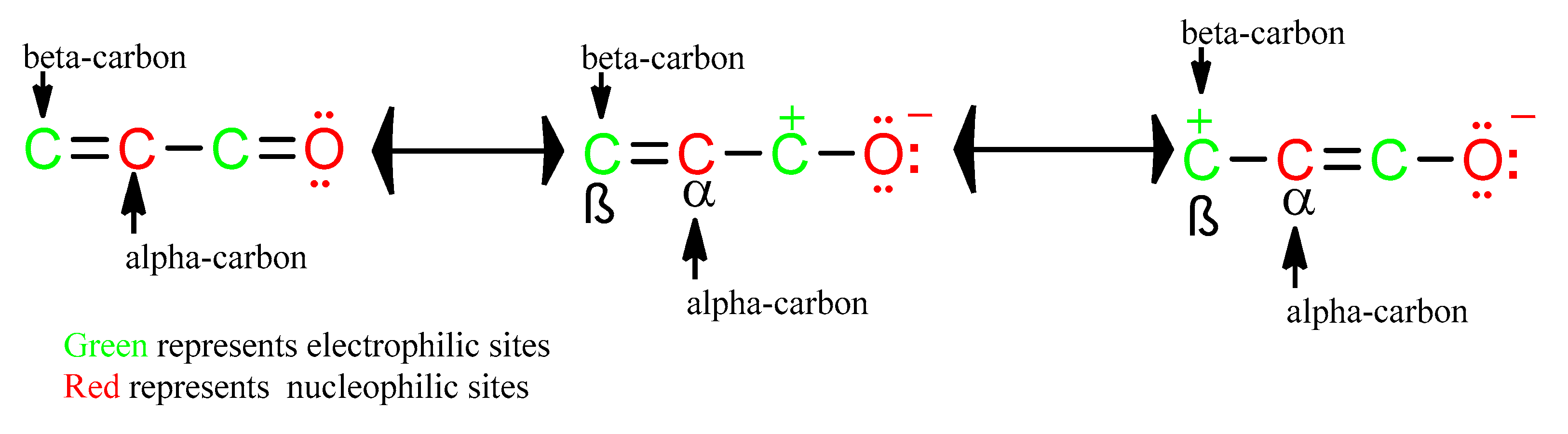
3. α,β-Unsaturated Carbonyls
3.1. Sesquiterpene Lactones
3.1.1. Parthenolide
3.1.2. Helenalin
3.1.3. Alantolactone
3.1.4. Costunolide
3.2. Curcumin
3.3. J-Series Cyclopentenone Prostaglandin
3.4. Chalcone and Its Derivatives
3.5. Dimethyl Fumarate
4. α,β-Unsaturated Sulfonyls
4.1. Vinyl Sulfones
4.2. Vinyl Sulfonamides
4.3. Vinyl Sulfonates
5. α,β-Unsaturated Sulfinyls
Vinyl Sulfoxide
This entry is adapted from the peer-reviewed paper 10.3390/ijms23158466
References
- Sulsen, V.P.; Martino, V.S. Sesquiterpene Lactones: Advances in Their Chemistry and Biological Aspects, 1st ed.; Springer Nature: Cham, Switzerland, 2018.
- Carlstrom, K.E.; Chinthkindi, P.K.; Espinosa, B.; Nimer, F.A.; Arner, E.S.J.; Arvidsson, P.I.; Piehl, F.; Johnasson, K. Characterization of more selective central nervous system NRF2-activating novel vinyl sulfoxime compounds compared to dimethyl fumarate. Neurotherapeutics 2020, 17, 114–1152.
- Shim, S.Y.; Hwang, H.S.; Nam, G.; Choi, K. Synthesis and NRF2 activating ability of thiourea and vinyl sulfoxide derivatives. Bull. Korean Chem. Soc. 2013, 34, 2317–2320.
- Mayer, R.J.; Allihn, P.W.A.; Hampel, N.; Mayer, P.; Sieber, S.A.; Ofial, A.R. Electrophilic reactivities of cyclic enones and α,β-unsaturated lactones. Chem. Sci. 2021, 12, 4850–4865.
- Li, Z.; Yazaki, R.; Ohsima, T. Chemo-and region selective direct functional group installation through catalytic hydroxyl group selective conjugation addition of amino alchohols to α,β-unsaturated sulfonyl compounds. Org. Lett. 2016, 18, 3350–3353.
- Lipton, S.; Rezaie, T.; Nutter, A.; Lopez, K.M.; Parker, J.; Kosaka, K.; Satoh, T.; McKercher, S.R.; Masiliah, E.; Nakanishi, N. Therapeutic advantage of pro-electrophoilic drugs to activate the Nrf2/ARE pathway in Alzheimer’s disease models. Cell Death Dis. 2016, 7, e2499.
- Egbujor, M.C.; Saha, S.; Buttari, B.; Profumo, E.; Saso, L. Activation of NRF2 signaling pathway by natural and synthetic chalcones: A therapeutic road map for oxidative stress. Expert Rev. Clin. Pharmacol. 2021, 14, 465–480.
- Robledinos-Anton, N.; Fernandez-Gines, R.; Manda, G.; Cuadrado, A. Activators and inhibitors of NRF2: A review of their potential for clinical development. Oxid. Med. Cell. Longev. 2019, 2019, 9372182.
- Chio, J.W.; Kim, S.; Park, J.; Kim, H.J.; Shin, S.J.; Kim, J.W.; Woo, S.Y.; Lee, C.; Han, S.M.; Lee, J.; et al. Optimization of vinyl sulfone derivatives as potent nuclear factor erythroid 2-related factor 2 activities for disease therapy. J. Med. Chem. 2019, 62, 811–830.
- Yagishita, Y.; Gatbonton-Schwager, T.N.; McCallum, M.L.; Kensler, T.W. Current landscape of NRF2 biomarkers in clinical trials. Antioxidants 2020, 9, 716.
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. therapeutic roles of curcumin: Lessons learned from clinical trials. AAPS J. 2013, 15, 195–218.
- Zhao, C.; Rakesh, K.P.; Ravidar, L.; Fang, W.Y.; Qin, H.L. Pharmaceutical and medicinal significance of sulfur (SVI)-containing motifs for drug discovery: A critical review. Eur. J. Med. Chem. 2019, 162, 679–734.
- Lu, L.; Feng, Q.; Su, T.; Cheng, Y.; Huang, Z.; Huang, Q.; Liu, Z. Chapter 35, Pharmacoepigenetics of Chinese herbal components in cancer. In Translational Epigenetics, Pharmacoepigenetics; Cacabelos, R., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 10, pp. 859–869.
- Rasul, A.; Parveen, S.; Ma, T. Costunolide: A novel anticancer sesquiterpene lactone. Bangladish J. Pharmacol. 2012, 7, 6–13.
- Chataigner, I.; Panel, C.; Gerard, H.; Piettre, S.R. Sulfonyl Vs. Carbonyl group: Which is the more electron–withdrawing? Chem. Commun. 2007, 31, 3288–3290.
- Lee, J.A.; Kim, J.H.; Woo, S.Y.; Son, H.Y.; Han, S.H.; Jang, B.K.; Choi, J.W.; Kim, D.J.; Park, K.D.; Hwang, O. A novel compound VSC2 has anti-inflammatory and antioxidant properties in microglia and in Parkinson’s disease model. Br. J. Pharmacol. 2015, 172, 1087–1100.
- Holland, R.; Fishbein, J.C. Chemistry of the cysteine sensors in Kelch-like ECH- associated protein 1. Antioxid. Redox Signal. 2010, 13, 1749–1761.
- Choi, J.W.; Shin, S.J.; Kim, H.J.; Park, J.-H.; Kim, J.K.; Lee, E.H.; Pae, A.N.; Bahn, Y.S.; Park, K.D. Antioxidant anti-inflammatory, and neuroprotective effects of novel vinyl sulfonate compounds as NRF2 activator. ACS Med. Chem. Lett. 2019, 10, 1061–1067.
- Pearson, R.G. Hard and soft acids and bases the evolution of a chemical concept. Coord. Chem. Rev. 1990, 100, 403–425.
- Saverland, M.; Mertes, R.; Morozzi, C.; Eggler, A.L.; Gamon, L.F. Kinetic assessment of Michael addition reaction of alpha, beta-unsaturated carbonyl compounds to amino acid and protein thiols. Free Radic. Biol. Med. 2021, 169, 1–11.
- Reusch, W. Nucleophilic addition to α,β-unsaturated carbonyl compounds. In Virtual Textbook of Organic Chemistry, 5th ed.; Michigan State University Press: East Lansing, MI, USA, 1999; p. 1162.
- Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Cole, R.N.; Itoh, K.; Wakabayashi, N.; Katoh, Y.; Yamamoto, M.; Talalay, P. Direct evidence that sulfhydryl groups of KEAP1 are the sensors regulating induction of pahse 2 enzymes that protect against carcinogens and oxidants. Proc. Natl. Acad. Sci. USA 2002, 99, 11908–11913.
- Silva, M.; Pruccoli, L.; Morronoi, F.; Sita, G.; Seghetti, f.; Viegas, C.; Tarozzi, A. The keap 1/NRF2-ARE pathway as a pharmacological target for chalcones. Molecules 2018, 23, 1803.
- Kobayashi, A.; Kang, M.I.; Watai, Y.; Tong, K.I.; Shibata, T.; Uchida, K.; Yamamoto, M. Oxidative and electrophilic stresses activate NRF2 through inhibition of ubiquitination activity of KEAP1. Mol. Cell. Biol. 2006, 26, 221–229.
- Eggler, A.L.; Liu, G.; Pezzuto, J.M.; Van Breemen, R.B.; Mesecar, A.D. Modifying specific cysteines of the electrophile-sensing human Keap1 proteinis insufficient to disrupt binding to the Nrf2 domain Neh2. Proc. Natl. Acad. Sci. USA 2005, 102, 10070–10075.
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; Leon, R.; Lopez, M.G.; Oliva, B.; et al. Transcription factor NRF2 as a therapeutic target for chronic diseases: A system medicine approach. Pharmacol. Rev. 2018, 70, 348–383.
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317.
- Taguchi, K.; Motohashi, H.; Yamamoto, M. Molecular mechanisms of the KEAP1–NRF2 pathway in stress response and cancer evolution. Genes Cells 2011, 16, 123–140.
- Egbujor, M.C.; Petrosino, M.; Zuhra, K.; Saso, L. The Role Of Organosulfur Compounds As Nrf2 Activators And Their Antioxidant Effects. Antioxidants 2022, 11, 1255.
- Tonelli, C.; Chio, I.I.C.; Tuveson, D.A. Transcriptional regulation by NRF2. Antioxid. Redox Signal. 2018, 29, 1727–1745.
- Ahmed, S.M.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. NRF2 signaling pathaway: Pivotal roles in inflammation. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 585–597.
- Cores, A.; Piquero, M.; Villacapa, M.; Leon, R.; Menendez, J.C. NRF2 regulation processes as a source of potential drug targets against neurodegenerative diseases. Biomolecules 2020, 10, 904.
- Saito, R.; Suzuki, T.; Hiramoto, K.; Asami, S.; Naganuma, E.; Suda, H.; Iso, T.; Yamamoto, H.; Morita, M.; Baird, L.; et al. Characterizations of Three Major Cysteine Sensors of Keap1 in Stress Response. Mol. Cell. Biol. 2015, 36, 271–284.
- Levonen, A.L.; Landar, A.; Ramachandran, A.; Ceaser, E.K.; Dickinson, D.A.; Zanoni, G.; Morrow, J.D.; Darley-Usmar, V.M. Cellular mechanisms of redox cell signaling: Role of cysteine modification in controlling antioxidant defences in response to electrophilic lipid oxidation products. Biochem. J. 2004, 378, 373–382.
- Wakabayashi, N.; Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Kang, M.; Kobayashi, A.; Yamamoto, M.; Kensler, T.W.; Talalay, P. Protection against electrophile and oxidant stress by induction of the phase 2 response: Fate of cysteines of the KEAP1 sensor modified by inducers. Proc. Natl. Acad. Sci. USA 2004, 101, 2040–2045.
- Yamamoto, T.; Suzuki, T.; Kobayashi, A.; Wakabayashi, J.; Maher, J.; Motohashi, H.; Yamamoto, M. Physiological significance of reactive cysteine residues of KEAP1 in determining NRF2 activity. Mol. Cell. Biol. 2008, 28, 2758–2770.
- Meadows, D.C.; Gervay-Hague, J. Vinyl sulfones: Synthetic preparations and medicinal chemistry applications. Med. Res. Rev. 2006, 26, 793–814.
- O’Connor, D.E.; Lyness, W.I. The effect of methyl-mercapto, methylsulfinyl, and methylsulfonyl groups on the equilibrium in three-carbon prototropic systems. J. Am. Chem. Soc. 1964, 86, 3840–3846.
- Cilento, G. The expansion of the sulfur outer shell. Chem. Rev. 1960, 60, 147–167.
- Inomata, K.; Hirata, T.; Suhara, H.; Kinoshita, H.; Kotake, H.; Senda, H. Stereochemistry of the conversion of γ-substituted (E)-vinyl sulfones to the corresponding allyl sulfones. Determination of the relative drgree of syn-effect. Chem. Lett. 1988, 17, 2009–2012.
- Snyder, H.R.; Eliel, E.L.; Charnahan, R.E. Studies in the sulfone series. J. Am. Chem. Soc. 1951, 73, 3258.
- Snyder, H.R.; Hallada, D.P. α,β-Unsaturated sulfonyl compounds in Diels-Alder reaction II. J. Am. Chem. Soc. 1952, 74, 5595–5597.
- Saddler, J.C.; Conrad, P.C.; Fuchs, P.L. A new (3+3) annulations route to isoquinoline-3-carboxylates. Tetrahedron Lett. 1978, 19, 5079–5081.
- Liu, L.K.; Chi, Y.; Jen, K.Y. Copper-catalyzed additions of sulfonyl iodides to simple and cyclic alkenes. J. Org. Chem. 1980, 45, 406–410.
- Taber, D.F.; Saleh, S.A. Branching strategy in organic synthesis 2-Reveral of olefin polarization with concomitant carbon-carbon bond formation. J. Org. Chem. 1981, 46, 4817–4819.
- Smith, M.B.; March, J. Advanced Organic Chemistry: Ractions, Mechanism and Structure, 6th ed.; Wiley-Interscience: New York, NY, USA, 2007.
- Rodrigues, T.; Recker, D.; Schneider, P.; Schneider, G. Counting on natural products for drug design. Nat. Chem. 2016, 8, 531–541.
- Arshad, L.; Jantan, T.; Bukhari, S.N.A.; Haque, A. Immunosuppressive effects of natural α,β-unsaturated carbonyl-based compounds, and their avalogs and derivatives, on immune cells: A review. Front. Pharmacol. 2017, 8, 22.
- Snyder, N.W.; Singh, B.; Buchan, G.; O’Brien, J.; Arroyo, A.D.; Liu, X.; Sobol, R.W.; Blair, I.A.; Mesaros, C.A.; Wendell, S.G. Primary saturation of α,β-unsaturated carbonyl containing fatty acid does not abolish electrophilicity. Chem.-Biol. Interact. 2021, 350, 109689.
- Thapa, P.; Upadhyay, S.P.; Suo, W.Z.; Singh, V.; Gurung, P.; Lee, E.S.; Sharma, R.; Sharm, M. Chalcone and its analogs:therapeutic and diagnostic applications in Alzheimer’s disease. Bioorganic Chem. 2021, 108, 104681.
- Naidu, S.D.; Dinkova-Kostova, A.T. KEAP1, a cysteine-based sensor and a drug target for the prevention and treatment of chronic disease. Open Biol. 2020, 10, 200105.
- Shin, J.W.; Chun, K.-S.; Kim, D.-H.; Kim, S.-J.; Kim, S.H.; Cho, N.-C.; Na, H.-K.; Surh, Y.-J. Curcumin induces stabilization of Nrf2 protein through Keap1 cysteine modification. Biochem. Pharmacol. 2020, 173, 113820.
- Grimsrud, P.A.; Xie, H.; Griffin, T.J.; Bernlohr, D.A. Oxidative stress and covalent modification of protein withbioactive aldehydes. J. Biol. Chem. 2008, 283, 21837–21841.
- Ahn, Y.-H.; Hwang, Y.; Liu, H.; Wang, X.J.; Zhang, Y.; Stephenson, K.K.; Boronina, T.N.; Cole, R.N.; Dinkova-Kostova, A.T.; Talalay, P.; et al. Electrophilic tuning of the chemoprotective natural product sulforaphane. Proc. Natl. Acad. Sci. USA 2010, 107, 9590–9595.
- Tirumalai, R.; Rajesh, K.T.; Mai, K.H.; Biswal, S. Acrolein causes transcriptional induction of phase II genes by activation of Nrf2 in human lung type II epithelial (A549) cells. Toxicol. Lett. 2002, 132, 27–36.
- Randall, M.J.; Spiess, P.C.; Hristova, M.; Hondal, R.J.; Vander, A. Acrolein-induced activation of mitogen-activated protein kinase signalling is mediated by alkylation of thioredoxin reductase and thioredoxin1. Redox Biol. 2013, 1, 265–275.
- Poganik, J.R.; Aye, Y. Electrophilic signaling and emerging immune and neuromodulatory electrophilic pharmaceuticals. Front. Aging Neurosci. 2020, 12, 1.
- Ploger, M.; Sendker, J.; Langer, K.; Schmidt, T.J. Covalent modification of human serum albumin by the natural sesquiterpene lactone partheolide. Molecules 2015, 20, 6211–6223.
- Wu, C.C.N.; Rosenbach, M.D.; Corr, M.; Schwab, R.B. Selectivity of electrophilic compounds in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 2010, 107, 7479–7484.
- Ghantous, A.; Gali-Muhtasib, H.; Vuorela, H.; Salibba, N.A.; Darwiche, N. What made sequiterpene lactones reach cancer clinical trials? Drug Discov. Today 2010, 15, 668–678.
- Moujir, L.; Callies, O.; Sousa, P.M.C.; Sharopov, F.; Seca, A.M.L. Applications of sequiterpene lactones: A review of sow potential success cases. Appl. Sci. 2020, 10, 3001.
- Gach, K.; Dlugosz, A.; Janecka, A. The role of oxidative stress in anticancer activity of sesquiterpene lactones. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2015, 388, 477–486.
- Esatbeyoglu, T.; Obermair, B.; Dorn, T.; Sims, K.; Rimbach, G.; Birringer, M. Sesquiterpene lactone composition and cellular Nrf2 induction of Taraxacum officinale leaves and roots and taraxinic acid β-d-glucopyranosyl ester. J. Med. Food 2017, 20, 71–78.
- Umemura, K.; Itoh, T.; Hamada, N.; Fujita, Y.; Ako, Y.; Nozawa, Y.; Matsuura, N.; Linuma, M.; Ito, M. Preconditioning by sesquiterpene lactone enhances H2O2-induced NRF2/ARE activation. Biochem. Biophys. Res. Commun. 2008, 368, 948–954.
- Fischedick, J.T.; Standford, M.; Johoson, D.A.; De Vos, R.C.H.; Todorovic, S.; Banjanac, T.; Verpoorte, R.; Johnson, J.A. Activation of antioxidant response element in mouse primary cortical cultures with sesquiterpene lactose isolated from Tanaetum parthenium. Planta Med. 2012, 78, 1725–1730.
- Formisano, C.; Rigano, D.; Millan, E.; Munoz, E.; Taglialatela-Scatati, O. Anti-inflammatory sesquiterpene lactones from Onopordum illyricum L. (Asteraceae) an Italian medicinal plant. Fitoterapia 2017, 116, 61–65.
- Nodwell, M.B.; Menz, H.; Kirsch, S.F.; Sieber, S.A. Rugulactone and its analogue exert antibacterial effects through multiple mechanisms including inhibition of thiamine biosynthesis. ChemBioChem 2012, 13, 1439–1446.
- Pareek, A.; Suthur, M.; Rathore, G.S.; Bansal, V. Feverfe (Tanacetum parthenium L): A systematic review. Pharmacogn. Rev. 2011, 5, 103–110.
- Shin, M.; McGonnor, A.; DiNatale, G.J.; Chiramaneqong, T.; Cai, T.; Connor, R.E. Hsp72 is an intracellular target of the α,β-unsaturated sesquiterpene lactone, parthenolide. ACS Omega 2017, 2, 7267–7274.
- Kupchan, S.M.; Fessler, D.C.; Eakin, M.A.; Giacobbe, T.J. Reactions of alpha methylene lactone tumor inhibitors with model biological neucleophiles. Science 1970, 168, 376–378.
- Kim, C.Y.; Kang, B.; Hong, J.; Choi, H.-S. Parthenolide inhibits lipid accumulation via activation of NRF2/KEAP1 signaling during adipocyte differentiation. Food Sci. Biotechnol. 2019, 29, 431–440.
- Kim, C.Y.; Kang, B.; Suh, H.J.; Choi, H.-S. Parthenolide, a fever few-derived phytochemical, ameliorates obesity and obesity-indvied infleammatory responses via the NRF2/Keap pathway. Pharmacol. Res. 2019, 145, 104259.
- Mao, W.; Zhu, Z. Parthenolide inhibirs hydrogen peroxide-induced osteoblast apoptosis. Mol. Med. Rep. 2018, 17, 8369–8376.
- Uchi, H.; Arrighi, J.F.; Aubry, J.P.; Furue, M.; Hauser, C. The sesquiterpene lactone parthenolide inhibits LSP- but not TNF-alpha induced maturation of human monocyte-derived dendritic cells by inhibition of the P38 mitogen-activated protein kinase pathway. J. Allergy Clin. Immunol. 2002, 110, 269–276.
- Hwang, D.; Fischer, N.H.; Jang, B.C.; Tak, H.; Kim, J.K. Inhibition of the expression of inducible cyclooxygenase and proinflammatory cytokines by sesquiterpene lactones in microphags correlates with the inhibition of MAP Kinases. Biochem. Biophys. Res. Commun. 1996, 226, 810–818.
- Bostwick, D.-G.; Alexander, E.E.; Singj, R.; Shan, A.; Qian, J.; Santella, R.M.; Oberley, L.W.; Yan, T.; Zhong, W.; Jiang, X.; et al. Antioxidant enzyme expression and reactive oxygen species damage in prostatic intraepithelial neoplasia and cancer. Cancer 2000, 89, 123–134.
- Dey, S.; Sarkar, M.; Giri, B. Anti-inflammatory and anti-tumor activities of parthenolide: An update. J. Chem. Biol. Ther. 2016, 1, 107.
- Li-Weber, M.; Palfi, K.; Giaisi, M.; Krammer, P.H. Dual role of the anti-inflammatory sesquiterpene lactone: Regulation of life and death by parthenolide. Cell. Death Differ. 2005, 12, 408–409.
- Carlisi, D.; De Blasio, A.; Drago-Ferrante, R.; Fiore, R.D.; Buttitta, G.; Morreale, M.; Scerri, C.; Vento, R.; Tosoriere, G. Parthenolide Prevents resistance of MDA-MB231 cells to doxorubicin and mitoxantrone: The role of NRF2. Cell. Death Discov. 2017, 3, 17078.
- Carlisi, D.; Buttitta, R.; Di Fiore, R.; Scerri, C.; Drago-Ferrante, R.; Vento, R.; Tesoriere, G. Parthenolide and DMAPT exert cytotoxic oxidative stress, mitochondrial dysfunction and necrosis. Cell. Death Dis. 2016, 7, e2194.
- Kriplani, P.; Guarve, K. Recent patents on anti-cancer potential of helenalin. Recent Pat. Anticancer Drug Discov. 2020, 15, 132–142.
- Shoaib, M.; Shah, I.; Ali, N.; Adhikari, A.; Tahir, M.N.; Shah, S.W.A.; Ishtiag, S.; Khan, J.; Khan, S.; Umer, M.N. Sesquiterpene lactone! A promising antioxidant, anticancer and moderate antinociceptive agent from artemesia macrocephala jacquem. BMC Complement. Altern. Med. 2017, 17, 27.
- Lyss, G.; Knorre, A.; Schmidt, T.J.; Pahl, H.L.; Merfort, I. The anti-inflammatory sesquiterpene lactone helenaline inhibits the transcription factor NF-kapper B by directly targeting p65. J. Biol. Chem. 1998, 273, 33508–33516.
- Li, Y.; Zeng, Y.; Huang, Q.; Wen, S.; Wei, Y.; Chen, Y.; Zhang, X.; Bai, F.; Lu, Z.; Wei, J.; et al. Helenalin from centipede minima ameliorates acute hepatic injury by protecting mitochondria function, activating NRF2 pathway and inhibiting NF-kB activation. Biomed. Pharmacother. 2019, 119, 109435.
- Lin, X.; Zhang, S.; Huang, R.; Wei, L.; Tan, S.; Liang, S.; Tian, Y.; Wu, X.; Lu, Z.; Huang, Q. Helenalin enhancing ethanol metabolism, inhibiting oxidative stress and suppressiong HSC activation. Fitoterapia 2014, 95, 203–213.
- Chun, J.; Li, R.-J.; Cheng, M.S.; Kim, Y.S. Alantolactone, a sesquiterpene lactone isolated from Inula helenium L. Selectrively suppress STAT3 actyivation and exhibits anticancer activity in MDA-MB-231 cells. Cancer Lett. 2015, 357, 393–403.
- Kahn, M.; Li, T.; Ahmad, K.M.K.; Rasul, A.; Nawaz, F.; Sun, M.; Zheng, Y.; Ma, T. Alantolactone induces apoptosis in HepG2 cells though GSH depletion, inhibition of STAT3 activation, and mitochondrial dysfunction. Biomed. Res. Int. 2013, 2013, 719858.
- Huo, J.; Shi, H.M.; Li, W.W.; Wang, M.Y.; Li, X.B. HPLC determination and NMR structural elucidatrion of sesquiterpene lactones in Inula helenuim. J. Pharm. Biomed. Anal. 2010, 51, 942–946.
- Liu, J.; Liu, M.; Wang, S.; He, Y.; Huo, Y.; Yang, Z.; Cao, X. Alntolactone induces apoptosis and suppresses migration in MCF-7 human breast cancer cells via the p38 MAPK, NF-kB and NRF2 signaling pathways. Int. J. Mol. Med. 2018, 42, 1847–1856.
- Seo, J.Y.; Lim, S.S.; Kim, J.R.; Lim, J.-S.; Ha, Y.R.; Lee, I.A.; Kim, E.J.; Park, J.H.Y.; Kim, J.-S. NRF2-mediated Induction of detoxifying enxymes by alantolactone present in Invla helenium. Phytother. Res. 2008, 22, 1500–1505.
- Dang, X.; Holt, B.; Ning, Q.; Liu, Y.; Guo, J.; Niu, G.; Chen, M. Alantolactone suppresses inflammation, apoptosis and oxidative stress in cigarette smoke-induced human bronchial epithelial cells through activation of NRF2/HO-1 and inhibition of the NF-kB pathways. Respirat. Res. 2020, 21, 95.
- Kim, D.Y.; Choi, B.Y. Costunolide, a bioactive sesquiterpene lactone with diverse therapentic potential. Int. J. Mol. Sci. 2019, 20, 2926.
- Mao, J.; Yi, M.; Wang, R.; Huang, Y.; Chen, M. Protective effects of costunolide against D-galactos-amine and lipopolysaccharide-induced acute liver injury in mice. Front. Pharmacol. 2018, 9, 1469.
- Pae, H.O.; Jeong, G.-S.; Kim, H.-S.; Woo, H.Y.; Rhew, H.S.; Kim, D.H.; Sohn, Y.C.; Kim, H.T.; Chung, H.-T. Costunolide inhibits production of tumor necrosis factor- K and interleukin-6 by inducing heme oxygenase-1 in RAW 264-7 Microphages. Inflamm. Res. 2007, 56, 520–526.
- Eliza, J.; Daisy, P.; Ignacimuthu, S. Antioxidant activity of costunolide and eremanthin isolated from costus speciosus (Ken ex. Retz) sm. Chem. Biol. Interact. 2010, 188, 467–472.
- Zheng, H.; Chen, Y.; Zhang, J.; Wang, L.; Jin, Z.; Huang, H.; Man, S.; Gao, W. Evaluation of costunolide and dehydrocostulactone on ethano-induced gastric ulcer in mice based on multi-pathway regulation. Chem. Biol. Interact. 2016, 250, 68–77.
- Peng, S.; Hou, Y.; Yao, J.; Fang, J. Activation of NRF2 by costunolide provides neuroprotective effect in PC12 Cells. Food Funct. 2019, 10, 4143.
- Mythri, R.B.; Bharath, M.M. Curcumin: A potential neuroprotective agent in Parkinson’s disease. Curr. Pharm. Des. 2012, 18, 91–99.
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the golden neutraceutical: Multitargeting for multiple chronic disease. Br. J. Pharmacol. 2017, 174, 1325–1348.
- Rahban, M.; Habibi-Rezaei, M.; Mazaheri, M.; Saso, L.; Moosavi-Movahedi, A.A. Anti-viral potential and modulation of NRF2 by curcumin: Pharmacological implications. Antioxidants 2020, 9, 1228.
- Madden, S.K.; Itzhaki, L.S. Structural and mechanistic insights into the KEAP1-NRF2 system as a route to drug discovery. Biochim. Et. Biophys. Acta (BBA)-Proteins Proteom. 2020, 1868, 140405.
- Park, J.-Y.; Sohn, H.-Y.; Koh, Y.H.; Jo, C. Curcumin activates NRF2 through PKCᴕ-mediated p62 phosphorylation at Ser351. Sci. Rep. 2021, 11, 8430.
- Ashrafizadeh, M.; Ahmadi, Z.; Mohammadinejad, R.; Farkhondeh, T.; Samarghandian, S. Curcumin activates the NRF2 pathway and induces cellular protection against oxidative injury. Curr. Mol. Med. 2020, 20, 116–133.
- Gonzalez-Reyes, S.; Guzman-Beltran, S.; Medina-Campos, O.N.; Pedraza-Chaverri, J. Curcumin Pretreatment induces NRF2 and an antioxidant response and prevents Hemin-induced toxicity in primary cultures of cerebellar granule neurons of rats. Oxid. Med. Cell. Longev. 2013, 2013, 801418.
- Ren, L.; Zhan, P.; Wang, Q.; Wang, C.; Liu, Y.; Yu, Z.; Zhang, S. Curcumin unregulates the NRF2 system by repressing inflammatory signalling-mediated KEAP1 expression in insulin-resistant conditions. Biochem Biophys Res. Commun. 2019, 514, 691–698.
- Kim, J.S.; Oh, J.-M.; Choi, H.; Kim, S.W.; Kim, B.G.; Cho, J.H.; Lee, J.; Lee, D.C. Activation of the NRF2/HO-1 pathway by curcumin inhibits oxidative stress in human nasal fibroblasts exposed to urban particulate matter. BMC Compl. Med. Ther. 2020, 20, 101.
- Mishra, P.; Paital, B.; Jena, S.; Swain, S.S.; Kumar, S.; Yadav, M.K.; Chainy, G.B.N.; Samanta, L. Possible activation of NRF2 by vitamin E/curcumin against altered thyroid hormone induced oxidative stress via NFkB/AKT/Mtor/kap1 signaling in rat heart. Sci. Rep. 2019, 9, 7408.
- Straus, D.S.; Glass, C.K. Cyclopentenone prostaglandins: New insights on biological activities and cellular targets. Med. Res. Rev. 2001, 21, 185–210.
- Kim, E.H.; Surh, Y.J. 15deoxy-D12,14-prostaglandin J2 as a potential endogeneous regulator of redox-sensitive transcription factors. Biochem. Pharmacol. 2006, 72, 1516–1528.
- Song, N.-Y.; Kim, E.-H.; Na, H.-K.; Surh, Y.-J. 15-Deoxy-delta 12, 14-prostaglandin J2 induces upregulation of multidrug resistance-associated protein 1 via NRF2 activation in human breast cancer cells. Natural compounds and their role in apoptotic cell signaling pathways. Am. N. Y. Acad. Sci. 2009, 1171, 210–216.
- Yu, X.; Egner, P.A.; Wakabayashi, J.; Wakabayashi, N.; Yamamoto, M.; Kensler, T.W. NRF2-mediated induction of cytoprotective enzymes by 15-deoxyD1214 –prostaglandin J2 is attenuated by alkenal/one oxidoreductase. J. Biol. Chem. 2006, 281, 26245–26252.
- Kansanen, E.; Kuosmanen, S.M.; Leinonen, H.; Levonen, A.-L. The KEAP1-NRF2 pathway: Mechanisms of activation and dysregulation in cancer. Redox Biol. 2013, 1, 45–49.
- Ugwu, D.I.; Ezema, B.E.; Okoro, U.C.; Eze, F.U.; Ekoh, O.C.; Egbujor, M.C.; Ugwuja, D. 1. ChemInform Abstract: Synthesis and pharmacological applications of chalcones: A review. ChemInform 2015, 13, 459–500.
- Constantinescu, T.; Lungu, C.N. Anticancer activity of natural and synthetic chalcones. Int. J. Mol. Sci. 2021, 22, 11306.
- Tang, Y.-L.; Zheng, X.; Qi, Y.; Pu, X.-J.; Liu, B.; Zhang, X.; Li, X.-S.; Xiao, W.-L.; Wan, C.-P.; Mao, Z.-W. Synthesis and anti-inflammatory evaluation of new chalcone derivatives bearing bispiperazine linker as IL-1β inhibitors. Bioorganic Chem. 2020, 98, 103748.
- Ngameni, B.; Cedric, K.; Mbaveng, A.T.; Erdogan, M.; Simo, I.; Kuete, V.; Dastan, A. Design, synthesis, characterization, and anticancer activity of a novel series of O-Substituted chalcone derivatives. Bioorganic Med. Chem. Lett. 2021, 35, 127827.
- Miranda-sapla, M.M.; Tomiotto-Pellissier, F.; Assolini, J.P.; Carloto, A.C.M.; Bortoleti, B.T.D.S.; Goncalves, M.D.; Tavares, E.R.; Rodrigues, J.H.D.S.; Simao, A.N.C.; Yamauchi, L.M.; et al. Trans-chalcone modulates Leishmania amazonensis infection in vitro by NRF2 over expression affecting iron availability. Eur. J. Pharmacol. 2019, 853, 275–288.
- Kuhnl, J.; Roggenkamp, D.; Gehrke, S.A.; Stab, F.; Wenck, H.; Kolbe, L.; Neufang, G. Lichochalcone A activates NRF2 in vitro and contributes to licorice extract-induced lowered cutaneous oxidative stress. Exp. Damatol. 2015, 24, 42–47.
- Zhang, M.; Wu, Y.-Q.; Xie, L.; Wu, J.; Xu, K.; Xiao, J.; Chen, D.Q. Isoliquiritigenin protects against pancreatic injury and intestinal dysfunction after severe acute pancreatitis via NRF2 signaling. Front. Pharmacol. 2018, 9, 936.
- Yang, H.-L.; Yang, T.-Y.; Gowrisankar, Y.V.; Liao, C.-H.; Liao, J.-W.; Huang, P.-J.; Hseu, Y.-C. Suppression of LPS-induced inflammation by chalcone flavokawain A through activation of NRF2/ARE-mediated antioxidant genes and inhibition of ROS/NFkB signaling pathways in primary splenocytes. Oxid. Med. Cell. Longev. 2020, 2020, 3476212.
- Fox, R.J.; Miller, D.H.; Phillips, J.T.; Hutchinson, M.; Havrdova, E.; Kita, M.; Yang, M.; Raghupathi, K.; Novas, M.; Sweetser, M.T.; et al. Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N. Engl.J. Med. 2012, 367, 1087–1097.
- Bomprezzi, R. Dimethyl fumarate in the treatment of relapsing-remitting multiple sclerosis: An overview. Ther. Adv. Neurol. Disord. 2015, 8, 20–30.
- Bista, P.; Zeng, W.; Ryan, S.; Lukashev, M.; Yamamoto, M. Dimethyl fumarate suppresses inflammation in vitro via both NRF2-dependent and NRF2-independent pathways. Neurology 2012, 78, P02.108.
- Akino, N.; Wada-Hiraike, O.; Isono, W.; Terao, H.; Honjo, H.; Miyamoto, Y.; Tanikawa, M.; Sone, K.; Hirano, M.; Harada, M.; et al. Activation of NRF2/KEAP1 pathway by oral dimethylfumarate administration alleviates oxidative stress and age-associated infertility might be delayed in the mouse ovary. Reprod. Biol. Endocrinol. 2019, 17, 23.
- Gopal, S.; Mikulskis, A.; Gold, R.; Fox, R.J.; Dawson, K.T.; Amaravadi, L. Evidance of activation of the NRF2 pathway in multiple sclerosis patients treated with delayed –release dimethyl fumarate in the phase 3 define and confirm studies. Mult. Scler. J. 2017, 2017, 1875–1883.
- Ahuja, M.; Kaidery, N.A.; Yang, L.; Calingasan, N.; Smirnova, N.; Gaisin, A.; Gaisina, I.N.; Gazaryan, I.; Hushpulian, D.M.; Kaddour-Djebbar, I.; et al. Distinct NRF2 signaling mechanisms of fumaric acid esters and their role in neuroprotection against 1-methyl-4-phenyl-1,2,3,6- tetrahydropyridine-induced experimental Parkinson’s like disease. J. Neurosci. 2016, 36, 6332–6351.
- Rosto, M.; Testi, C.; Parisi, G.; Cortese, B.; Baiocco, P.; Di Angelantonio, S. Exploring the use of dimethyl fumarate as microglia modulator for neurodegenerative diseases treatment. Antioxidants 2020, 9, 700.
- Egbujor, M.C.; Okoro, U.C.; Egu, A.S.; Okonkwo, V.I.; Okafor, S.N.; Emeruwa, C.N.; Egwuatu, P.I.; Umeh, O.R.; Eziafakaego, M.I.; Amasiatu, I.S.; et al. Synthesis and biological evaluation of sulfamoyl carboxamide derivatives from sulfur-containing α-amino acids. Chiang Mai J. Sci. 2022, 49, 1100–1115.
- Strating, J. Organic Sulfur Compounds, 1st ed.; Pergamon Press: Oxford, UK, 1961.
- Egbujor, M.C.; Okoro, U.C.; Okafor, S. Design, synthesis, molecular docking, antimicrobial and antioxidant activities of new phenylsulfamoyl carboxylic acids of pharmacological interest. Med. Chem. Res. 2019, 28, 2118–2127.
- Egbujor, M.C.; Okoro, U.C. New methionine-based p-toluenesulphonamoyl carboxamide derivatives as antimicrobial and antioxidant agents: Design, synthesis. J. Pharm. Res. Int. 2019, 28, 1–12.
- Egbujor, M.C.; Okoro, U.C.; Okafor, S.; Nwankwo, N.E. Design, synthesis and molecular docking of novel serine-based sulphonamide bioactive compounds as potential antioxidant and antimicrobial agents. Attach. Am. J. Pharm. Sci. 2019, 6, 12232–12240.
- Abdel-Aziz, A.A.; Angeli, A.; El-Azab, A.S.; Hammouda, M.E.A.; El-Sherbeny, M.A.; Supuran, C.T. Synthesis and anti-inflammatory activity of sulphonamides and carboxylates incorporating trimellitimides: Dual cyclooxygenase/carbonic anhydrase inhibitory actions. Bioorganic Chem. 2019, 84, 260–268.
- Simpkins, N.S. Sulfone in Organic Synthesis, 1st ed.; Pergamon Press: Oxford, UK, 1993.
- Forristal, I. The chemistry of α,β-unsaturated sulfoxides and sulfones: An update. J. Sulfur Chem. 2005, 26, 163–195.
- Palmer, J.T.; Rasnick, D.; Klaus, J.L.; Bromme, D. Vinyl sulfones as mechanism-based cysteine protease inhibitor. J. Med. Chem. 1995, 38, 3193–3196.
- Liu, S.; Hanzlik, R.P. Structure-activity relationship for inhibition of papain by peptide Michael acceptors. J. Med. Chem. 1992, 35, 1067–1075.
- Engel, J.C.; Doyle, P.S.; Hsieh, I.; Mckerrow, J.H. Cysteine protease inhibitors cure an exprermental trypanosome cruzi infection. J. Exp. Med. 1998, 188, 725–734.
- Frankel, B.A.; Bentley, M.; Kruger, R.G.; McCatterty, D.G. Vinyl sulfones: Inhibitors of SrtA, a transpeptidase required for cell wall protein anchoring and virvlence in staphylococcus aureus. J. Am. Chem. Soc. 2004, 126, 3404–3405.
- Roush, W.R.; Gwaltney, S.L.; Cheng, J.; Scheidt, K.A.; Mckerrow, J.H.; Hansell, E. Vinyl sulfonates esters and vinyl sulphonamides: Potent, irreversible inhibitors of cystein proteases. J. Am. Chem. Soc. 1998, 120, 10994–10995.
- Kover, G.E.; Kam, C.M.; Powers, J.C.; Hudig, D. Dipeptide vinyl sulfones suitable for intracellular inhibition of dipeptidyl peptidase I. Int. J. Immunopharmacol. 2001, 1, 21–32.
- Kam, C.M.; Gotz, M.G.; Koot, G.; McGuire, M.; Thiele, D.; Hudig, D.; Powers, J.C. Design and evaluation of inhibitors for dipeptidyl peptidase I (Cathepsin C). Arch. Biochem. Biophys. 2004, 427, 123–134.
- Woo, S.Y.; Kim, J.H.; Moon, M.K.; Han, S.-H.; Yeon, S.K.; Choi, J.W.; Jang, B.K.; Song, H.J.; Kang, Y.G.; Kim, J.W.; et al. Discovery of vinyl sulfones as novel class of neuroprotective agents towards Parkinson’s disease therapy. J. Med. Chem. 2014, 57, 1473–1487.
- Smith, D.A.; Cucurull-Sanchez, L. The adaptive in combo strategy. In Comprehensive Medicinal Chemistry II; Pfizer Global Research and Developmental: Sandwich, UK, 2007; Volume 5, pp. 957–969.
- Song, Z.-L.; Hou, Y.; Bai, F.; Fang, J. Generation of potent NRF2 activators via tuning the electrophilicity and steric hinderance of vinyl sulfones for neuroprotection. Bioorganic Chem. 2020, 107, 104520.
- Egbujor, M.C.; Okoro, U.C.; Okafor, S. Novel alanine-based antimicrobial and antioxidant agents: Synthesis and molecular docking. Indian J. Sci. Technol. 2020, 13, 1003–1014.
- Egbujor, M.C.; Okoro, U.C.; Nwobodo, D.C.; Ezeagu, C.U.; Amadi, U.B.; Okenwa-Ani, C.G.; Ugwu, J.I.; Okoye, I.G.; Abu, I.P.; Egwuatu, P.I. Design, synthesis, antimicrobial and antioxidant activities of novel threonine-based sulfonamide derivatives. J. Pharm. Res. Int. 2020, 32, 51–61.
- Onoabedje, E.A.; Ibezim, A.; Okoro, U.C.; Batra, S. Synthesis, molecular docking, antiplasmodial and antioxidant activities of new sulfonamido-peptode derivatives. Heliyon 2020, 6, e04958.
- Egbujor, M.C.; Okoro, U.C.; Egu, S.A.; Egwuatu, P.I.; Eze, F.U.; Amasiatu, I.S. Design, synthesis and biological evaluation of alanine-based sulphonamide derivatives. Int. J. Res. Pharm. Sci. 2020, 11, 6449–6458.
- Egbujor, M.C.; Okoro, U.C.; Okafor, S.; Nwankwo, N.E. Synthesis, characterization and in silico studies of novel alkanoylated 4-methylphenyl sulphonamoyl carboxylic acids as potential antimicrobial and antioxidant agents. Int. J. Pharm. Phytopharm. Res. 2019, 9, 89–97.
- Egbujor, M.C.; Egu, S.A.; Okonkwo, V.I.; Jacob, A.D.; Egwuatu, P.I.; Amasiatu, I.S. Antioxidant drug design:historical and recent developments. J. Pharm. Res. Int. 2021, 32, 36–56.
- Egbujor, M.C.; Okoro, U.C.; Okafor, S.N.; Amasiatu, I.S.; Amadi, U.B.; Egwuatu, P.I. Synthesis, molecular docking and pharmacological evaluation of new 4-methylphenylsulphamoyl carboxylic acids analogs. Int. J. Res. Pharm. Sci. 2020, 11, 5357–5366.
- Luan, F.; Wei, L.; Zhang, J.; MI, Y.; Dong, F.; Li, Q.; Guo, Z. Autioxidant activity of chitosan derivatives with propane sulfonate groups. Polymer 2018, 10, 395.
- Arshia, A.J.; Faheem, A.; Khan, K.M.; Shah, S.; Perveen, S. Benzophenone esters and sulfonates synthesis and their potential as anti-inflammatory agents. Med. Chem. 2019, 15, 162–174.
- Gilchrist, T.L. Synthesis: Carbon with three or four attached heteroatoms. In Comprehensive Organic Functional Group Transformations; Elsevier Science: Oxford, UK, 2017; Volume 6.
- Otacka, S.; Kwiatknowska, M.; Madalinska, L.; Kielbasinski, P. Chiral organosulfur ligands/catalysts with a stereogenic sulfur atom: Applications in asymmetric synthesis. Chem. Rev. 2017, 117, 414–4181.
- Elisia, I.; Nakamura, H.; Lam, V.; Hofs, E.; Cederberg, R.; Cait, J.; Hughes, M.R.; Lee, L.; Jia, W.; Adomat, H.H.; et al. DMSO represses inflammatory cytokine production from human blood cells and reduces autoimmune arthritis. PLoS ONE 2016, 11, e0152538.
- Sanmartin-Suarez, C.; Soto-Otero, R.; Sanchez-Sellero, I.; Mendez-Alvarez, E. Antioxidant properties of dimethyl sulfoxide and its viability as a solvent in the evaluation of neuroprotective antioxidants. J. Pharmacol. Toxicol. Methods 2011, 63, 209–215.
- Forristal, I.; Rayner, C.M. Advances in Sulfur Chemistry. In Recent Advances in the Chemistry of α,β-Unsaturated Sulfoxides and Sulfones; Rayner, C.M., Ed.; JAI Press: Greenwich, CT, USA, 2000; Volume 2, pp. 155–213.
- Posner, G.H. Asymmetric Synthesis; Morrison, J., Ed.; Academic Press: New York, NY, USA, 1983; Volume 2, p. 225.
- Lyzwa, P.; Jankowiak, A.; Kwiatowska, M.; Mikolajczyk, M.; Kielbasinski, P.; Betz, A.; Jaffres, P.-A.; Gavmont, A.-C.; Gulea, M. Diastereoselective Michael additions to α,β-unsaturated sulfinyl phosphonates in the thiolane series. Tetrahedron Lett. 2007, 48, 351–355.
- Unnikrishria, M.K.; Rao, M.N.A. Anti-inflammatory activity of methionine, methionine sulfoxide and methionone sulfone. Agents Actions 1990, 31, 110–112.
