The electrospun nanofiber membrane has a unique structure and biological function similar to the extracellular matrix (ECM), and is considered an advanced wound dressing. They have significant potential in encapsulating and delivering active substances that promote wound healing.
- wound dressing
- electrospinning
- nanostructure
- nanocomposite
1. Introduction
2. Wound and Wound Dressing
2.1. Wounds Classification
2.2. Types of Wound Dressing
|
Nature |
Category |
Advantages |
Disadvantages |
Ref. |
|---|---|---|---|---|
|
Traditional wound dressing |
Gauze, lint, bandage |
Easy to use and economical |
1. Dry, unable to maintain a moist healing environment 2. Adhering to the wound site is difficult to remove |
[46] |
|
Modern wound dressing |
Film |
1. Transparent, can observe wound changes 2. Form a bacterial barrier 3. Gas and water vapor permeability |
1. Absorptive capacity is not strong 2. Obstruct the regeneration of epithelial tissue |
[47] |
|
Foam |
1. High water absorption performance to maintain the moist environment of the wound 2. Change the dressing without damage |
1. Weak adhesion 2. Completely opaque |
[48] |
|
|
Hydrocolloid |
1. Stimulate tissue autolysis and debridement 2. The closed structure blocks the invasion of external bacteria |
1. Poor degradability 2. Produce a special smell |
[49] |
|
|
Hydrogel |
1. Ability to replenish water and maintain a humid environment 2. Comfortable and easy to replace |
1. No adhesion, low mechanical strength 2. High water content, limited absorption capacity, not suitable for wounds with high exudate |
[50] |
|
|
Alginate |
1. Non-toxic, fast hemostasis 2. Good air permeability 3. Biodegradation |
Not suitable for dry wounds |
[51] |
|
|
Bioactive wound dressing |
Drug-loaded dressing, antibacterial dressing |
1. Good biocompatibility 2. Anti-inflammatory and antibacterial 3. Promote the growth of cells and tissues |
Induce immune response |
[52] |
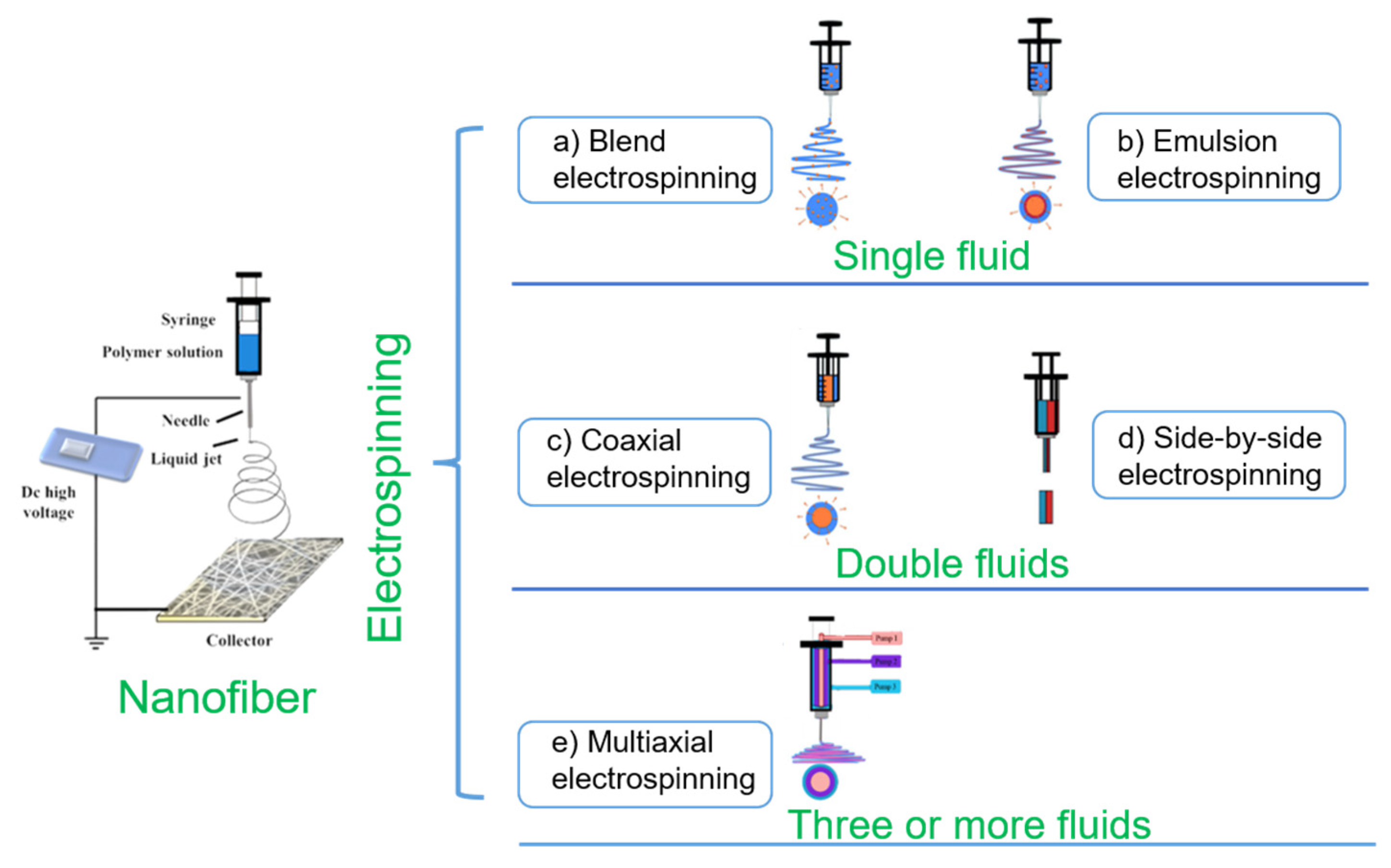
3. Electrospinning Technology
4. Electrospun Nanofibers in Wound Dressing
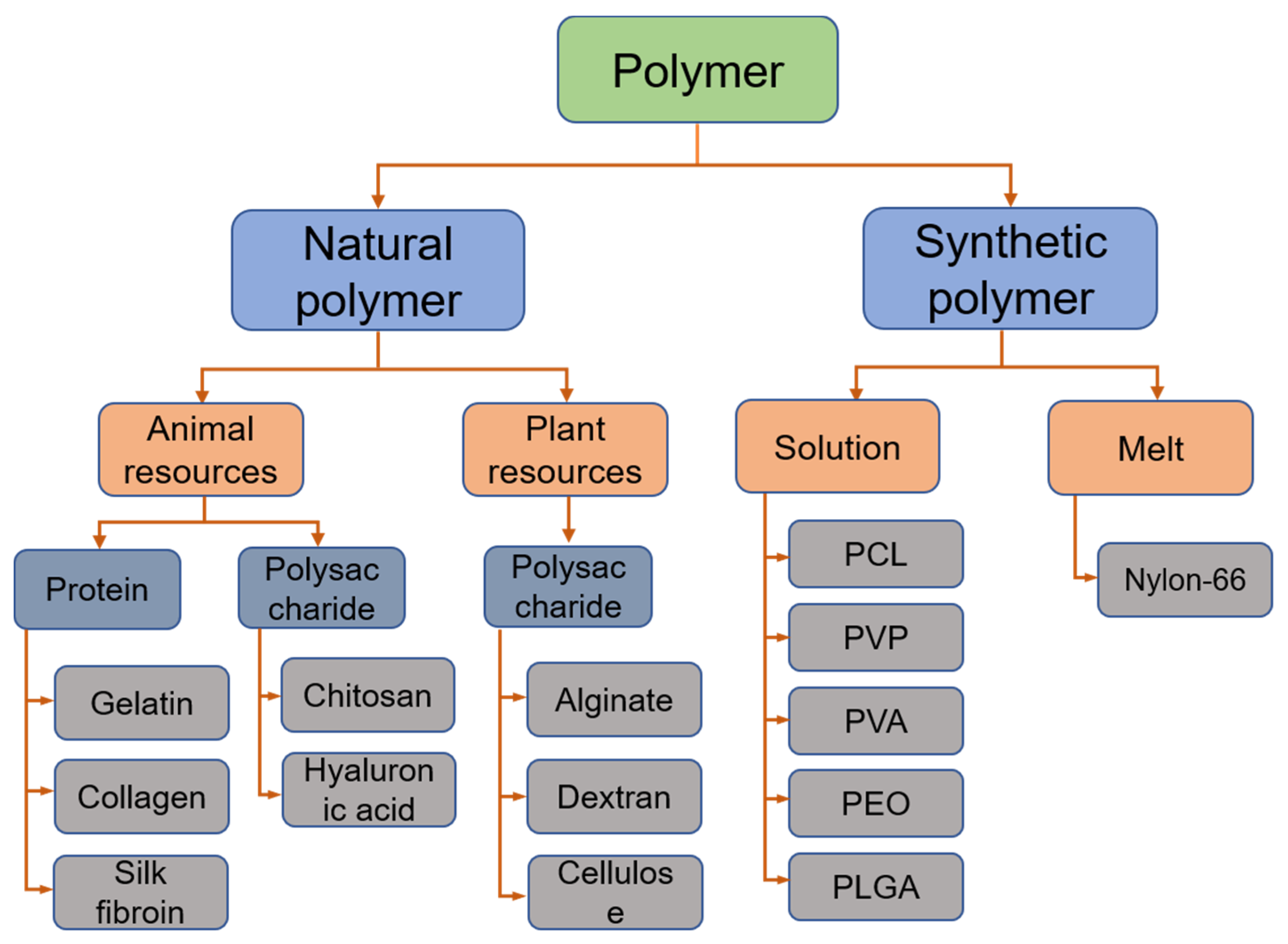
4.1. Polymer in Electrospun Wound Dressing
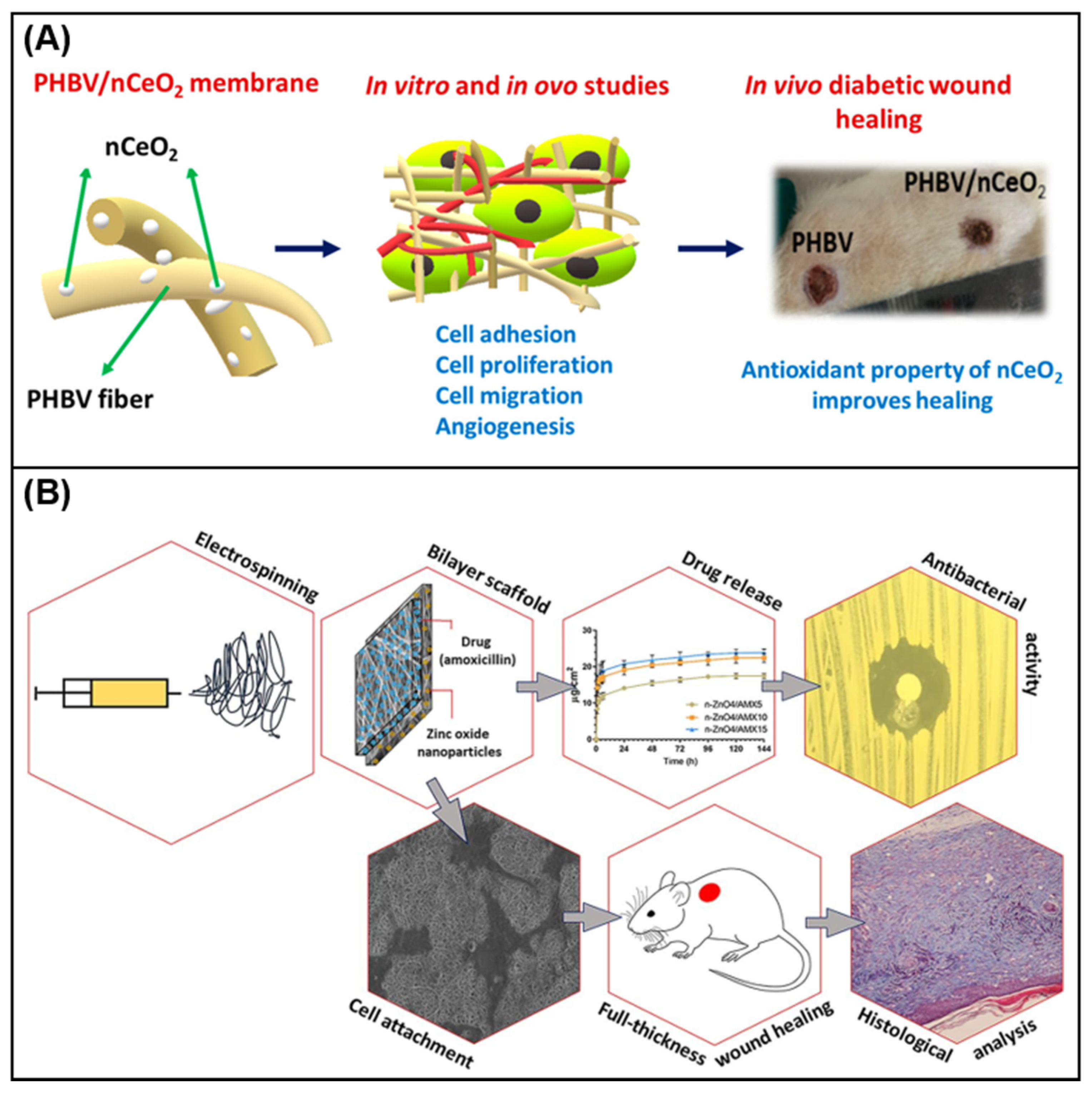
4.2. Bioactive Ingredients in Electrospun Wound Dressing
|
Scaffold Material |
Additional Polymer |
Bioactive Ingredients |
Solvent |
Electrospinning Technique |
Highlights |
Ref. |
|---|---|---|---|---|---|---|
|
Gelatin |
CA |
Berberine |
HFP |
Blend |
Has strong antibacterial activity and is suitable for the management and treatment of diabetic foot ulcer |
[72] |
|
CA/PVP |
Gentamicin |
Acetic acid, ethanol |
Bi-layer |
Thermal stability, wettability characteristics and antibacterial activity |
[73] |
|
|
Collagen |
EC/PLA |
Silver sulfadiazine |
Chloroform, ethanol |
Blend |
The antibacterial performance showed inhibitory activity against Bacillus (9.71 ± 1.15 mm) and E. coli (12.46 ± 1.31 mm), promoted cell proliferation and adhesion |
[74] |
|
Zein/PCL |
n-ZnO, aloe vera |
Chloroform, ethanol |
Blend |
The developed nanofibers revealed good cell compatibility |
[75] |
|
|
CS |
PCL |
Lidocaine hydrochloride, mupirocin |
HFIP, DCM |
Dual |
Have the functions of promoting hemostasis, antibacterial, and drug release. |
[76] |
|
PEO/CNC |
Acacia extract |
Acetic acid |
Blend |
A continuous release of natural acacia extract from nanofibers occurred during 24 h |
[77] |
|
|
SF |
PLGA |
Artemisinin |
HFIP |
Blend |
The fabricated membrane shows anti-inflammatory properties without cytotoxicity |
[78] |
|
PCL/PVA |
Curcumin |
Formic acid, dichloromethane |
Blend |
Accelerate wound healing in diabetic mice |
[79] |
|
|
Alginate |
PVA/CS |
Dexpanthenol |
Acetic acid |
Coaxial |
Not only is it non-toxic to fibroblasts, but it also has a certain effect on cell attachment and morphology |
[80] |
|
PVA |
Cardamom extract |
Distilled water |
Blend |
Have good biocompatibility and antibacterial properties |
[81] |
|
|
PVP |
EC |
CIP, AgNP |
Ethanol, acetic acid, acetone |
Side-by-side |
Janus fiber has good bactericidal activity |
[70] |
|
PLA/PEO/Collagen |
Cefazolin |
DCM, DMF, HFIP, ethanol |
Coaxial |
Antibacterial studies on wounds show that they can effectively inhibit the growth of microorganisms. |
[82] |
|
|
PCL |
CS |
Aloe vera |
Acetic acid |
Blend |
Have good antibacterial properties and biocompatibility |
[83] |
|
CS |
Curcumin |
Ethanol, acetic acid |
Blend |
Shows antibacterial, anti-oxidant and wound healing capabilities |
[84] |
|
|
Gelatin |
Oregano oil |
HFIP |
Blend |
Good biocompatibility and antibacterial activity |
[85] |
|
|
/ |
Urtica dioica, n-ZnO |
DMF, DCM |
Blend |
The hybrid scaffold shows high antibacterial activity and cell viability |
[86] |
|
|
Gelatin |
Clove essential oil |
Glacial acetic acid |
Blend |
Antibacterial activity |
[87] |
|
|
PVA |
CS/Starch |
/ |
Double-distilled water, acetic acid |
Blend |
Proper tensile strength and elongation, excellent biocompatibility and antibacterial activity |
[88] |
|
CS |
/ |
Acetic acid |
Blend |
Good physical and chemical properties, biocompatibility and antibacterial properties |
[89] |
|
|
PEO |
CS |
Vancomycin |
Acetic acid |
Blend |
Antibacterial effects against S. aureus |
[90] |
|
CS |
Teicoplanin |
Acetic acid |
Dual |
Wound closure was significantly improved |
[91] |
HFP: Hexafluoropropylene, EC: Ethylcellulose, PLA: Polylactic acid, HFIP: 1,1,1,3,3,3-hexafluoro-2-propanol, DCM: Dichloromethane, PEO: Polyethene oxide, CNC: Cellulose nanocrystals, DMF: N,N-dimethylformamide.
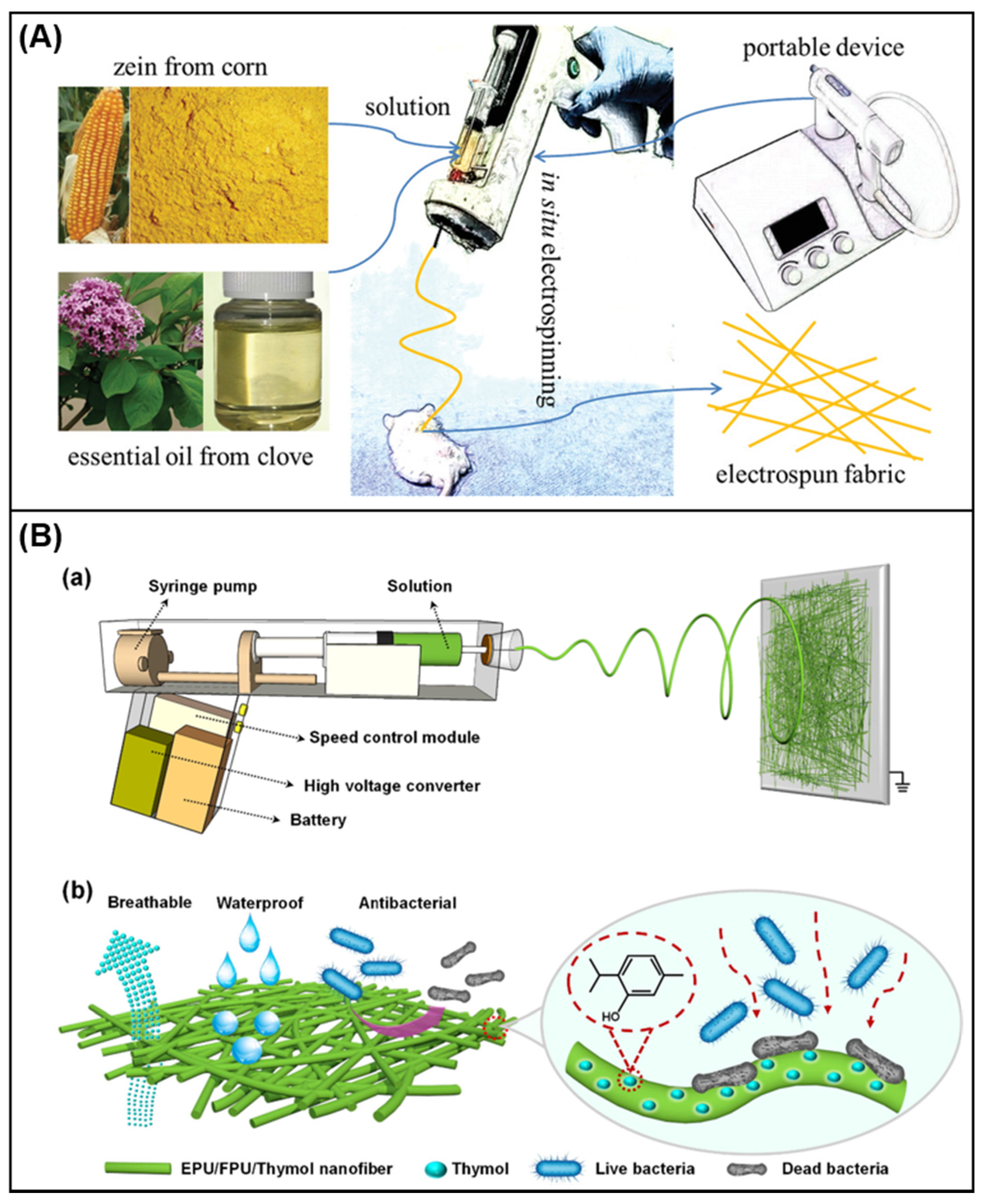
4.3. In Situ Electrospinning in Wound Dressing
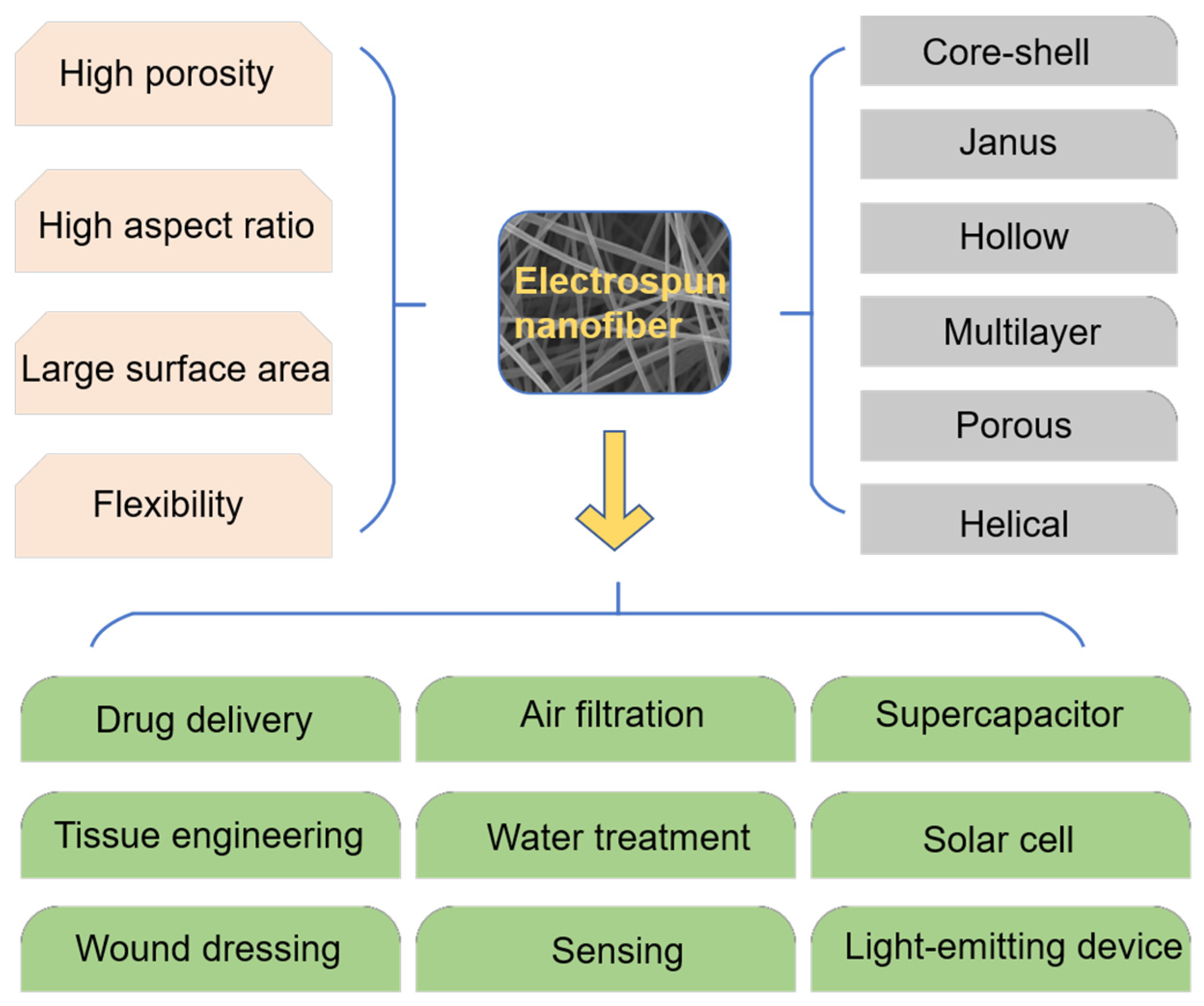
4.4. Application of Electrospinning Technology in Other Fields
This entry is adapted from the peer-reviewed paper 10.3390/membranes11100770
References
- Nosrati, H.; Aramideh Khouy, R.; Nosrati, A.; Khodaei, M.; Banitalebi-Dehkordi, M.; Ashrafi-Dehkordi, K.; Sanami, S.; Alizadeh, Z. Nanocomposite scaffolds for accelerating chronic wound healing by enhancing angiogenesis. J. Nanobiotechnology 2021, 19, 1–22.
- Fatehi, P.; Abbasi, M. Medicinal plants used in wound dressings made of electrospun nanofibers. J. Tissue Eng. Regen. Med. 2020, 14, 1527–1548.
- El Ayadi, A.; Jay, J.W.; Prasai, A. Current approaches targeting the wound healing phases to attenuate fibrosis and scarring. Int. J. Mol. Sci. 2020, 21, 1105.
- Chen, M.; Tian, J.; Liu, Y.; Cao, H.; Li, R.; Wang, J.; Wu, J.; Zhang, Q. Dynamic covalent constructed self-healing hydrogel for sequential delivery of antibacterial agent and growth factor in wound healing. Chem. Eng. J. 2019, 373, 413–424.
- Chen, K.; Wang, F.; Liu, S.; Wu, X.; Xu, L.; Zhang, D. In situ reduction of silver nanoparticles by sodium alginate to obtain silver-loaded composite wound dressing with enhanced mechanical and antimicrobial property. Int. J. Biol. Macromol. 2020, 148, 501–509.
- Fahimirad, S.; Ajalloueian, F. Naturally-derived electrospun wound dressings for target delivery of bioactive agents. Int. J. Pharm. 2019, 566, 307–328.
- Kanikireddy, V.; Varaprasad, K.; Jayaramudu, T.; Karthikeyan, C.; Sadiku, R. Carboxymethyl cellulose-based materials for infection control and wound healing: A review. Int. J. Biol. Macromol. 2020, 164, 963–975.
- Das, A.; Uppaluri, R.; Das, C. Feasibility of poly-vinyl alcohol/starch/glycerol/citric acid composite films for wound dressing applications. Int. J. Biol. Macromol. 2019, 131, 998–1007.
- Weng, W.; Chi, J.; Yu, Y.; Zhang, C.; Shi, K.; Zhao, Y. Multifunctional composite inverse opal film with multiactives for wound healing. ACS Appl. Mater. Interfaces 2021, 13, 4567–4573.
- Bužarovska, A.; Dinescu, S.; Lazar, A.D.; Serban, M.; Pircalabioru, G.G.; Costache, M.; Gualandi, C.; Avérous, L. Nanocomposite foams based on flexible biobased thermoplastic polyurethane and ZnO nanoparticles as potential wound dressing materials. Mater. Sci. Eng. C 2019, 104, 109893.
- Cui, H.; Liu, M.; Yu, W.; Cao, Y.; Zhou, H.; Yin, J.; Liu, H.; Que, S.; Wang, J.; Huang, C.; et al. Copper peroxide-loaded gelatin sponges for wound dressings with antimicrobial and accelerating healing properties. ACS Appl. Mater. Interfaces 2021, 13, 26800–26807.
- Zhao, X.; Pei, D.; Yang, Y.; Xu, K.; Yu, J.; Zhang, Y.; Zhang, Q.; He, G.; Zhang, Y.; Li, A.; et al. Green tea derivative driven smart hydrogels with desired functions for chronic diabetic wound treatment. Adv. Funct. Mater. 2021, 31, 2009442.
- Pan, X.; Kong, D.; Wang, W.; Liu, W.; Ou-Yang, W.; Zhang, C.; Wang, Q.; Huang, P.; Zhang, C.; Li, Y. Synthetic polymeric antibacterial hydrogel for methicillin-resistant staphylococcus aureus-infected wound healing: Nanoantimicrobial self-assembly, drug- and cytokine-free strategy. ACS Nano 2020, 14, 12905–12917.
- Chen, L.; Zhang, L.; Zhang, H.; Sun, X.; Liu, D.; Zhang, J.; Zhang, Y.; Cheng, L.; Santos, H.A.; Cui, W. Programmable immune activating electrospun fibers for skin regeneration. Bioact. Mater. 2021, 6, 3218–3230.
- Guo, X.; Liu, Y.; Bera, H.; Zhang, H.; Chen, Y.; Cun, D.; Foderà, V.; Yang, M. α-Lactalbumin-based nanofiber dressings improve burn wound healing and reduce scarring. ACS Appl. Mater. Interfaces 2020, 12, 45702–45713.
- Toriello, M.; Afsari, M.; Shon, H.K.; Tijing, L.D. Progress on the fabrication and application of electrospun nanofiber composites. Membranes 2020, 10, 1–35.
- Akhmetova, A.; Heinz, A. Electrospinning proteins for wound healing purposes: Opportunities and challenges. Pharmaceutics 2021, 13, 1–22.
- Jao, D.; Beachley, V.Z. Continuous dual-track fabrication of polymer micro-/nanofibers based on direct drawing. ACS Macro Lett. 2019, 8, 588–595.
- Shin, S.; Menk, F.; Kim, Y.; Lim, J.; Char, K.; Zentel, R.; Choi, T.L. Living light-induced crystallization-driven self-assembly for rapid preparation of semiconducting nanofibers. J. Am. Chem. Soc. 2018, 140, 6088–6094.
- Qin, W.; Li, J.; Tu, J.; Yang, H.; Chen, Q.; Liu, H. Fabrication of porous chitosan membranes composed of nanofibers by low temperature thermally induced phase separation, and their adsorption behavior for Cu2+. Carbohydr. Polym. 2017, 178, 338–346.
- Kamin, Z.; Abdulrahim, N.; Misson, M.; Chiam, C.K.; Sarbatly, R.; Krishnaiah, D.; Bono, A. Use of melt blown polypropylene nanofiber templates to obtain homogenous pore channels in glycidyl methacrylate/ethyl dimethacrylate-based monoliths. Chem. Eng. Commun. 2021, 208, 661–672.
- Bazmandeh, A.Z.; Mirzaei, E.; Fadaie, M.; Shirian, S.; Ghasemi, Y. Dual spinneret electrospun nanofibrous/gel structure of chitosan-hyaluronic acid as a wound dressing: In-vitro and in-vivo studies. Int. J. Biol. Macromol. 2020, 162, 359–373.
- Sabra, S.; Ragab, D.M.; Agwa, M.M.; Rohani, S. Recent advances in electrospun nanofibers for some biomedical applications. Eur. J. Pharm. Sci. 2020, 144, 105224.
- Abazari, M.F.; Nasiri, N.; Nejati, F.; Kohandani, M.; Hajati-Birgani, N.; Sadeghi, S.; Piri, P.; Soleimanifar, F.; Rezaei-Tavirani, M.; Mansouri, V. Acceleration of osteogenic differentiation by sustained release of BMP2 in PLLA/graphene oxide nanofibrous scaffold. Polym. Adv. Technol. 2021, 32, 272–281.
- Mozaffari, A.; Gashti, M.P.; Mirjalili, M.; Parsania, M. Argon and argon-oxygen plasma surface modification of gelatin nanofibers for tissue engineering applications. Membranes 2021, 11, 1–13.
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control. Release 2021, 334, 463–484.
- Li, Z.; Wen, W.; Chen, X.; Zhu, L.; Cheng, G.; Liao, Z.; Huang, H.; Ming, L. Release characteristics of an essential oil component encapsulated with cyclodextrin shell matrices. Curr. Drug Deliv. 2021, 18, 487–499.
- Yu, D.G. Preface-bettering drug delivery knowledge from pharmaceutical techniques and excipients. Curr. Drug Deliv. 2021, 18, 2–3.
- Schuhladen, K.; Raghu, S.N.V.; Liverani, L.; Neščáková, Z.; Boccaccini, A.R. Production of a novel poly(ɛ-caprolactone)-methylcellulose electrospun wound dressing by incorporating bioactive glass and Manuka honey. J. Biomed. Mater. Res. Part B 2021, 109, 180–192.
- Bootdee, K.; Nithitanakul, M. Poly (d, l-lactide-co-glycolide) nanospheres within composite poly (vinyl alcohol)/aloe vera electrospun nanofiber as a novel wound dressing for controlled release of drug. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 223–230.
- Lan, X.; Liu, Y.; Wang, Y.; Tian, F.; Miao, X.; Wang, H.; Tang, Y. Coaxial electrospun PVA/PCL nanofibers with dual release of tea polyphenols and ε-poly(L-lysine) as antioxidant and antibacterial wound dressing materials. Int. J. Pharm. 2021, 601, 120525.
- Bonferoni, M.C.; Rossi, S.; Sandri, G.; Caramella, C.; Del Fante, C.; Perotti, C.; Miele, D.; Vigani, B.; Ferrari, F. Bioactive medications for the delivery of platelet derivatives to skin wounds. Curr. Drug Deliv. 2019, 16, 472–483.
- Keshvardoostchokami, M.; Majidi, S.S.; Huo, P.; Ramachandran, R.; Chen, M.; Liu, B. Electrospun nanofibers of natural and synthetic polymers as artificial extracellular matrix for tissue engineering. Nanomaterials 2021, 11, 1–23.
- Augustine, R.; Rehman, S.R.U.; Ahmed, R.; Zahid, A.A.; Sharifi, M.; Falahati, M.; Hasan, A. Electrospun chitosan membranes containing bioactive and therapeutic agents for enhanced wound healing. Int. J. Biol. Macromol. 2020, 156, 153–170.
- Ather, S.; Harding, K.G.; Tate, S.J. Wound management and dressings. In Advanced Textiles for Wound Care, 2nd ed.; The Textile Institute Book Series; Woodhead Publishing: Cambridge, UK, 2019; pp. 1–22.
- Wang, W.; Lu, K.J.; Yu, C.H.; Huang, Q.L.; Du, Y.Z. Nano-drug delivery systems in wound treatment and skin regeneration. J. Nanobiotechnology 2019, 17, 1–15.
- Iacob, A.T.; Drăgan, M.; Ionescu, O.M.; Profire, L.; Ficai, A.; Andronescu, E.; Confederat, L.G.; Lupascu, D. An overview of biopolymeric electrospun nanofibers based on polysaccharides for wound healing management. Pharmaceutics 2020, 12, 1–49.
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin wound healing process and new emerging technologies for skin wound care and regeneration. Pharmaceutics 2020, 12, 1–30.
- Wang, Y.; Feng, Q.; Li, Z.; Bai, X.; Wu, Y.; Liu, Y. Evaluating the effect of integra seeded with adipose tissue-derived stem cells or fibroblasts in wound healing. Curr. Drug Deliv. 2020, 17, 629–635.
- Smet, S.; Probst, S.; Holloway, S.; Fourie, A.; Beele, H.; Beeckman, D. The measurement properties of assessment tools for chronic wounds: A systematic review. Int. J. Nurs. Stud. 2021, 121, 103998.
- Sen, C.K. Human wounds and its burden: An updated compendium of estimates. Adv. Wound Care 2019, 8, 39–48.
- Homaeigohar, S.; Boccaccini, A.R. Antibacterial biohybrid nanofibers for wound dressings. Acta Biomater. 2020, 107, 25–49.
- Eaglstein, W.H. Moist wound healing with occlusive dressings: A clinical focus. Dermatol. Surg. 2001, 27, 175–182.
- Driver, V.R.; Gould, L.J.; Dotson, P.; Gibbons, G.W.; Li, W.W.; Ennis, W.J.; Kirsner, R.S.; Eaglstein, W.H.; Bolton, L.L.; Carter, M.J. Identification and content validation of wound therapy clinical endpoints relevant to clinical practice and patient values for FDA approval. Part 1. Survey of the wound care community. Wound Repair Regen. 2017, 25, 454–465.
- Mouro, C.; Gomes, A.P.; Ahonen, M.; Fangueiro, R.; Gouveia, I.C. Chelidonium majus 1. Incorporated emulsion electrospun PCL/PVA_PEC nanofibrous meshes for antibacterial wound dressing applications. Nanomaterials 2021, 11, 1785.
- Montaser, A.S.; Rehan, M.; EI-Senousy, W.M.; Zaghloul, S. Designing strategy for coating cotton gauze fabrics and its application in wound healing. Carbohydr. Polym. 2020, 244, 116479.
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound dressings—A review. Biomedicine 2015, 5, 24–28.
- Chaganti, P.; Gordon, I.; Chao, J.H.; Zehtabchi, S. A systematic review of foam dressings for partial thickness burns. Am. J. Emerg. Med. 2019, 37, 1184–1190.
- Rezvani Ghomi, E.; Khalili, S.; Nouri Khorasani, S.; Esmaeely Neisiany, R.; Ramakrishna, S. Wound dressings: Current advances and future directions. J. Appl. Polym. Sci. 2019, 136, 1–12.
- Cascone, S.; Lamberti, G. Hydrogel-based commercial products for biomedical applications: A review. Int. J. Pharm. 2020, 573, 118803.
- Ahmad, A.; Mubarak, N.M.; Jannat, F.T.; Ashfaq, T.; Santulli, C.; Rizwan, M.; Najda, A.; Bin-Jumah, M.; Abdel-Daim, M.M.; Hussain, S.; et al. A critical review on the synthesis of natural sodium alginate based composite materials: An innovative biological polymer for biomedical delivery applications. Processes 2021, 9, 1–27.
- Tang, Y.; Lan, X.; Liang, C.; Zhong, Z.; Xie, R.; Zhou, Y.; Miao, X.; Wang, H.; Wang, W. Honey loaded alginate/PVA nanofibrous membrane as potential bioactive wound dressing. Carbohydr. Polym. 2019, 219, 113–120.
- Wang, C.; Wang, J.; Zeng, L.; Qiao, Z.; Liu, X.; Liu, H.; Zhang, J.; Ding, J. Fabrication of electrospun polymer nanofibers with diverse morphologies. Molecules 2019, 24, 834.
- Aidana, Y.; Wang, Y.; Li, J.; Chang, S.; Wang, K.; Yu, D.-G. Fast dissolution electrospun medicated nanofibers for effective delivery of poorly water-soluble drugs. Curr. Drug Deliv. 2021, 18.
- Nauman, S.; Lubineau, G.; Alharbi, H.F. Post processing strategies for the enhancement of mechanical properties of enms (Electrospun nanofibrous membranes): A review. Membranes 2021, 11, 1–38.
- Ma, H.; Burger, C.; Chu, B.; Hsiao, B.S. Electrospun nanofibers for environmental protection. Handb. Fibrous Mater. 2020, 773–806.
- Zhao, K.; Kang, S.X.; Yang, Y.Y.; Yu, D.G. Electrospun functional nanofiber membrane for antibiotic removal in water: Review. Polymers 2021, 13, 1–33.
- Wang, M.; Yu, D.-G.; Li, X.; Williams, G.R. The development and bio-applications of multifluid electrospinning. Mater. Highlights 2020, 1, 1–13.
- Wang, Y.; Tian, L.; Zhu, T.; Mei, J.; Chen, Z.; Yu, D.G. Electrospun aspirin/Eudragit/lipid hybrid nanofibers for colon-targeted delivery using an energy-saving process. Chem. Res. Chin. Univ. 2021, 37, 443–449.
- Li, Y.; Zhu, J.; Cheng, H.; Li, G.; Cho, H.; Jiang, M.; Gao, Q.; Zhang, X. Developments of advanced electrospinning techniques: A critical review. Adv. Mater. Technol. 2021, 2100410.
- Xin, R.; Ma, H.; Venkateswaran, S.; Hsiao, B.S. Electrospun nanofibrous adsorption membranes for wasterwater treatment: Mechanical strength enhancement. Chem. Res. Chinese Univ. 2021, 37, 355–365.
- Zare, M.; Dziemidowicz, K.; Williams, G.R.; Ramakrishna, S. Encapsulation of pharmaceutical and nutraceutical active ingredients using electrospinning processes. Nanomaterials 2021, 11, 1968.
- Wang, F.; Hu, S.; Jia, Q.; Zhang, L. Advances in electrospinning of natural biomaterials for wound dressing. J. Nanomater. 2020, 2020, 8719859.
- Memic, A.; Abudula, T.; Mohammed, H.S.; Joshi Navare, K.; Colombani, T.; Bencherif, S.A. Latest progress in electrospun nanofibers for wound healing applications. ACS Appl. Bio Mater. 2019, 2, 952–969.
- Li, H.; Chen, X.; Lu, W.; Wang, J.; Xu, Y.; Guo, Y. Application of electrospinning in antibacterial field. Nanomaterials 2021, 11, 1–29.
- Keirouz, A.; Chung, M.; Kwon, J.; Fortunato, G.; Radacsi, N. 2D and 3D electrospinning technologies for the fabrication of nanofibrous scaffolds for skin tissue engineering: A review. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, 1–32.
- Naskar, A.; Kim, K.S. Recent advances in nanomaterial-based wound-healing therapeutics. Pharmaceutics 2020, 12, 499.
- Abdalla, S.S.I.; Katas, H.; Azmi, F.; Busra, M.F.M. Antibacterial and anti-biofilm biosynthesised silver and gold nanoparticles for medical applications: Mechanism of action, toxicity and current status. Curr. Drug Deliv. 2020, 17, 88–100.
- Augustine, R.; Hasan, A.; Patan, N.K.; Dalvi, Y.B.; Varghese, R.; Antony, A.; Unni, R.N.; Sandhyarani, N.; Moustafa, A.E.A. Cerium oxide nanoparticle incorporated electrospun poly(3-hydroxybutyrate-co-3-hydroxyvalerate) membranes for diabetic wound healing applications. ACS Biomater. Sci. Eng. 2020, 6, 58–70.
- Yang, J.; Wang, K.; Yu, D.G.; Yang, Y.; Bligh, S.W.A.; Williams, G.R. Electrospun Janus nanofibers loaded with a drug and inorganic nanoparticles as an effective antibacterial wound dressing. Mater. Sci. Eng. C 2020, 111, 110805.
- Jafari, A.; Amirsadeghi, A.; Hassanajili, S.; Azarpira, N. Bioactive antibacterial bilayer PCL/gelatin nanofibrous scaffold promotes full - thickness wound healing. Int. J. Pharm. 2020, 583, 119413.
- Samadian, H.; Zamiri, S.; Ehterami, A.; Farzamfar, S.; Vaez, A.; Khastar, H.; Alam, M.; Ai, A.; Derakhshankhah, H.; Allahyari, Z.; et al. Electrospun cellulose acetate/gelatin nanofibrous wound dressing containing berberine for acetate/gelatin nanofibrous wound dressing containing berberine diabetic foot ulcer healing: In vitro and in vivo studies. Sci. Rep. 2020, 10, 1–12.
- López-Calderón, H.D.; Avilés-Arnaut, H.; Galán Wong, L.J.; Almaguer - Cantú, V.; Laguna-Camacho, J.R.; Calderón-Ramón, C.; Escalante- Martínez, J.E.; Arévalo-Niño, K. Electrospun polyvinylpyrrolidone-gelatin and cellulose acetate bi-Layer scaffold loaded with gentamicin as possible wound dressing. Polymers 2020, 12, 2311.
- Ahmadian, S.; Ghorbani, M.; Mahmoodzadeh, F. Silver sulfadiazine-loaded electrospun ethyl cellulose/polylactic acid/collagen nanofibrous mats with antibacterial properties for wound healing. Int. J. Biol. Macromol. 2020, 162, 1555–1565.
- Ghorbani, M.; Nezhad-Mokhtari, P.; Ramazani, S. Aloe vera-loaded nanofibrous scaffold based on zein/polycaprolactone/collagen for wound healing. Int. J. Biol. Macromol. 2020, 153, 921–930.
- Yang, S.; Li, X.; Liu, P.; Zhang, M.; Wang, C.; Zhang, B. Multifunctional chitosan/polycaprolactone nanofiber scaffolds with varied dual-drug release for wound-healing applications. ACS Biomater. Sci. Eng. 2020, 6, 4666–4676.
- Ribeiro, A.S.; Costa, S.M.; Ferreira, D.P.; Calhelha, R.C.; Barros, L.; Stoiković, D.; Soković, M.; Ferreira, I.C.F.R.; Fangueiro, R. Chitosan/nanocellulose electrospun fibers with enhanced antibacterial and antifungal activity for wound dressing applications. React. Funct. Polym. 2021, 159, 104808.
- Peng, Y.; Ma, Y.; Bao, Y.; Liu, Z.; Chen, L.; Dai, F.; Li, Z. Electrospun PLGA/SF/artemisinin composite nanofibrous membranes for wound dressing. Int. J. Biol. Macromol. 2021, 183, 68–78.
- Agarwal, Y.; Rajinikanth, P.S.; Ranjan, S.; Tiwari, U.; Balasubramnaiam, J.; Pandey, P.; Arya, D.K.; Anand, S.; Deepak, P. Curcumin loaded polycaprolactone-/polyvinyl alcohol-silk fibroin based electrospun nanofibrous mat for rapid healing of diabetic wound: An in-vitro and in-vivo studies. Int. J. Biol. Macromol. 2021, 176, 376–386.
- Najafiasl, M.; Osfouri, S.; Azin, R.; Zaeri, S. Alginate-based electrospun core/shell nanofibers containing dexpanthenol: A good candidate for wound dressing. J. Drug Deliv. Sci. Technol. 2020, 57, 101708.
- Najafi, S.; Gholipour- Kanani, A.; Eslahi, N.; Bahrami, S.H. Study on release of cardamom extract as an antibacterial agent from electrospun scaffold based on sodium alginate. J. Text. Inst. 2021, 112, 1482–1490.
- Hajikhani, M.; EMAM-Djomeh, Z.; Askari, G. Fabrication and characterization of mucoadhesive bioplastic patch via coaxial polylactic acid (PLA) based electrospun nanofibers with antimicrobial and wound healing application. Int. J. Biol. Macromol. 2021, 172, 143–153.
- Yin, J.; Xu, L. Batch preparation of electrospun polycaprolactone/chitosan/aloe vera blended nanofiber membranes for novel wound dressing. Int. J. Biol. Macromol. 2020, 160, 352–363.
- Fahimirad, S.; Abtahi, H.; Satei, P.; Ghaznavi-Rad, E.; Moslehi, M.; Ganji, A. Wound healing performance of PCL/chitosan based electrospun nanofiber electrosprayed with curcumin loaded chitosan nanoparticles. Carbohydr. Polym. 2021, 259, 117640.
- El Fawal, G.; Hong, H.; Mo, X.; Wang, H. Fabrication of scaffold based on gelatin and polycaprolactone (PCL) for wound dressing application. J. Drug Deliv. Sci. Technol. 2021, 63, 102501.
- Ghiyasi, Y.; Salahi, E.; Esfahani, H. Synergy effect of Urtica dioica and ZnO NPs on microstructure, antibacterial activity and cytotoxicity of electrospun PCL scaffold for wound dressing application. Mater. Today Commun. 2021, 26, 102163.
- Unalan, I.; Endlein, S.J.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Detsch, R.; Boccaccini, A.R. Evaluation of electrospun poly(ε-caprolactone)/gelatin nanofiber mats containing clove essential oil for antibacterial wound dressing. Pharmaceutics 2019, 11, 570.
- Adeli, H.; Khorasani, M.T.; Parvazinia, M. Wound dressing based on electrospun PVA/chitosan/starch nanofibrous mats: Fabrication, antibacterial and cytocompatibility evaluation and in vitro healing assay. Int. J. Biol. Macromol. 2019, 122, 238–254.
- Wang, S.; Yan, F.; Ren, P.; Li, Y.; Wu, Q.; Fang, X.; Chen, F.; Wang, C. Incorporation of metal-organic frameworks into electrospun chitosan/poly (vinyl alcohol) nanofibrous membrane with enhanced antibacterial activity for wound dressing application. Int. J. Biol. Macromol. 2020, 158, 9–17.
- Kalalinia, F.; Taherzadeh, Z.; Jirofti, N.; Amiri, N.; Foroghinia, N.; Beheshti, M.; Bazzaz, B.S.F.; Hashemi, M.; Shahroodi, A.; Pishavar, E.; et al. Evaluation of wound healing efficiency of vancomycin-loaded electropunk chitosan/poly ethylene oxide nanofibers in full thickness wound model of rat. Int. J. Biol. Macromol. 2021, 177, 100–110.
- Amiri, N.; Ajami, S.; Shahroodi, A.; Jannatabadi, N.; Amiri Darban, S.; Fazly Bazzaz, B.S.; Pishavar, E.; Kalalinia, F.; Movaffagh, J. Teicoplanin-loaded chitosan-PEO nanofibers for local antibiotic delivery and wound healing. Int. J. Biol. Macromol. 2020, 162, 645–656.
- Dong, W.H.; Liu, J.X.; Mou, X.J.; Liu, G.S.; Huang, X.W.; Yan, X.; Ning, X.; Ning, X.; Russell, S.J.; Long, Y.Z. Performance of polyvinyl pyrrolidone-isatis root antibacterial wound dressings produced in situ by handheld electrospinner. Colloids Surf. B 2020, 188, 110766.
- Qin, M.; Mou, X.J.; Dong, W.H.; Liu, J.X.; Liu, H.; Dai, Z.; Huang, X.W.; Wang, N.; Yan, X. In situ electrospinning wound healing films composed of zein and clove essential oil. Macromol. Mater. Eng. 2020, 305, 1–6.
- Yue, Y.; Gong, X.; Jiao, W.; Li, Y.; Yin, X.; Si, Y.; Yu, J.; Ding, B. In-situ electrospinning of thymol-loaded polyurethane fibrous membranes for waterproof, breathable, and antibacterial wound dressing application. J. Colloid Interface Sci. 2021, 592, 310–318.
- Xu, H.; Xu, X.; Li, S.; Song, W.-L.; Yu, D.-G.; Annie Bligh, S.W. The effects of drug heterogeneous distributions with core-sheath nanostructures on its sustained release profiles. Biomolecules 2021, 11, 1330.
- Xiaoxia, X.; Jing, S.; Dongbin, X.; Yonggang, T.; Jingke, Z.; Hulai, W. Realgar nanoparticles inhibit migration, invasion and metastasis in a mouse model of breast cancer by suppressing matrix metalloproteinases and angiogenesis. Curr. Drug Deliv. 2020, 17, 148–158.
- Eskiler, G.G.; Cecener, G.; Dikmen, G.; Egeli, U.; Tunca, B. Talazoparib loaded solid lipid nanoparticles: Preparation, characterization and evaluation of the therapeutic efficacy in vitro. Curr. Drug Deliv. 2019, 16, 511–529.
- Tan, G.Z.; Zhou, Y. Electrospinning of biomimetic fibrous scaffolds for tissue engineering: A review. Int. J. Polym. Mater. Polym. Biomater. 2019, 69, 947–960.
- Islam, M.S.; Ang, B.C.; Andriyana, A.; Afifi, A.M. A review on fabrication of nanofibers via electrospinning and their applications. SN Appl. Sci. 2019, 1, 1–16.
- Yang, X.; Wang, J.; Guo, H.; Liu, L.; Xu, W.; Duan, G. Structural design toward functional materials by electrospinning: A review. E-Polymers 2020, 20, 682–712.