Saporin is a ribosome-inactivating protein that can cause inhibition of protein synthesis and causes cell death when delivered inside a cell. Development of commercial Saporin results in a technology termed ‘molecular surgery’, with Saporin as the scalpel. Its low toxicity (it has no efficient method of cell entry) and sturdy structure make Saporin a safe and simple molecule for many purposes. The most popular applications use experimental molecules that deliver Saporin via an add-on targeting molecule. These add-ons come in several forms: peptides, protein ligands, antibodies, even DNA fragments that mimic cell-binding ligands. Cells that do not express the targeted cell surface marker will not be affected.
- Saporin
- immunotoxins
- lesion
- Alzheimer’s
- pain
- RIP
- internalization
- animal model
- cancer
- Alzheimer’s disease
1. Introduction
1.1. RIPs in Cancer Research
1.2. RIPs in Neuroscience Research
2. Targeting Alzheimer’s Disease
2.1. 192-IgG-SAP
2.2. 192-IgG-SAP Species Specific Alternatives
3. Behavior, Disease and Animal Models
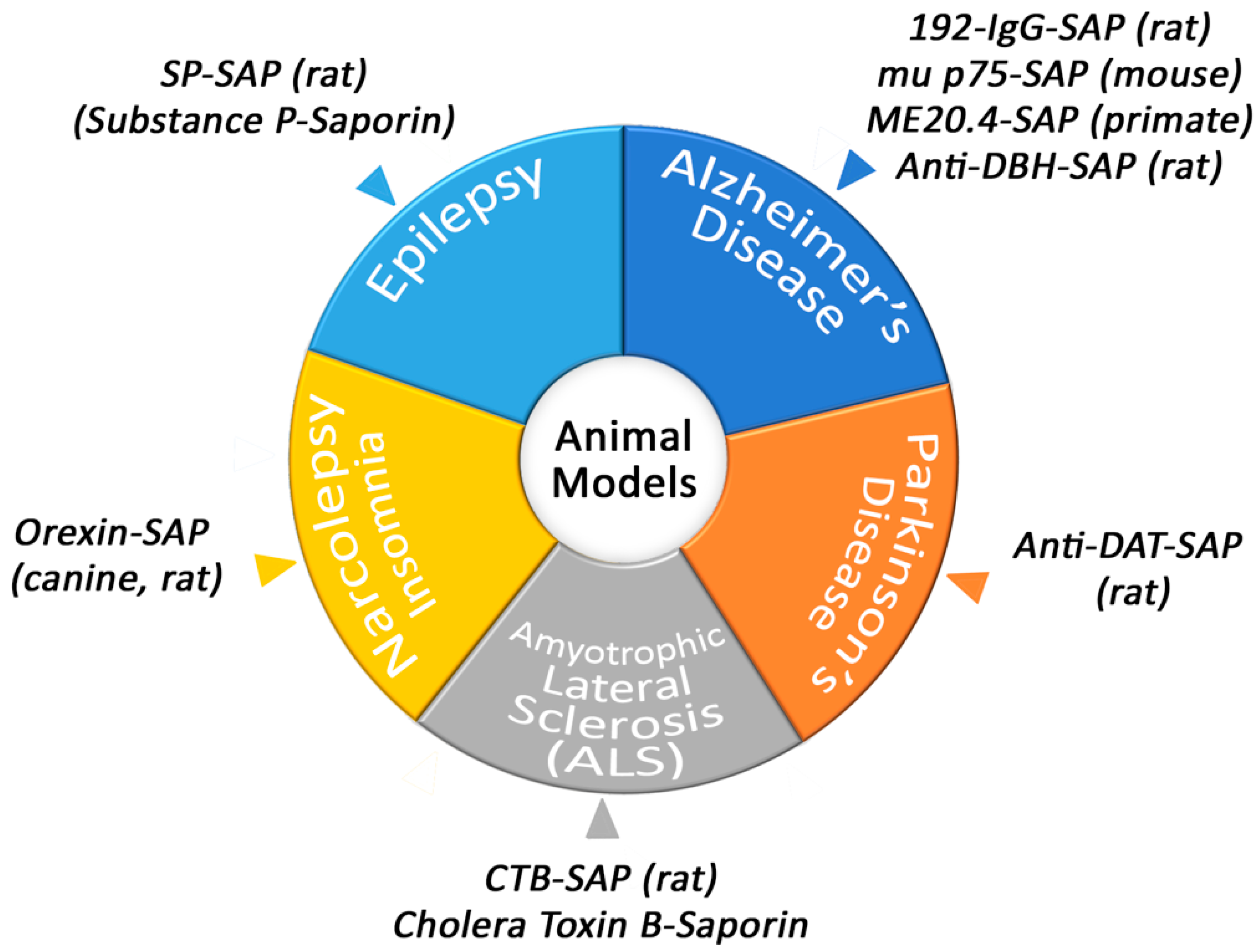
3.1. Narcolepsy/Insomnia
3.2. Amyotrophic Lateral Sclerosis
3.3. Parkinson’s Disease
3.4. Itch
3.5. Epilepsy
4. Secondary Conjugates and Streptavidin-ZAP
5. Clinical Trial for Cancer Pain
5.1. The Road to Human Clinical Trials
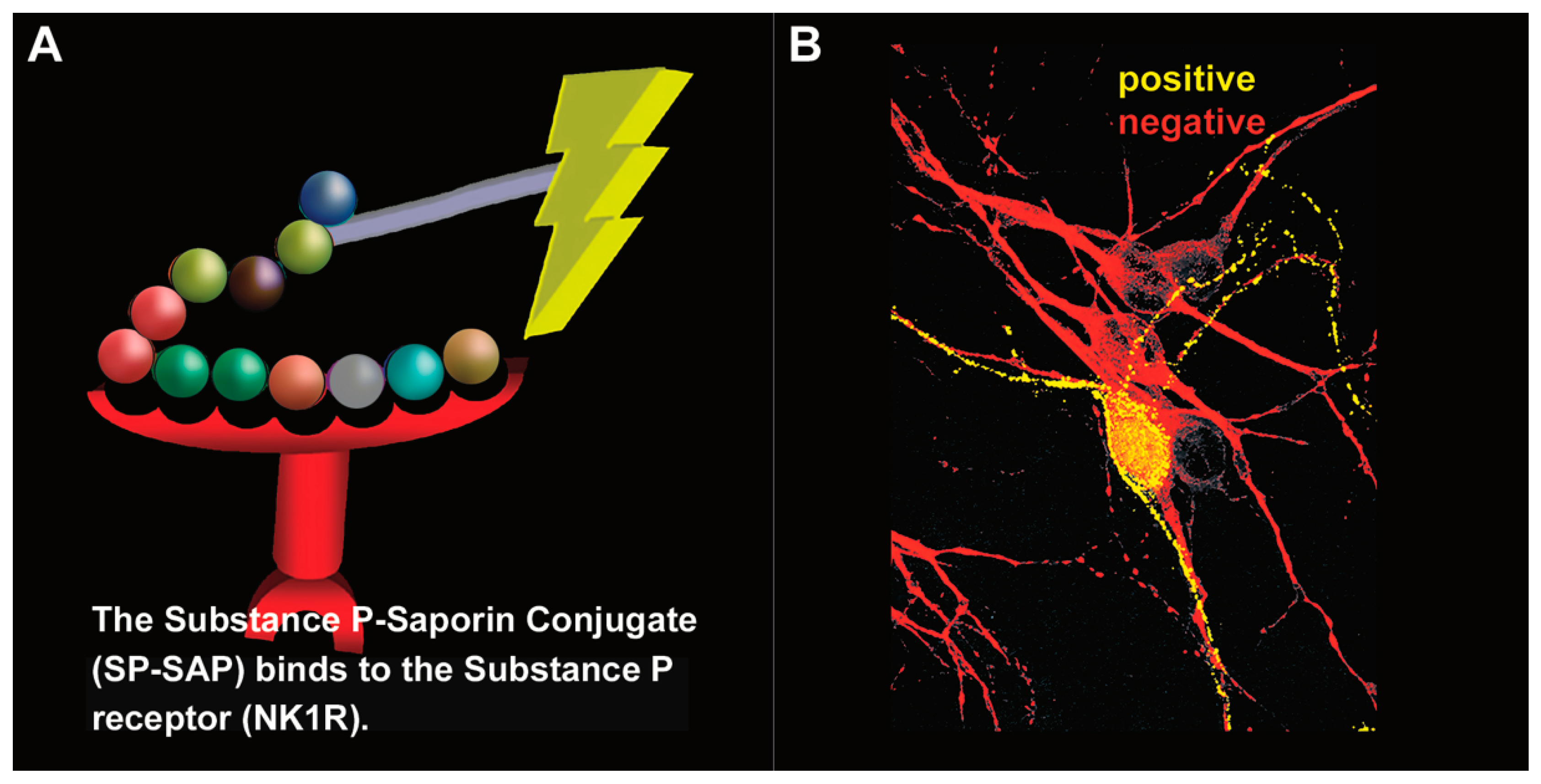
5.2. Human Clinical Trial
5.3. Preclinical Work Using Stable Substance P-Saporin (SSP-SAP)
This entry is adapted from the peer-reviewed paper 10.3390/toxins14030184
References
- Schwartz, R.S. Paul Ehrlich’s Magic Bullets. N. Engl. J. Med. 2004, 350, 1079–1080.
- Stirpe, F.; Barbieri, L.; Battelli, M.G.; Soria, M.; Lappi, D. Ribosome–Inactivating Proteins from Plants: Present Status and Future Prospects. Bio/Technology 1992, 10, 405–412.
- Bourrie, B.J.P.; Casellas, P.; Blythman, H.E.; Jansen, F.K. Study of the plasma clearance of antibody–ricin-A-chain immunotoxins. Evidence for specific recognition sites on the A chain that mediate rapid clearance of the immunotoxin. JBIC J. Biol. Inorg. Chem. 1986, 155, 1–10.
- Siena, S.; Bregni, M.; Formosa, A.; Martineau, D.; Lappi, D.A.; Bonadonna, G.; Gianni, A.M. Evaluation of anti-human T-lymphocyte saporin immunotoxins potentially useful in human transplantation. Transplantation 1988, 46, 747–753.
- Chaudhary, V.K.; FitzGerald, D.J.; Adhya, S.; Pastan, I. Activity of a recombinant fusion protein between transforming growth factor type alpha and Pseudomonas toxin. Proc. Natl. Acad. Sci. USA 1987, 84, 4538–4542.
- Kreitman, R.J.; Siegall, C.B.; Chaudhary, V.K.; Fitzgerald, D.J.; Pastan, I. Properties of chimeric toxins with two recognition domains: Interleukin 6 and transforming growth factor alpha at different locations in Pseudomonas exotoxin. Bioconjug. Chem. 1992, 3, 63–68.
- Kaplan, G.; Lee, F.; Onda, M.; Kolyvas, E.; Bhardwaj, G.; Baker, D.; Pastan, I. Protection of the Furin Cleavage Site in Low-Toxicity Immunotoxins Based on Pseudomonas Exotoxin A. Toxins 2016, 8, 217.
- Chaudhary, V.K.; Jinno, Y.; FitzGerald, D.; Pastan, I. Pseudomonas extoxin contains a specific sequence at the carboxyl ter-minus that is required for cytotoxicity. Proc. Natl. Acad. Sci. USA 1990, 87, 308–312.
- Mazor, R.; Pastan, I. Immunogenicity of Immunotoxins Containing Pseudomonas Exotoxin A: Causes, Consequences, and Mitigation. Front. Immunol. 2020, 11, 1261.
- Collier, R.J. Effect of diphtheria toxin on protein synthesis: Inactivation of one of the transfer factors. J. Mol. Biol. 1967, 25, 83–98.
- Stirpe, F.; Gasperi-Campani, A.; Barbieri, L.; Falasca, A.; Abbondanza, A.; Stevens, A.W. Ribosome-inactivating proteins from the seeds of Saponaria officinalis L. (soapwort), of Agrostemma githago L. (corn cockle) and of Asparagus officinalis L. (asparagus), and from the latex of Hura crepitans L. (sandbox tree). Biochem. J. 1983, 216, 617–625.
- Thorpe, P.E.; Brown, A.N.F.; Bremner, J.A.G.; Foxwell, B.M.J.; Stirpe, F. An Immunotoxin Composed of Monoclonal Anti-Thy 1.1 Antibody and a Ribosome-Inactivating Protein from Saponaria officinalis: Potent Antitumor Effects In Vitro and In Vivo. JNCI J. Natl. Cancer Inst. 1985, 75, 151–159.
- Siena, S.; Lappi, D.A.; Bregni, M.; Formosa, A.; Villa, S.; Soria, M.; Bonadonna, G.; Gianni, A.M. Synthesis and characterization of an antihuman T-lymphocyte saporin immunotoxin (OKT1-SAP) with in vivo stability into nonhuman primates. Blood 1988, 72, 756–765.
- Glennie, M.J.; Brennand, D.M.; Bryden, F.; McBride, H.M.; Stirpe, F.; Worth, A.T.; Stevenson, G.T. Bispecific F(ab’ gamma)2 antibody for the delivery of saporin in the treatment of lymphoma. J. Immunol. 1988, 141, 3662–3670.
- Flavell, D.J.; Noss, A.; Pulford, K.A.F.; Ling, N.; Flavell, S.U. Systemic delivery with 3BIT, a triple combination cocktail of anti-CD19, -CD22, and -CD38-saporin immunotoxins, is curative of human B-cell lymphoma in severe combined immunode-ficient mice. Cancer Res. 1997, 57, 4824–4829.
- Giansanti, F.; Flavell, D.J.; Angelucci, F.; Fabbrini, M.S.; Ippoliti, R. Strategies to Improve the Clinical Utility of Saporin-Based Targeted Toxins. Toxins 2018, 10, 82.
- Zuppone, S.; Assalini, C.; Minici, C.; Bertagnoli, S.; Branduardi, P.; Degano, M.; Fabbrini, M.S.; Montorsi, F.; Salonia, A.; Vago, R. The anti-tumoral potential of the saporin-based uPAR-targeting chimera ATF-SAP. Sci. Rep. 2020, 10, 2521.
- Dementia. Available online: https://www.who.int/news-room/fact-sheets/detail/dementia (accessed on 27 July 2021).
- Bolshakov, A.P.; Stepanichev, M.Y.; Dobryakova, Y.V.; Spivak, Y.S.; Markevich, V.A. Saporin from Saponaria officinalis as a Tool for Experimental Research, Modeling, and Therapy in Neuroscience. Toxins 2020, 12, 546.
- Rossner, S.; Roßner, S. Cholinergic immunolesions by 192IgG-saporin—A useful tool to simulate pathogenic aspects of alzheimer’s disease. Int. J. Dev. Neurosci. 1997, 15, 835–850.
- Arendt, T. Impairment in memory function and neurodegenerative changes in the cholinergic basal forebrain system induced by chronic intake of ethanol. Cell Anim. Models Aging Dement. Res. 1994, 44, 173–187.
- Giovannini, M.G.; Scali, C.; Prosperi, C.; Bellucci, A.; Vannucchi, M.G.; Rosi, S.; Pepeu, G.; Casamenti, F. β-Amyloid-Induced Inflammation and Cholinergic Hypofunction in the Rat Brain in Vivo: Involvement of the p38MAPK Pathway. Neurobiol. Dis. 2002, 11, 257–274.
- Verkhratsky, A.; Rodrigues, J.J.; Pivoriunas, A.; Zorec, R.; Semyanov, A. Astroglial atrophy in Alzheimer’s disease. Pflüg. Arch. Eur. J. Physiol. 2019, 471, 1247–1261.
- Davies, P.; Maloney, A.J. Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 1976, 308, 1403.
- Blanco-Centurion, C.; Xu, M.; Murillo-Rodriguez, E.; Gerashchenko, D.; Shiromani, A.M.; Salin-Pascual, R.J.; Hof, P.R.; Shiromani, P.J. Adenosine and Sleep Homeostasis in the Basal Forebrain. J. Neurosci. 2006, 26, 8092–8100.
- Wrenn, C.C.; Wiley, R.G. The behavioral functions of the cholinergic basal forebrain: Lessons from 192 IgG-SAPORIN. Int. J. Dev. Neurosci. 1998, 16, 595–602.
- McGaughy, J.; Everitt, B.; Robbins, T.; Sarter, M. The role of cortical cholinergic afferent projections in cognition: Impact of new selective immunotoxins. Behav. Brain Res. 2000, 115, 251–263.
- Beach, T.G.; Potter, E.P.; Kuo, Y.-M.; Emmerling, M.R.; Durham, A.R.; Webster, S.D.; Walker, D.G.; Sue, I.L.; Scott, S.; Layne, K.J.; et al. Cholinergic deafferentation of the rabbit cortex: A new animal model of Aβ deposition. Neurosci. Lett. 2000, 283, 9–12.
- Ferreira, G.; Meurisse, M.; Tillet, Y.; Lévy, F. Distribution and co-localization of choline acetyltransferase and p75 neurotrophin receptors in the sheep basal forebrain: Implications for the use of a specific cholinergic immunotoxin. Neuroscience 2001, 104, 419–439.
- Berger-Sweeney, J.; Stearns, N.A.; Murg, S.L.; Floerke-Nashner, L.R.; Lappi, D.; Baxter, M.G. Selective Immunolesions of Cholinergic Neurons in Mice: Effects on Neuroanatomy, Neurochemistry, and Behavior. J. Neurosci. 2001, 21, 8164–8173.
- Xu, Q.; Wang, D.R.; Dong, H.; Chen, L.; Lu, J.; Lazarus, M.; Cherasse, Y.; Chen, G.H.; Qu, W.M.; Huang, Z.L. Medial Para-brachial Nucleus Is Essential in Controlling Wakefulness in Rats. Front. Neurosci. 2021, 15, 645877.
- Sakurai, T.; Amemiya, A.; Ishii, M.; Matsuzaki, I.; Chemelli, R.M.; Tanaka, H.; Williams, S.C.; Richardson, J.A.; Kozlowski, G.P.; Wilson, S.; et al. Orexins and Orexin Receptors: A Family of Hypothalamic Neuropeptides and G Protein-Coupled Receptors that Regulate Feeding Behavior. Cell 1998, 92, 573–585.
- de Lecea, L.; Kilduff, T.S.; Peyron, C.; Gao, X.-B.; Foye, P.E.; Danielson, P.E.; Fukuhara, C.; Battenberg, E.L.F.; Gautvik, V.T.; Bartlett, F.S., III; et al. The hypocretins: Hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl. Acad. Sci. USA 1998, 95, 322–327.
- Lind, L.A.; Murphy, E.R.; Lever, T.E.; Nichols, N.L. Hypoglossal Motor Neuron Death Via Intralingual CTB–saporin (CTB–SAP) Injections Mimic Aspects of Amyotrophic Lateral Sclerosis (ALS) Related to Dysphagia. Neuroscience 2018, 390, 303–316.
- Meredith, G.E.; Sonsalla, P.K.; Chesselet, M.-F. Animal models of Parkinson’s disease progression. Acta Neuropathol. 2008, 115, 385–398.
- Wiley, R.G.; Harrison, M.B.; Levey, A.I.; Lappi, D. Destruction of midbrain dopaminergic neurons by using immunotoxin to dopamine transporter. Cell. Mol. Neurobiol. 2003, 23, 839–850.
- Weisshaar, E. Epidemiology of Itch. Immunodefic. Ski. 2016, 50, 5–10.
- Turski, W.A.; Cavalheiro, E.; Schwarz, M.; Czuczwar, S.J.; Kleinrok, Z.; Turski, L. Limbic seizures produced by pilocarpine in rats: Behavioural, electroencephalographic and neuropathological study. Behav. Brain Res. 1983, 9, 315–335.
- Curia, G.; Longo, D.; Biagini, G.; Jones, R.S.; Avoli, M. The pilocarpine model of temporal lobe epilepsy. J. Neurosci. Methods 2008, 172, 143–157.
- Wiley, R.G.; Lappi, D.A. (Eds.) Molecular Surgery with Targeted Toxins; Humana Press: Totowa, NJ, USA, 2005; Volume 1, p. 311.
- Polito, L.; Bortolotti, M.; Mercatelli, D.; Battelli, M.G.; Bolognesi, A. Saporin-S6: A Useful Tool in Cancer Therapy. Toxins 2013, 5, 1698–1722.
- Till, M.; May, R.D.; Uhr, J.W.; Thorpe, E.P.; Vitetta, E.S. An assay that predicts the ability of monoclonal antibodies to form potent ricin A chain-containing immunotoxins. Cancer Res. 1988, 48, 1119–1123.
- Kohls, M.D.; Lappi, D. Mab-Zap: A Tool for Evaluating Antibody Efficacy for Use in an Immunotoxin. Biotechniques 2000, 28, 162–165.
- Mantyh, P.W.; Rogers, S.D.; Honore, P.; Allen, B.J.; Ghilardi, J.R.; Li, J.; Daughters, R.S.; Lappi, D.A.; Wiley, R.G.; Simone, D.A. Inhibition of Hyperalgesia by Ablation of Lamina I Spinal Neurons Expressing the Substance P Receptor. Science 1997, 278, 275–279.
- Allen, J.W.; Mantyh, P.W.; Horais, K.; Tozier, N.; Rogers, S.D.; Ghilardi, J.R.; Cizkova, D.; Grafe, M.R.; Richter, P.; Lappi, D.A.; et al. Safety Evaluation of Intrathecal Substance P-Saporin, a Targeted Neurotoxin, in Dogs. Toxicol. Sci. 2006, 91, 286–298.
