In the human cornea, regeneration of the epithelium is regulated by the stem cell reservoir of the limbus, which is the marginal region of the cornea representing the anatomical and functional border between the corneal and conjunctival epithelium. In support of this concept, extensive limbal damage, e.g., by chemical or thermal injury, inflammation, or surgery, may induce limbal stem cell deficiency (LSCD) leading to vascularization and opacification of the cornea and eventually vision loss. These acquired forms of limbal stem cell deficiency may occur uni- or bilaterally, which is important for the choice of treatment. Moreover, a variety of inherited diseases, such as congenital aniridia or dyskeratosis congenita, are characterized by LSCD typically occurring bilaterally. Several techniques of autologous and allogenic stem cell transplantation have been established. The limbus can be restored by transplantation of whole limbal grafts, small limbal biopsies or by ex vivo-expanded limbal cells.
1. Introduction
In 1868, the term “stem cell” was first proposed in the worldwide scientific literature by the German biologist Ernst Haeckel
[1]. Stem cells are unspecialized cells with differentiation potential that can simultaneously self-renew, differentiate and generate any cell type in the organism, with slow cycling during homeostasis in vivo
[2][3]. Over the years, stem cells became attractive for therapeutic applications in many branches of medicine, and a growing amount of knowledge is being gathered in this field
[2]. Based on their differentiation potential, stem cells can be classified into totipotent, pluripotent, multipotent, oligopotent, and unipotent stem cells
[2]. Moreover, according to the stage of development, stem cells can be divided into embryonic and adult stem cells
[4]. Embryonic stem cells are pluripotent and derived from blastocysts
[5]. Adult stem cells exist in adult tissues and have less differentiation potential than embryonic stem cells
[6]. However, some adult forms of stem cells are thought to possess multipotent, oligopotent, and unipotent properties
[4][7]. Some adult stem cell types, such as bone marrow and dental pulp stem cells, are believed to even possess pluripotent properties
[8][9]. Under physiological conditions, adult stem cells, which can create progenitor cells, are usually quiescent
[10][11]. However, adult stem cells can be reactivated to proliferate for tissue regeneration under various extrinsic stimuli, such as stress and injury
[12]. In 1971, Davanger and Evensen proposed that renewal of the corneal epithelium is driven by migration of epithelial cells located at the limbus of the cornea
[13]. The hypothesis of limbal epithelial stem cells (LESCs) has been studied and proven in various in vivo and in vitro studies
[14][15][16]. Moreover, there is some evidence for stem-like and progenitor cells in the peripheral corneal endothelium
[17][18]. Furthermore, small populations of stem cells in the human corneal stroma have been identified
[19]. Taken together, not only LESCs but also corneal stromal stem cells and endothelial stem cells appear to exist in the cornea, which are all considered to play a vital role in maintaining corneal homeostasis and repair
[20][21][22][23].
The cornea is a transparent and refractive structure at the front of the eye that transmits and focuses light to the retina and lacks blood vessels
[24]. Hence, loss of corneal transparency results in severe visual impairment or blindness
[25]. The World Health Organization estimates that around 4.2 million worldwide are suffering from moderate to severe distance vision loss or blindness due to corneal opacities, which has become the fourth leading cause of visual impairment
[26]. There is a wide variety of corneal diseases that can cause corneal opacification or deformation, e.g., corneal trauma, microbial infection, neurotrophic keratopathy, endothelial dystrophy, or limbal stem cell deficiency, which can be acquired or inherited
[27][28][29][30].
Various corneal transplantation techniques have been developed to treat loss of corneal transparency in cases where conservative strategies are unavailable or unsuccessful
[31]. Due to the relative immune privilege of the cornea, the prognosis of corneal grafts is relatively good
[32]. However, transplantation of epithelial stem cells is challenging, since they are believed to be localized at the densely vascularized limbal region, which is highly immunogenic
[33]. Therefore, a variety of conservative strategies is under investigation to treat persistent corneal epithelial defects and limbal stem cell disease.
2. Localization of Limbal Epithelial Stem Cells
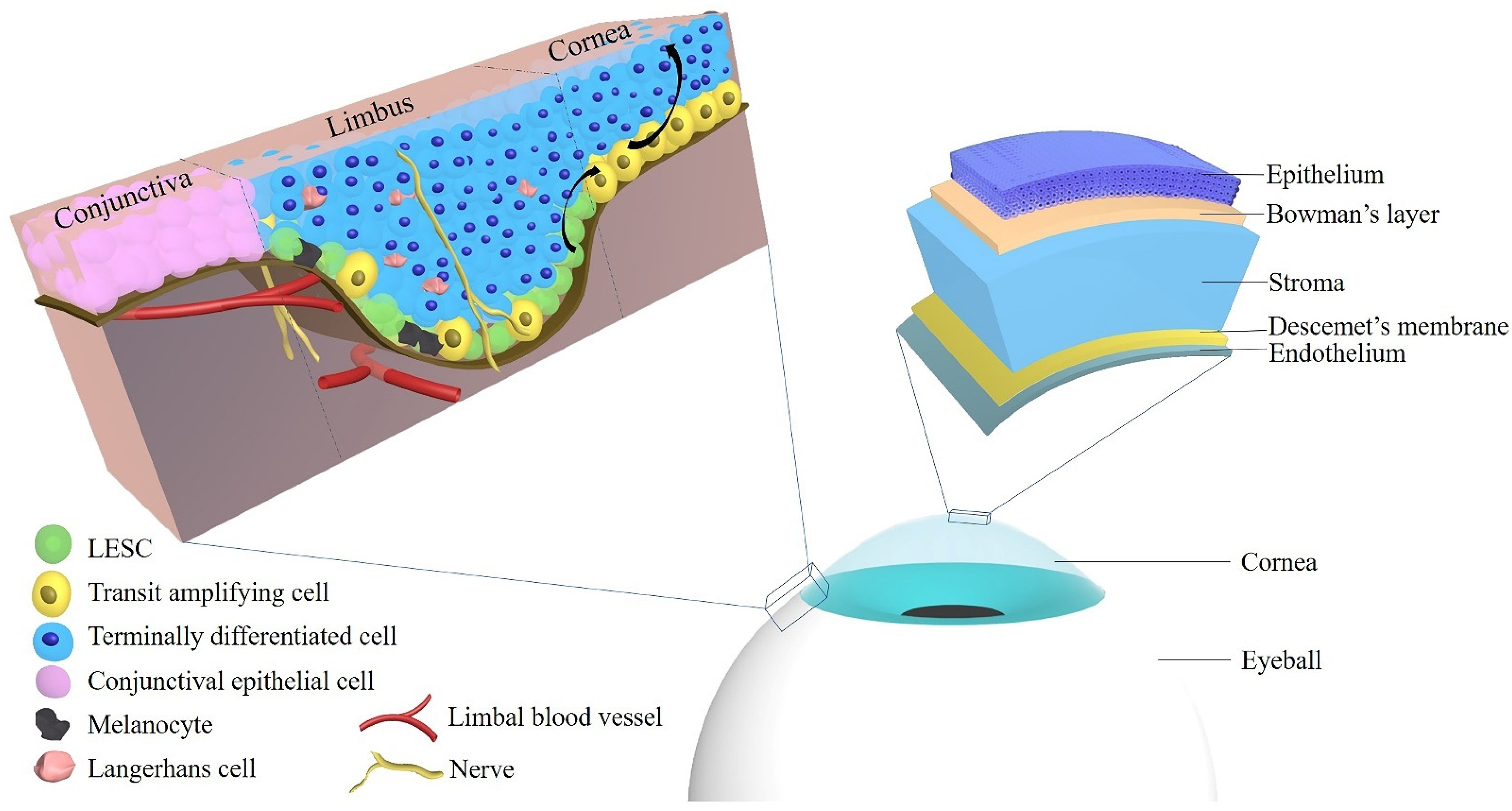
The cornea is composed of five layers: the epithelium, Bowman’s layer, stroma, Descemet’s membrane, and the endothelium, and each of those layers exerts an essential role in maintaining corneal transparency and stable visual function
[34]. In 2013, Dua et al. proposed a novel sixth layer in the human cornea located between the stroma and Descemet’s membrane
[35]. The authors suggested the terms “Dua’s layer”, “pre-Descemet’s layer (PDL)” and “pre-posterior limiting lamina layer” for this anatomical part of the cornea
[35][36]. Although there are still many controversies about this anatomical structure, it already had a significant impact on corneal research and therapy
[35][36][37][38][39][40]. Clear vision requires a concertedly functional cooperation of the individual corneal layers to maintain a smooth and transparent corneal surface
[33]. The corneal epithelium is the cornea’s outermost layer with five to seven cell layers that are together 50 to 60 μm thick
[15][34][41]. It consists of inner basal cells, middle wing cells and superficial squamous cells
[34]. The limbal corneal epithelium is composed of several layers of epithelial cells with Langerhans cells and melanocytes
[15]. The role of the corneal epithelium includes maintenance of corneal transparency and protection of the eye from the external environment
[42].
In 1983, Thoft and Friend proposed the X, Y, Z hypothesis of corneal epithelial maintenance
[43]. The authors suggested that proliferation of corneal basal epithelial cells (X) and centripetal movement of peripheral cells (Y) equals the epithelial cell loss from the corneal surface (Z) so that cell replacement may maintain and replace cell loss in the cornea
[43]. These peripheral cells reside exclusively in the limbal zone and simultaneously maintain a steady cell number by giving rise to transit amplifying cells (TAC)
[44]. By a scanning slit confocal microscope, investigators tested the hypothesis that cells migrate centripetally in the epithelial layer of the normal human anterior cornea
[45]. However, the origin of these corneal epithelial cells was not described in the X, Y, Z hypothesis in 1983. Thoft presented another assumption later that conjunctival cells in the limbus could supply peripheral corneal cells by crossing the limbus, which has been termed “conjunctival transdifferentiation” theory
[46]. In 1986, Schermer et al. used a new monoclonal antibody, AE5, in vivo and in culture to recognize a 64-kDa basic (K3) keratin, a specific corneal epithelial differentiation marker in an advanced stage of basal epithelial cells of the central cornea, but not in conjunctival cells
[47]. Therefore, Schermer et al. considered that not conjunctival cells, but corneal epithelial cells, are responsible for the maintenance of the corneal epithelium, and they suggested that corneal epithelial stem cells are located at the limbus, a zone between the cornea and the conjunctiva
[47]. By using the monoclonal antibody, 4G10.3, directed against a 50-kD protein and binding to the limbal basal cells, Chung et al. studied the localization of corneal epithelial stem cells in rats during development in life (from day one to six weeks of age)
[48]. The authors suggested that stem cells or stem-like cells are localized throughout the basal layer of the corneal and limbal epithelium from day 1 through the whole adult life
[48]. These epithelial stem cells are sequestered in the limbus when the cornea matures
[48]. Two positive (p63, CK14) and one negative stem cell marker (CK3) have been used to identify the location of corneal epithelial stem cells in horses
[49]. The results revealed that equine corneal epithelial stem cells are localized in the epithelial basal layer of the corneal limbus, which is rich in resident melanocytes. Hence, melanocytes might serve as an indicator of the collecting area of corneal epithelial stem cells
[49]. Moreover, a member of the ATP binding cassette (ABC) transporters, ABCG2, was shown to be expressed by limbal basal cells
[50]. In a rabbit model, in which the limbal epithelium was surgically removed, recurrent erosions and corneal vascularization with delayed healing occurred whereas the controls healed normally
[16]. These signs could be related to limbal stem cell dysfunction
[16].
Figure 1 shows the anatomy of the cornea and limbus and the localization of LESCs.
Figure 1. The localization of limbal epithelial stem cells and the anatomy of the cornea. Abbreviations: LESC: limbal epithelial stem cell.
Pellegrini et al. proposed that the location of stem cells can be strongly supported by the evaluation of their cell division capacity, even though some corneal differentiation markers for locating epithelial stem cells have been reported
[15]. By serially cultivating cells from the ocular surface, Pellegrini et al. found that the cells from nine limbal biopsies underwent 85 doublings, whereas cells from central cornea could not be serially cultivated, indicating that cells in the limbal region of the corneal epithelium have a high proliferative capacity, indicative of stem cell existence
[15]. Ebato et al. compared the mitotic rate of cells from the corneal limbus, the peripheral epithelium, and the central epithelium in tissue culture
[51][52]. The results indicated that the mitotic rate of outgrowths of limbal cells was significantly higher than that of peripheral epithelium, which in turn was higher than that of the central epithelium
[51][52]. Altogether, the existing data strongly suggest that corneal epithelial stem cells reside in the basal layer of the corneal limbal epithelium
[53]. The stem cells are believed to be especially rich at the limbal palisades of Vogt, which are radially oriented fibrovascular ridges especially concentrated along the superior and inferior limbus
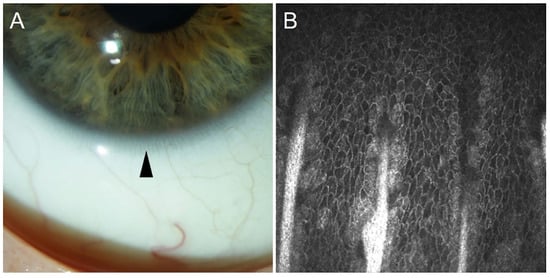
[54]. Images of Vogt palisades taken by slit lamp and in vivo confocal microscopy are depicted in
Figure 2.
Figure 2. The limbal palisades of Vogt taken by slit lamp (arrowhead, (A)) and by in vivo confocal microscopy (Heidelberg Engineering GmbH, Heidelberg, (B)).
Based on studies using in vivo confocal microscopy and three-dimensional (3D) imaging, Shortt et al. modeled the regional distribution and structure of the corneal epithelial stem cell niche, and indicated how epithelial stem cells can be located and observed in vivo in humans
[55]. Their results suggested that corneal epithelial stem cells line the edges and bases of limbal crypts and are also located around the sides and tips of focal stromal projections, surrounded by a complex vascular plexus
[55]. Their findings reveal three different stem cell niches: limbal epithelial crypts, limbal crypts, and focal stromal projections
[55]. The limbal crypts are most densely distributed in the superior and inferior limbal quadrants
[55].
3. Limbal Epithelial Stem Cell Markers
Several potential markers have been used to identify LESCs, which will target the emerging therapeutic topics. It is therefore useful to summarize these markers of LESCs.
3.1. p63
The p63 gene produces full-length (TAp63) and N-terminally truncated (ΔNp63) transcripts that can be spliced to encode three different p63 isoforms: p63α, p63β, and p63γ
[56][57]. The p63 protein was found in various human tissues and is a transcription factor regulating the progression of the cell through its cycle and cell death in response to environmental stimuli
[56]. Pellegrini et al. have shown that the nuclear p63 transcription factor (ΔNp63α), a p53 homolog related to epithelial regenerative proliferation, is expressed by basal cells of the limbal epithelium in vivo and in vitro, but not by TACs
[58][59]. The p63β and p63γ isoforms, which are absent in the resting limbus, are inclined to participate in epithelial differentiation during corneal regeneration
[60]. Some scientists suggest that evaluation of p63 in cultivated limbal epithelial cell sheets is vital for assessing and selecting the quality of the cell sheet
[61]. In addition, p63 knockout mice lack all stratified squamous epithelia and do not express differentiation markers
[62]. In a clinical study, Rama et al. cultivated limbal stem cells on fibrin to treat 112 patients with corneal damage
[63]. Their results showed that successful transplantation of patients was associated with the percentage of p63-bright holoclone-forming stem cells in culture
[63]. Via performing in situ hybridization with [
35S]-labeled sense and antisense p63α RNA riboprobes, strong p63 mRNA signals were found only in the basal layer of limbal epithelia
[64].
3.2. ABCG2
ABCG2, also known as breast cancer resistance protein 1 (BCRP1), is a member of the ABC family, as a universal marker for stem cells in many tissues
[65]. For example, ABCG2/BCRP1 expression is related to the side population (SP) phenotype based on the ability to efflux Hoechst 33342 dye, as a marker for hematopoietic stem cells
[65]. The limbal epithelium contains SP cells that express ABCG2, which are considered LESCs
[66]. By immunocytochemistry and quantitative PCR, ABCG2 was detected in the basal layer of the limbal epithelium
[64][67]. Moreover, ABCG2 mRNA was shown to be expressed at low levels by corneal epithelium and at higher levels by limbal epithelium
[50]. As shown by flow cytometry, ABCG2-positive limbal basal cells exhibited greater colony-forming efficiency than corneal epithelium and possessed stem cell properties on a 3T3 fibroblast feeder layer (mouse embryo fibroblast)
[50]. In an in vitro study, Kethiri et al. elucidated the ideal limbal explant size and number for limbal transplantation by assessing the expression of ABCG2
[68]. Their findings revealed that a minimum amount of 0.3 mm
2 vital tissue would be adequate for ample limbal cell expansion, and that larger cadaveric explants (≥0.5 mm
2) had a similar growth rate and proliferative potential to the live tissue
[68]. In an ABCG2 knockout mouse model, cultured corneal epithelial cells lacking ABCG2 had an increased susceptibility to oxidative damage induced by mitoxantrone and hypoxia, suggesting that ABCG2 may protect corneal epithelial cells by exerting anti-oxidative effects
[69]. In one study, pieces of the limbus from the donor’s limbal zone have been cultured. From these pieces, ABCG2-positive limbal stem cells could be expanded as cell sheets after three weeks of culturing on amniotic membrane. The cells could also be cryopreserved
[70].
3.3. Growth Factor Receptors
Based on the theory that the epidermal growth factor receptor (EGFR) is localized in various stratified squamous epithelia, Zieske and Wasson studied the growth factor receptor in the rat cornea in 1993
[71]. The authors showed by an antibody binding to the EGFR that staining was more intense in basal cells from the corneal limbal zone
[71]. In studies on human ocular surface epithelia, EGFR immunostaining was observed in basal corneal epithelial cells
[72]. Keratinocyte growth factor (KGF) is a mesenchymal-cell-derived paracrine growth factor, which is consistently more produced by limbal fibroblasts than by central corneal fibroblasts
[73]. KGF stimulates the differentiation, proliferation, and migration of limbal epithelial cells via a KGF paracrine loop, inducing ΔNp63α expression
[74]. In a human in vitro limbal epithelial cell model, KGF induced higher expression of the epithelial stem marker, ΔNp63α, compared with epidermal growth factor
[74]. Moreover, it has recently been shown that limbal epithelial cells cultured with KGF and the rho kinase inhibitor, Y-27632, could maintain the expression of several LESC markers, which may become an improved in vitro model in regenerative medicine
[75]. Furthermore, the keratinocyte growth factor receptor (KGFR) has been mainly expressed on the mRNA level by cultured limbal epithelial cells
[76].
3.4. Integrins
The integrins, consisting of individual α and β subunits, are the primary metazoan receptors that are vital for a metazoan existence and play critical roles in mediating cell adhesion
[77]. There are several integrin subunits reported to be expressed in the corneal epithelium, including β1, β4, β5, α2, α3, α5, α6, and αv
[78]. The β1 integrin subunit is mainly located at the membranes of basal epithelial cells in the cornea, and anti-β4 immunofluorescence was only shown in the basal part of the epithelium
[79]. Pajoohesh-Ganji et al. reported that limbal basal cells at the superior and inferior quadrants of adult mouse corneas expressed higher levels of β1 and β4 integrin and less α9 integrin than limbal basal cells at the nasal and temporal quadrants
[80]. Hayashi et al. reported that LESCs could be enriched by integrin α6 and the transferrin receptor (CD71), localized immunohistochemically in the basal region of the limbal epithelium, suggesting that α6 integrin and CD71 are the cell surface markers of LESCs
[81]. After mesenchymal stem cell transplantation in an experimental rabbit model of LSCD, expression of β1 integrin and connexin 43 (Cx43) were used as indicators for putative LESCs
[82][83]. These studies provided crucial evidence that corneal integrins are potential markers of LESCs and are essential for supporting the function of LESCs
[80][84][85][86].
3.5. Keratins
Keratins are the major structural proteins of epithelia, which play a critical role in the integrity and mechanical stability of epithelial cells and tissues
[87]. In addition, several keratins mediate tissue differentiation and participate in intracellular signaling under various pathologic conditions
[88]. Like epidermal epithelium, the corneal epithelium contains significant amounts of keratins, such as K3/K12, K5/K14, K5/K12, K8/K18, and K8/K19
[89][90][91]. However, K14 and K5 are not only located at the limbus but also reside in epithelial cells of the corneal center, suggesting that K5/K14 is an unreliable marker for LESCs
[92]. The corneal epithelial cells’ intermediate filament cytoskeleton comprises the cornea-specific keratins, K3 and K12
[93]. Additionally, RT-PCR studies revealed lower K3 and K12 mRNA levels in limbal epithelia than in the corneal epithelia
[64]. Other studies proved that the limbal basal cells lack the corneal keratins, K3 and K12
[47][94]. During corneal wound healing, K14 was expressed in niches at the limbal area seven days after birth in the mouse cornea
[95]. K8 and K19 were present in the limbal niches at day 21 after birth
[95]. The corneal epithelium showed enhanced K14 expression by day 28 after wounding, suggesting that LESCs migrate centripetally towards the central cornea
[95]. In contrast, the expression of K8 in limbal niches does not change with wound size
[96]. In 2019, Park et al. showed that K14
+ basal epithelia migrate into the wound bed by increased population pressure gradient from the limbus to the wound edge
[97]. Thus, the authors demonstrated that LESCs participate in corneal wound healing by using bromodeoxyuridine (BrdU) labeling to visualize their contribution in real-time
[97].
Table 1 lists the keratins in different species during the development and injury healing of the cornea.
Table 1. Corneal keratins in the development and wound healing of corneal and limbal epithelia.
| Keratins |
Species |
Stage |
Location |
References |
| K3 |
Rabbit |
21-day embryos |
The peridermal layer of the cornea |
[98] |
| 23-day embryos |
The suprabasal layer of the cornea |
| 7 to 12 days after birth |
The basal layer of the cornea |
| Adult |
The basal layer of the corneal epithelium and the suprabasal layer of the limbal epithelium |
| K4 |
Mouse |
16-, 18-, and 20-day embryos
2 and 4 days after birth |
The superficial layer of the cornea |
[94] |
| Adult |
Not observed |
| K5/K12 |
Mouse |
7 days after birth |
The basal and apical cells of the central and peripheral cornea and limbus |
[95] |
| K12 |
Rabbit |
17-day embryos |
The peridermal layer of the cornea |
[98] |
| 23-day embryos |
The basal layer of the cornea |
| Adult |
The basal layer of the corneal epithelium and the suprabasal layer of the limbal epithelium |
| K12 |
Chick |
12-day embryos |
Both peridermal and basal ectodermal layers of the cornea |
[98] |
| 14- to 21-day embryos |
All the epithelial strata of the
central cornea |
| 21-day embryos |
The suprabasal layer of the limbal epithelium |
| K12 |
Mouse |
15-day embryos |
The superficial layer of the corneal epithelium |
[94] |
| 18-day embryos |
The suprabasal layer of the cornea |
| 0 h after wounding |
The peripheral corneal epithelium |
[97] |
| 24 h after wounding |
Occasionally in superficial cells of the central and peripheral cornea |
| K14 |
Mouse |
15-day embryos |
Corneal epithelial cells |
[94] |
| 7 days after birth |
The basal layers over the entire mouse ocular surface with higher expression at the limbal region compared with the central cornea |
[95] |
| 49 days after birth |
The localization decreases in the central corneal epithelium but remains strong at the limbus. |
| 0 h after wounding |
Restricted to the limbus |
[97] |
| 24 h after wounding |
The layer of epithelial cells that covered the defect |
| 1 day after wounding |
Corneal epithelial cells at and behind the leading edge |
[95] |
| 7 days after wounding |
The corneal center |
| 28 days after wounding |
Cells adjacent to an erosion at the corneal center and
around goblet cell clusters at the limbus |
| K15 |
Mouse |
7 days after birth |
Limbal cells and well spread apical cells of the corneal periphery and center |
[95] |
| 49 days after birth |
The localization decreases in the central corneal epithelium but remains strong at the limbus. |
| 1 day after wounding |
Extending toward the leading edge |
| 7 days after wounding |
The corneal center |
| 28 days after wounding |
Cells adjacent to an erosion at the corneal center and
around goblet cell clusters at the limbus |
| K17 |
Human |
Within 24 h (for ex vivo) to 48 h after death |
Clusters of limbal basal cells in normal corneas and significantly decreased in diabetic limbal basal epithelium |
[99] |
| K18 |
Mouse |
14 days after birth |
The limbus |
[95] |
| 7 days after wounding, |
The center of the clusters |
| 28 days after wounding |
Compound niches |
| K8/K19 |
Mouse |
7 days after birth |
Basal and suprabasal cells throughout the corneal epithelium and limbus |
[95] |
| 14 days after birth |
Fewer cells on the central cornea and more restricted toward the peripheral and limbal region |
| 1 day after wounding |
K19 + clusters are migrating away from the limbus |
| 7 days after wounding |
K19 is localized to cells at the edges of large clusters |
| 28 days after wounding |
Near the limbal region and on the peripheral cornea |
[95][96] |
This entry is adapted from the peer-reviewed paper 10.3390/cells10092302