Sodium-glucose cotransporter 2 (SGLT2) inhibitors are a novel class of oral hypoglycemic agents which increase urinary glucose excretion by suppressing glucose reabsorption at the proximal tubule in the kidney. SGLT2 inhibitors lower glycated hemoglobin (HbA1c) by 0.6–0.8% (6–8 mmol/mol) without increasing the risk of hypoglycemia and induce weight loss and improve various metabolic parameters including blood pressure, lipid profile and hyperuricemia.
- sodium-glucose cotransporter 2 inhibitor
- type 2 diabetes
- cardiovascular outcome trial
- cardiorenal protection
1. Introduction
|
Generic Name |
Dosage |
SGLT1/2 Selectivity |
Half-Life (t1/2) |
Indication |
|---|---|---|---|---|
|
Ipragliflozin |
50–100 mg once daily |
254:1 |
15 h |
Type 1 and type 2 diabetes |
|
Dapagliflozin |
5–10 mg once daily |
1242:1 |
8–12 h |
Type 1 and type 2 diabetes |
|
Canagliflozin |
100 mg once daily |
155:1 |
12 h |
Type 2 diabetes |
|
Empagliflozin |
10–25 mg once daily |
2680:1 |
14–18 h |
Type 2 diabetes |
|
Luseogliflozin |
2.5–5.0 mg once daily |
1770:1 |
9 h |
Type 2 diabetes |
|
Tofogliflozin |
20 mg once daily |
2912:1 |
5 h |
Type 2 diabetes |
2. Mechanisms of Action of SGLT2 Inhibitors
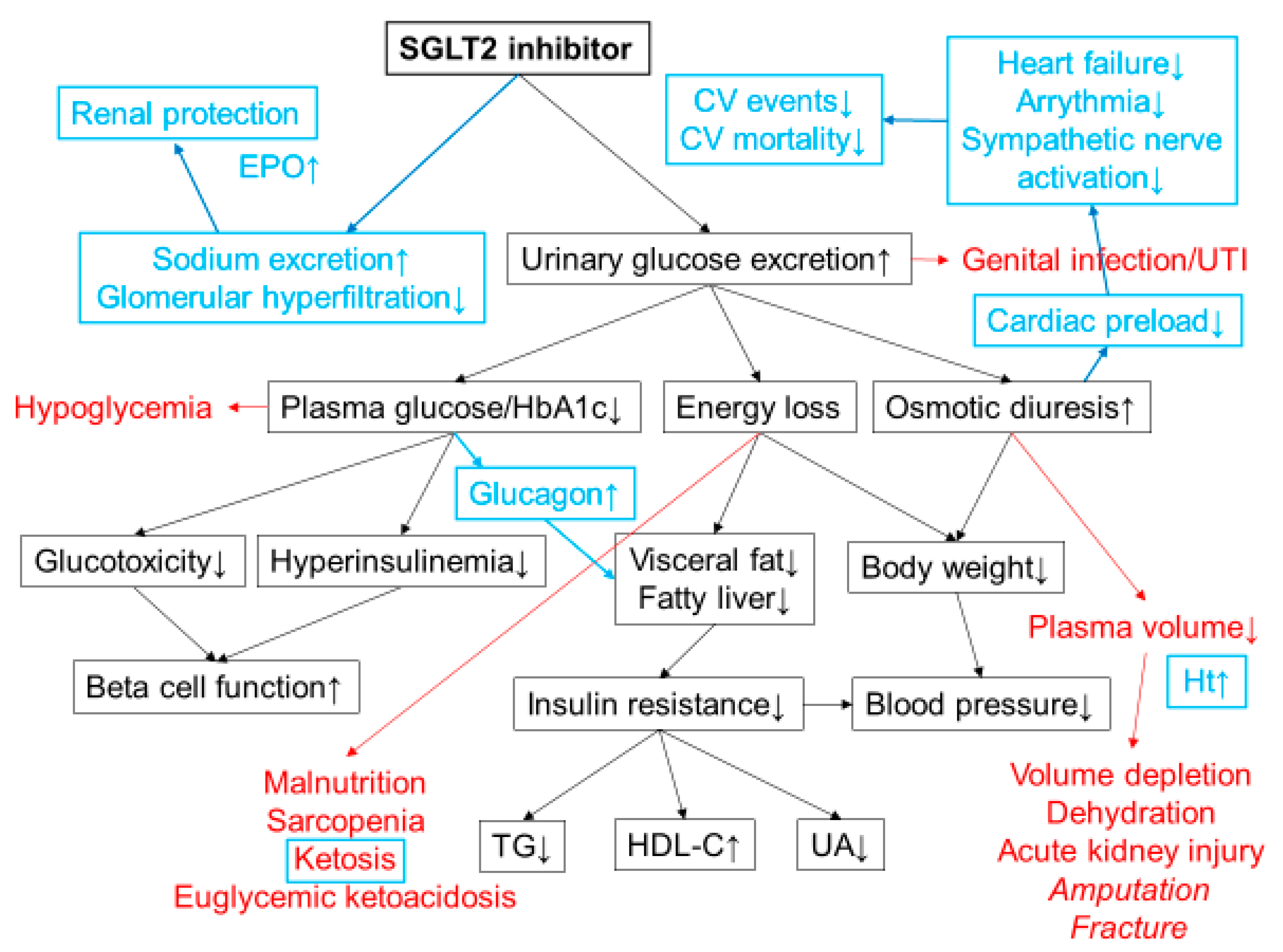
3. Clinical Efficacy of SGLT2 Inhibitors
3.1. Glucose-Lowering Effect
3.2. Body Weight, Blood Pressure, and Other Metablic Parameters
3.3. Hypoglycemia
3.4. Beta Cell Function
4. Adverse Effects
5. Positioning of SGLT2 Inhibitors in Treatment of T2DM
This entry is adapted from the peer-reviewed paper 10.3390/diseases8020014
References
- Ghezzi, C.; Loo, D.D.F.; Wright, E.M. Physiology of renal glucose handling via SGLT1, SGLT2 and GLUT2. Diabetologia 2018, 61, 2087–2097.
- Abdul-Ghani, M.A.; DeFronzo, R.A.; Norton, L. Novel hypothesis to explain why sglt2 inhibitors inhibit only 30-50% of filtered glucose load in humans. Diabetes 2013, 62, 3324–3328.
- Poulsen, S.B.; Fenton, R.A.; Rieg, T. Sodium-glucose cotransport. Curr. Opin. Nephrol. Hypertens. 2015, 24, 463–469.
- DeFronzo, R.A.; Hompesch, M.; Kasichayanula, S.; Liu, X.; Hong, Y.; Pfister, M.; Morrow, L.A.; Leslie, B.R.; Boulton, D.W.; Ching, A.; et al. Characterization of renal glucose reabsorption in response to dapagliflozin in healthy subjects and subjects with type 2 diabetes. Diabetes Care 2013, 36, 3169–3176.
- Norton, L.; Shannon, C.E.; Fourcaudot, M.; Hu, C.; Wang, N.; Ren, W.; Song, J.; Abdul-Ghani, M.; DeFronzo, R.A.; Ren, J.; et al. Sodium-glucose co-transporter (sglt) and glucose transporter (glut) expression in the kidney of type 2 diabetic subjects. Diabetes Obes. Metab. 2017, 19, 1322–1326.
- Nagahisa, T.; Saisho, Y. Cardiorenal protection: Potential of sglt2 inhibitors and glp-1 receptor agonists in the treatment of type 2 diabetes. Diabetes Ther. 2019, 10, 1733–1752.
- Polidori, D.; Sha, S.; Mudaliar, S.; Ciaraldi, T.P.; Ghosh, A.; Vaccaro, N.; Farrell, K.; Rothenberg, P.; Henry, R.R. Canagliflozin lowers postprandial glucose and insulin by delaying intestinal glucose absorption in addition to increasing urinary glucose excretion: Results of a randomized, placebo-controlled study. Diabetes Care 2013, 36, 2154–2161.
- Takebayashi, K.; Hara, K.; Terasawa, T.; Naruse, R.; Suetsugu, M.; Tsuchiya, T.; Inukai, T. Effect of canagliflozin on circulating active glp-1 levels in patients with type 2 diabetes: A randomized trial. Endocr. J. 2017, 64, 923–931.
- Neuen, B.L.; Young, T.; Heerspink, H.J.L.; Neal, B.; Perkovic, V.; Billot, L.; Mahaffey, K.W.; Charytan, D.M.; Wheeler, D.C.; Arnott, C.; et al. SGLT2 inhibitors for the prevention of kidney failure in patients with type 2 diabetes: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2019, 7, 845–854.
- Jardine, M.J.; Zhou, Z.; Mahaffey, K.W.; Oshima, M.; Agarwal, R.; Bakris, G.; Bajaj, H.S.; Bull, S.; Cannon, C.P.; Charytan, D.M.; et al. Renal, cardiovascular, and safety outcomes of canagliflozin by baseline kidney function: A secondary analysis of the credence randomized trial. J. Am. Soc. Nephrol. 2020, 31, 1128–1139.
- Buse, J.B.; Wexler, D.J.; Tsapas, A.; Rossing, P.; Mingrone, G.; Mathieu, C.; D’Alessio, D.A.; Davies, M.J. 2019 update to: Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the american diabetes association (ADA) and the european association for the study of diabetes (EASD). Diabetologia 2020, 63, 221–228.
- Vasilakou, D.; Karagiannis, T.; Athanasiadou, E.; Mainou, M.; Liakos, A.; Bekiari, E.; Sarigianni, M.; Matthews, D.R.; Tsapas, A. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: A systematic review and meta-analysis. Ann. Intern. Med. 2013, 159, 262–274.
- Nishimura, R.; Osonoi, T.; Kanada, S.; Jinnouchi, H.; Sugio, K.; Omiya, H.; Ubukata, M.; Sakai, S.; Samukawa, Y. Effects of luseogliflozin, a sodium-glucose co-transporter 2 inhibitor, on 24-h glucose variability assessed by continuous glucose monitoring in japanese patients with type 2 diabetes mellitus: A randomized, double-blind, placebo-controlled, crossover study. Diabetes Obes. Metab. 2015, 17, 800–804.
- Henry, R.R.; Strange, P.; Zhou, R.; Pettus, J.; Shi, L.; Zhuplatov, S.B.; Mansfield, T.; Klein, D.; Katz, A. Effects of dapagliflozin on 24-hour glycemic control in patients with type 2 diabetes: A randomized controlled trial. Diabetes Technol. Ther. 2018, 20, 715–724.
- Hayashi, T.; Fukui, T.; Nakanishi, N.; Yamamoto, S.; Tomoyasu, M.; Osamura, A.; Ohara, M.; Yamamoto, T.; Ito, Y.; Hirano, T. Dapagliflozin decreases small dense low-density lipoprotein-cholesterol and increases high-density lipoprotein 2-cholesterol in patients with type 2 diabetes: Comparison with sitagliptin. Cardiovasc. Diabetol. 2017, 16, 8.
- McGill, J.B.; Subramanian, S. Safety of sodium-glucose co-transporter 2 inhibitors. Am. J. Cardiol. 2019, 124, S45–S52.
- Danne, T.; Garg, S.; Peters, A.L.; Buse, J.B.; Mathieu, C.; Pettus, J.H.; Alexander, C.M.; Battelino, T.; Ampudia-Blasco, F.J.; Bode, B.W.; et al. International consensus on risk management of diabetic ketoacidosis in patients with type 1 diabetes treated with sodium–glucose cotransporter (sglt) inhibitors. Diabetes Care 2019, 42, 1147–1154.
- Takahara, M.; Shiraiwa, T.; Matsuoka, T.A.; Katakami, N.; Shimomura, I. Ameliorated pancreatic beta cell dysfunction in type 2 diabetic patients treated with a sodium-glucose cotransporter 2 inhibitor ipragliflozin. Endocr. J. 2015, 62, 77–86.
- Polidori, D.; Mari, A.; Ferrannini, E. Canagliflozin, a sodium glucose co-transporter 2 inhibitor, improves model-based indices of beta cell function in patients with type 2 diabetes. Diabetologia 2014, 57, 891–901.
- Saisho, Y. Changing the concept of type 2 diabetes: Beta cell workload hypothesis revisited. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 121–127.
- Saisho, Y. Beta cell dysfunction: Its critical role in prevention and management of type 2 diabetes. World J. Diabetes 2015, 6, 109–124.
- Saisho, Y. How can we develop more effective strategies for type 2 diabetes mellitus prevention? A paradigm shift from a glucose-centric to a beta cell-centric concept of diabetes. EMJ Diabet 2018, 6, 46–52.
- Ferrannini, E.; Muscelli, E.; Frascerra, S.; Baldi, S.; Mari, A.; Heise, T.; Broedl, U.C.; Woerle, H.J. Metabolic response to sodium-glucose cotransporter 2 inhibition in type 2 diabetic patients. J. Clin. Investig. 2014, 124, 499–508.
- Merovci, A.; Solis-Herrera, C.; Daniele, G.; Eldor, R.; Fiorentino, T.V.; Tripathy, D.; Xiong, J.; Perez, Z.; Norton, L.; Abdul-Ghani, M.A.; et al. Dapagliflozin improves muscle insulin sensitivity but enhances endogenous glucose production. J. Clin. Investig. 2014, 124, 509–514.
- Sheu, W.H.H.; Chan, S.P.; Matawaran, B.J.; Deerochanawong, C.; Mithal, A.; Chan, J.; Suastika, K.; Khoo, C.M.; Nguyen, H.M.; Linong, J.; et al. Use of sglt-2 inhibitors in patients with type 2 diabetes mellitus and abdominal obesity: An asian perspective and expert recommendations. Diabetes Metab. J. 2020, 44, 11–32.
- Bonner, C.; Kerr-Conte, J.; Gmyr, V.; Queniat, G.; Moerman, E.; Thevenet, J.; Beaucamps, C.; Delalleau, N.; Popescu, I.; Malaisse, W.J.; et al. Inhibition of the glucose transporter sglt2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat. Med. 2015, 21, 512–517.
- Saponaro, C.; Mühlemann, M.; Acosta-Montalvo, A.; Piron, A.; Gmyr, V.; Delalleau, N.; Moerman, E.; Thévenet, J.; Pasquetti, G.; Coddeville, A.; et al. Interindividual heterogeneity of sglt2 expression and function in human pancreatic islets. Diabetes 2020, 69, 902–914.
- Suga, T.; Kikuchi, O.; Kobayashi, M.; Matsui, S.; Yokota-Hashimoto, H.; Wada, E.; Kohno, D.; Sasaki, T.; Takeuchi, K.; Kakizaki, S.; et al. Sglt1 in pancreatic alpha cells regulates glucagon secretion in mice, possibly explaining the distinct effects of sglt2 inhibitors on plasma glucagon levels. Mol. Metab. 2019, 19, 1–12.
- Kuhre, R.E.; Ghiasi, S.M.; Adriaenssens, A.E.; Wewer Albrechtsen, N.J.; Andersen, D.B.; Aivazidis, A.; Chen, L.; Mandrup-Poulsen, T.; Orskov, C.; Gribble, F.M.; et al. No direct effect of sglt2 activity on glucagon secretion. Diabetologia 2019, 62, 1011–1023.
- Bersoff-Matcha, S.J.; Chamberlain, C.; Cao, C.; Kortepeter, C.; Chong, W.H. Fournier gangrene associated with sodium-glucose cotransporter-2 inhibitors: A review of spontaneous postmarketing cases. Ann. Intern. Med. 2019, 170, 764–769.
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R.; et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N. Engl. J. Med. 2017, 377, 644–657.
- Perkovic, V.; Jardine, M.J.; Neal, B.; Bompoint, S.; Heerspink, H.J.L.; Charytan, D.M.; Edwards, R.; Agarwal, R.; Bakris, G.; Bull, S.; et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306.
- Wing, R.R.; Bolin, P.; Brancati, F.L.; Bray, G.A.; Clark, J.M.; Coday, M.; Crow, R.S.; Curtis, J.M.; Egan, C.M.; Espeland, M.A.; et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N. Engl. J. Med. 2013, 369, 145–154.
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2015, 373, 2117–2128.
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 2019, 380, 347–357.
- McMurray, J.J.V.; Solomon, S.D.; Inzucchi, S.E.; Kober, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; Belohlavek, J.; et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N. Engl. J. Med. 2019, 381, 1995–2008.
- Petrie, M.C.; Verma, S.; Docherty, K.F.; Inzucchi, S.E.; Anand, I.; Belohlavek, J.; Bohm, M.; Chiang, C.E.; Chopra, V.K.; de Boer, R.A.; et al. Effect of dapagliflozin on worsening heart failure and cardiovascular death in patients with heart failure with and without diabetes. JAMA 2020.
- Lam, C.S.P.; Chandramouli, C.; Ahooja, V.; Verma, S. Sglt-2 inhibitors in heart failure: Current management, unmet needs, and therapeutic prospects. J. Am. Heart Assoc. 2019, 8, e013389.
- Dekkers, C.C.J.; Gansevoort, R.T. Sodium-glucose cotransporter 2 inhibitors: Extending the indication to non-diabetic kidney disease? Nephrol. Dial. Transplant. 2020, 35, i33–i42.
- Taylor, S.I.; Blau, J.E.; Rother, K.I.; Beitelshees, A.L. Sglt2 inhibitors as adjunctive therapy for type 1 diabetes: Balancing benefits and risks. Lancet Diabetes Endocrinol. 2019, 7, 949–958.
- Sims, H.; Smith, K.H.; Bramlage, P.; Minguet, J. Sotagliflozin: A dual sodium-glucose co-transporter-1 and -2 inhibitor for the management of type 1 and type 2 diabetes mellitus. Diabet. Med. 2018, 35, 1037–1048.
- Cefalo, C.M.A.; Cinti, F.; Moffa, S.; Impronta, F.; Sorice, G.P.; Mezza, T.; Pontecorvi, A.; Giaccari, A. Sotagliflozin, the first dual sglt inhibitor: Current outlook and perspectives. Cardiovasc. Diabetol. 2019, 18, 20.
