Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Veterinary Sciences
Anaplasma phagocytophilum and Anaplasma ovis, tick-borne pathogens with zoonotic potential, have been detected in small ruminants in Europe and North America in the past. However, knowledge about the distribution of these pathogens in the German small ruminant population is scarce. These intracellular bacteria cause tick borne fever and ovine anaplasmosis, respectively.
- Anaplasma phagocytophilum
- Anaplasma ovis
- tick-borne fever
- ovine anaplasmosis
- sheep
- goat
- Germany
1. Introduction
Across Europe and North America, sheep and goats can become infected with obligate intracellular bacteria of the genus Anaplasma. Whereas Anaplasma phagocytophilum is widespread in many European countries, an infection with Anaplasma ovis mainly occurs in the Mediterranean Basin [1]. However, reports about the focal occurrence of A. ovis in Central European countries like Hungary, Slovakia and Germany are increasing [2][3][4]. Both pathogens are also present in the US sheep population, but detailed information about the dissemination is lacking [5][6].
Wild ruminants may act as a reservoir for both pathogens in Europe and North America [3][7][8][9][10][11]. The transmission of Anaplasma spp. usually happens through tick bites [1]. The main vectors of A. phagocytophilum are Ixodes ricinus in Europe, as well as Ixodes scapularis, Ixodes pacificus and Ixodes spinipalpis in North America [1][11][12][13]. Different tick species belonging to the genera Dermacentor, Rhipicephalus and Hyalomma are considered to transmit A. ovis [13][14]. In recent years, A. ovis was also found in sheep keds (Melophagus ovinus) but their vector competence remains doubtful [15][16].
The replication of A. phagocytophilum takes place within the vacuoles of neutrophil granulocytes and sometimes also lymphocytes [17]. This causes granulocytic anaplasmosis in many domestic animals, such as horses [18][19], cattle [20][21], dogs [22][23] and cats [24][25], and also in humans [12]. In small ruminants, A. phagocytophilum results in tick-borne fever (TBF) and affected animals suffer from high fever, anorexia and dullness [26][27][28]. Neutropenia and thrombocytopenia are the haematological key findings in affected sheep and goats [26][29]. Immunosuppression causes a high susceptibility to secondary infections like Mannheima haemolytica and Bibersteina trehalosi and leads to respiratory distress in lambs [30][31]. Furthermore, A. phagocytophilum favours co-infections with staphylococcal bacteria which cause tick-pyaemia with polyarthritis [29]. TBF and co-infections can be fatal for lambs [29][30][31]. However, mild courses of A. phagocytophilum were reported but affected lambs had reduced growth rates [28]. Goats show similar clinical signs to sheep after an infection with A. phagocytophilum, but to a lesser extent [26][32][33].
Anaplasma ovis mainly affects the ovine and caprine erythrocytes [34] but can also be found in wild ungulates like roe deer (Capreolus capreolus) and red deer (Cervus elaphus) [7][15][35]. Humans rarely become infected [36]. The pathogen causes ovine anaplasmosis especially in sheep in poor health [37]. Main clinical signs in sheep are fever, severe anaemia, extreme weakness, anorexia, and weight loss [34][37][38][39]. Moreover, haemoglobinuria and icteric carcasses were also described in sheep infected with A. ovis [2][39]. An acute infection results in decreased values of red blood cells, haemoglobin and packed cell volume [40]. Although the same signs are described for goats as for sheep, A. ovis appears to be more pathogenic for goats [41].
In Germany, A. phagocytophilum was identified in I. ricinius across the country with detection rates between 1.9% and 5.4% [42][43][44]. Although A. phagocytophilum has been well described in domestic animals [19][20][25][45] and wild ungulates [9][46], knowledge of the occurrence of the pathogen in German sheep and goat flocks is still limited. A molecular investigation revealed an infection rate of 4% (n = 255) in sheep from Northern Germany [8] and a clinical case of TBF was described in a goat from western Germany [47]. Recently, A. phagocytophilum was detected by PCR in five sheep flocks located in the southern part of the country [4]. In the same study, A. ovis was identified for the first time in a German sheep flock.
2. Animals
Serum samples from sheep and goats were available from a Q fever study conducted from winter 2017 to spring 2018, and details were described elsewhere [48]. In brief, the specimens were collected from 3178 small ruminants (2731 sheep, 447 goats) within 71 flocks located in five federal states: Schleswig-Holstein (SH), Lower Saxony (LS), North Rhine-Westphalia (NRW), Baden-Wuerttemberg (BW) and Bavaria (BAV) (Figure 1).
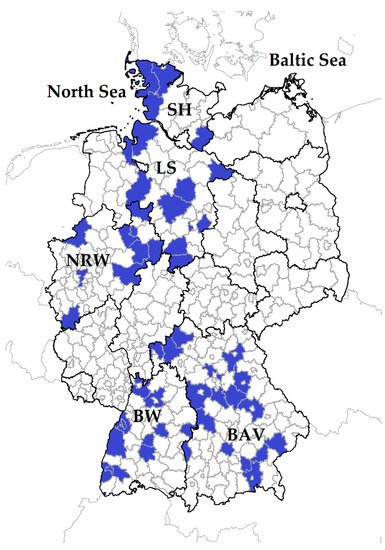
Figure 1. In total, 71 small ruminant flocks were sampled in five German federal states: Schleswig-Holstein: SH; Lower Saxony: LS, North Rhine-Westphalia: NRW, Baden-Wuerttemberg: BW, Bavaria: BAV. Districts with participating farms are coloured in blue.
These states have the largest sheep populations within Germany and the farms were selected based on the owners’ willingness to participate in the study. The number of required samples to determine the intra-flock prevalence (IFP) was calculated on the assumption of 3% expected prevalence, 95% confidence interval, 80% power and 5% precision. A maximum of 44 animals per flock were sampled. In sheep flocks with goats, the sample size for the goats was calculated under the same assumptions as for sheep. Individual ear tag number, species (sheep or goat), sex and age of every sampled animal were recorded.
3. Occurrence of Anaplasma spp. in German Small Ruminant Flocks
In total, antibodies against Anaplasma spp. were detected in 70 out of 71 small ruminant flocks. The IFP ranged between 0% and 97.7%. However, the level of IFP did not differ in principle between the federal states (p > 0.05). There was only one sheep flock in BAV without antibodies to Anaplasma species.
Overall, the mean antibody levels against Anaplasma spp. in sheep from the northern federal states, SH and LS, were significantly lower compared to NRW, BW and BAV (Figure 2). Among the northern federal states, there was no difference in the sheep’s antibody response (p > 0.05). Moreover, the mean antibody levels were also not statistically different between BW vs. NRW and BAV vs. NRW (p > 0.05), but sheep from BAV had significantly lower Anaplasma spp. values than animals from BW.
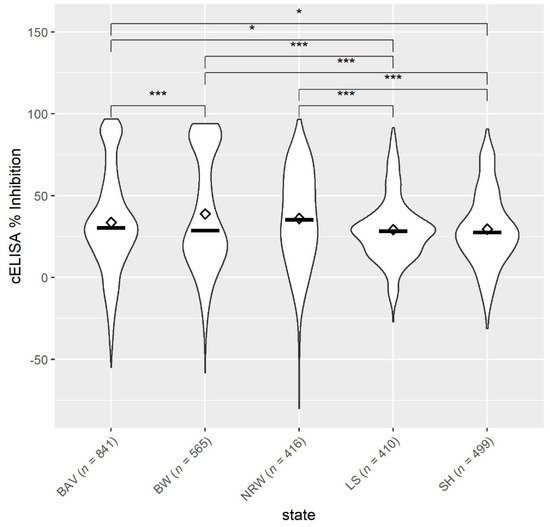
Figure 2. Mean ◊ and median—values of antibodies to Anaplasma spp. from sheep (n = 2731, 71 flocks) located in five German federal states: BAV = Bavaria, BW = Baden-Wuerttemberg, NRW = North Rhine-Westphalia, LS = Lower Saxony, SH = Schleswig-Holstein. * p < 0.05. *** p < 0.001.
4. Univariable Analysis
4.1. Risk Factors at Animal Level for Exposure to Anaplasma spp.
Sheep had a 2.5 times higher risk acquiring an Anaplasma spp. infection than goats. Females had a 37% increased chance of being seropositive, but the likelihood of antibody detection in young animals (≤2 years) was reduced by one quarter.
4.2. Risk Factors at Flock Level for Exposure to Anaplasma spp.
At flock level, only landscape conservation and cats had a significant p-value. Small ruminant flocks used for landscape conservation were four times more likely to have an IFP of above 20%. Farms with cats had an almost four-fold higher risk of having more than 20% IFP than farms without cats.
5. Multivariable Analysis
5.1. Risk Factors at Animal Level for Exposure to Anaplasma spp.
The results of the multivariable analysis were in line with the univariable analysis and revealed no additional information (Table 1).
Table 1. Multivariable risk factor model at animal level for exposure to Anaplasma spp.
| Variable | Category | Odds Ratio (OR) | 95% Confidence Interval | p-Value | Quasilikelihood under the Independence Model Criterion (QIC) |
|---|---|---|---|---|---|
| Species | Goat | 2.525 | 1.443–4.417 | 0.001 | 4342.598 |
| Sex | Male | 1.378 | 1.029–1.846 | 0.032 | |
| Age | >2 years | 0.739 | 0.970–0.970 | 0.029 |
5.2. Risk Factors at Flock Level for Exposure to Anaplasma spp.
The resulting multivariate model at flock level included four risk factors (Table 2) which were all significant except for contact to deer.
Table 2. Multivariate risk factor model at flock level for the risk of having an Anaplasma spp. intra-flock seroprevalence of above 20%.
| Variable | Category | Odds Ratio (OR) | 95% Confidence Interval | C- p-Value | LR- p-Value | Akaike Information Criterion (AIC) |
|---|---|---|---|---|---|---|
| Landscape conservation | No | 5.348 | 1.026–7.877 | 0.047 | 0.0002 | 49.614 |
| Deer | No | 0.149 | 0.015–1.461 | 0.102 | ||
| Cats | No | 10.731 | 1.681–68.514 | 0.012 | ||
| Dogs | No | 166.328 | 3.606–>999.999 | 0.009 |
Small ruminant flocks which performed landscape conservation had about a five times higher risk of having more than 20% seropositive animals compared to flocks without this farming purpose. Observations of deer near the flock reduced the risk to less than one-sixth, while the presence of cats and dogs on the farms increased the probability of having an IFP above 20% 10-fold and 166-fold, respectively.
This entry is adapted from the peer-reviewed paper 10.3390/ani11102793
References
- Stuen, S. Haemoparasites in small ruminants in European countries: Challenges and clinical relevance. Small Rumin. Res. 2016, 142, 22–27.
- Hornok, S.; Elek, V.; de la Fuente, J.; Naranjo, V.; Farkas, R.; Majoros, G.; Foldvari, G. First serological and molecular evidence on the endemicity of Anaplasma ovis and Anaplasma marginale in Hungary. Vet. Microbiol. 2007, 122, 316–322.
- Víchová, B.; Majláthová, V.; Nováková, M.; Stanko, M.; Hviščová, I.; Pangrácová, L.; Chrudimský, T.; Čurlík, J.; Peťko, B. Anaplasma infections in ticks and reservoir host from Slovakia. Infect. Genet. Evol. 2014, 22, 265–272.
- Bauer, B.U.; Răileanu, C.; Tauchmann, O.; Fischer, S.; Ambros, C.; Silaghi, C.; Ganter, M. Anaplasma phagocytophilum and Anaplasma ovis—Emerging pathogens in the German sheep population. Pathogens 2021. submitted.
- Mason, K.L.; Gonzalez, M.V.; Chung, C.; Mousel, M.R.; White, S.N.; Taylor, J.B.; Scoles, G.A. Validation of an improved Anaplasma antibody competitive ELISA for detection of Anaplasma ovis antibody in domestic sheep. J. Vet. Diagn. Investig. 2017, 29, 763–766.
- Gorman, J.K.; Hoar, B.R.; Nieto, N.C.; Foley, J.E. Evaluation of Anaplasma phagocytophilum infection in experimentally inoculated sheep and determination of Anaplasma spp. seroprevalence in 8 free-ranging sheep flocks in California and Oregon. Am. J. Vet. Res. 2012, 73, 1029–1034.
- de la Fuente, J.; Ruiz-Fons, F.; Naranjo, V.; Torina, A.; Rodriguez, O.; Gortazar, C. Evidence of Anaplasma infections in European roe deer (Capreolus capreolus) from southern Spain. Res. Vet. Sci. 2008, 84, 382–386.
- Scharf, W.; Schauer, S.; Freyburger, F.; Petrovec, M.; Schaarschmidt-Kiener, D.; Liebisch, G.; Runge, M.; Ganter, M.; Kehl, A.; Dumler, J.S.; et al. Distinct host species correlate with Anaplasma phagocytophilum ankA gene clusters. J. Clin. Microbiol. 2011, 49, 790–796.
- Kauffmann, M.; Rehbein, S.; Hamel, D.; Lutz, W.; Heddergott, M.; Pfister, K.; Silaghi, C. Anaplasma phagocytophilum and Babesia spp. in roe deer (Capreolus capreolus), fallow deer (Dama dama) and mouflon (Ovis musimon) in Germany. Mol. Cell. Probes 2017, 31, 46–54.
- de la Fuente, J.; Atkinson, M.W.; Naranjo, V.; de Mera, I.G.F.; Mangold, A.J.; Keating, K.A.; Kocan, K.M. Sequence analysis of the msp4 gene of Anaplasma ovis strains. Vet. Microbiol. 2007, 119, 375–381.
- Dugat, T.; Lagrée, A.-C.; Maillard, R.; Boulouis, H.-J.; Haddad, N. Opening the black box of Anaplasma phagocytophilum diversity: Current situation and future perspectives. Front. Cell. Infect. Microbiol. 2015, 5, 61.
- Matei, I.A.; Estrada-Pena, A.; Cutler, S.J.; Taussat, M.V.; Varela-Castro, L.; Potkonjak, A.; Zeller, H.; Mihalca, A.D. A review on the eco-epidemiology and clinical management of human granulocytic anaplasmosis and its agent in Europe. Parasites Vectors 2019, 12, 599.
- Rymaszewska, A.; Grenda, S. Bacteria of the genus Anaplasma—Characteristics of Anaplasma and their vectors: A review. Vet. Med. 2008, 53, 573–584.
- Friedhoff, K.T. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp. Parassitologia 1997, 39, 99–109.
- Hornok, S.; De La Fuente, J.; Biró, N.; de Mera, I.G.F.; Meli, M.L.; Elek, V.; Gönczi, E.; Meili, T.; Tánczos, B.; Farkas, R.; et al. First molecular evidence of Anaplasma ovis and Rickettsia spp. in keds (Diptera: Hippoboscidae) of sheep and wild ruminants. Vector Borne Zoonotic Dis. 2011, 11, 1319–1321.
- Zhang, Q.X.; Wang, Y.; Li, Y.; Han, S.Y.; Wang, B.; Yuan, G.H.; Zhang, P.Y.; Yang, Z.W.; Wang, S.L.; Chen, J.Y.; et al. Vector-borne pathogens with Veterinary and Public Health significance in Melophagus ovinus (Sheep Ked) from the Qinghai-Tibet Plateau. Pathogens 2021, 10, 249.
- Henniger, T.; Henniger, P.; Grossmann, T.; Distl, O.; Ganter, M.; von Loewenich, F.D. Congenital infection with Anaplasma phagocytophilum in a calf in northern Germany. Acta Vet. Scand. 2013, 55, 38.
- von Loewenich, F.D.; Stumpf, G.; Baumgarten, B.U.; Röllinghoff, M.; Dumler, J.S.; Bogdan, C. A case of Equine Granulocytic Ehrlichiosis provides molecular evidence for the presence of pathogenic Anaplasma phagocytophilum (HGE Agent) in Germany. Eur. J. Clin. Microbiol. Infect. Dis. 2003, 22, 303–305.
- Silaghi, C.; Liebisch, G.; Pfister, K. Genetic variants of Anaplasma phagocytophilum from 14 equine granulocytic anaplasmosis cases. Parasites Vectors 2011, 4, 161.
- Tegtmeyer, P.; Ganter, M.; von Loewenich, F.D. Simultaneous infection of cattle with different Anaplasma phagocytophilum variants. Ticks Tick Borne Dis. 2019, 10, 1051–1056.
- Silaghi, C.; Nieder, M.; Sauter-Louis, C.; Knubben-Schweizer, G.; Pfister, K.; Pfeffer, M. Epidemiology, genetic variants and clinical course of natural infections with Anaplasma phagocytophilum in a dairy cattle herd. Parasites Vectors 2018, 11, 20.
- Jensen, J.; Simon, D.; Escobar, H.M.; Soller, J.T.; Bullerdiek, J.; Beelitz, P.; Pfister, K.; Nolte, I. Anaplasma phagocytophilum in Dogs in Germany. Zoonoses Public Health 2007, 54, 94–101.
- Silaghi, C.; Kohn, B.; Chirek, A.; Thiel, C.; Nolte, I.; Liebisch, G.; Pfister, K. Relationship of molecular and clinical findings on Anaplasma phagocytophilum involved in natural infections of dogs. J. Clin. Microbiol. 2011, 49, 4413–4414.
- Hamel, D.; Bondarenko, A.; Silaghi, C.; Nolte, I.; Pfister, K. Seroprevalence and bacteriamea of Anaplasma phagocytophilum in cats from Bavaria and Lower Saxony (Germany). Berl. Munch. Tierarztl. Wochenschr. 2012, 125.
- Schäfer, I.; Kohn, B.; Müller, E. Anaplasma phagocytophilum in domestic cats from Germany, Austria and Switzerland and clinical/laboratory findings in 18 PCR-positive cats (2008–2020). J. Feline Med. Surg. 2021, 1098612X211017459.
- Gokce, H.I.; Woldehiwet, Z. Differential Haematological Effects of Tick-borne Fever in Sheep and Goats. J. Vet. Med. Ser. B 1999, 46, 105–115.
- Stuen, S.; Bergstrom, K.; Palmer, E. Reduced weight gain due to subclinical Anaplasma phagocytophilum (formerly Ehrlichia phagocytophila) infection. Exp. Appl. Acarol. 2002, 28, 209–215.
- Grøva, L.; Olesen, I.; Steinshamn, H.; Stuen, S. Prevalence of Anaplasma phagocytophilum infection and effect on lamb growth. Acta Vet. Scand. 2011, 53, 30.
- Sargison, N.; Edwards, G. Tick infestations in sheep in the UK. Practice 2009, 31, 58–65.
- Overås, J.; Lund, A.; Ulvund, M.J.; Waldeland, H. Tick-borne fever as a possible predisposing factor in septicaemic pasteurellosis in lambs. Vet. Rec. 1993, 133, 398.
- Daniel, R.G.; Carson, A.; Evans, C.; Cookson, R.; Wessels, M. Pathological observations of tick-borne fever and intercurrent bacterial infections in lambs. Vet. Rec. Case Rep. 2016, 4, e000357.
- Van Miert, A.S.J.P.A.M.; Van Duin, C.T.M.; Schotman, A.J.H.; Franssen, F.F. Clinical, haematological and blood biochemical changes in goats after experimental infection with tick-borne fever. Vet. Parasitol. 1984, 16, 225–233.
- Gray, D.; Webster, K.; Berry, J.E. Evidence of louping ill and tick-borne fever in goats. Vet. Rec. 1988, 122, 66.
- Jiménez, C.; Benito, A.; Arnal, J.L.; Ortín, A.; Gómez, M.; López, A.; Villanueva-Saz, S.; Lacasta, D. Anaplasma ovis in sheep: Experimental infection, vertical transmission and colostral immunity. Small Rumin. Res. 2019, 178, 7–14.
- Pereira, A.; Parreira, R.; Nunes, M.; Casadinho, A.; Vieira, M.L.; Campino, L.; Maia, C. Molecular detection of tick-borne bacteria and protozoa in cervids and wild boars from Portugal. Parasites Vectors 2016, 9, 251.
- Chochlakis, D.; Ioannou, I.; Tselentis, Y.; Psaroulaki, A. Human anaplasmosis and Anaplasma ovis variant. Emerg. Infect. Dis. 2010, 16, 1031–1032.
- Torina, A.; Galindo, R.C.; Vicente, J.; Di Marco, V.; Russo, M.; Aronica, V.; Fiasconaro, M.; Scimeca, S.; Alongi, A.; Caracappa, S.; et al. Characterization of Anaplasma phagocytophilum and A. ovis infection in a naturally infected sheep flock with poor health condition. Trop. Anim. Health Prod. 2010, 42, 1327–1331.
- Torina, A.; Alongi, A.; Naranjo, V.; Scimeca, S.; Nicosia, S.; Di Marco, V.; Caracappa, S.; Kocan, K.M.; de la Fuente, J. Characterization of anaplasma infections in Sicily, Italy. Ann. N. Y. Acad. Sci. 2008, 1149, 90–93.
- Lacasta, D.; Ferrer, L.M.; Sanz, S.; Labanda, R.; González, J.M.; Benito, A.Á.; Ruiz, H.; Rodríguez-Largo, A.; Ramos, J.J. Anaplasmosis Outbreak in Lambs: First Report Causing Carcass Condemnation. Animals 2020, 10, 1851.
- Yasini, S.; Khaki, Z.; Rahbari, S.; Kazemi, B.; Amoli, J.S.; Gharabaghi, A.; Jalali, S. Hematologic and clinical aspects of experimental ovine anaplasmosis caused by Anaplasma ovis in Iran. Iran. J. Parasitol. 2012, 7, 91–98.
- Stuen, S.; Longbottom, D. Treatment and control of chlamydial and rickettsial infections in sheep and goats. Vet. Clin. North. Am. Food Anim. Pr. 2011, 27, 213–233.
- Hartelt, K.; Oehme, R.; Frank, H.; Brockmann, S.O.; Hassler, D.; Kimmig, P. Pathogens and symbionts in ticks: Prevalence of Anaplasma phagocytophilum (Ehrlichia sp.), Wolbachia sp., Rickettsia sp., and Babesia sp. in Southern Germany. Int. J. Med. Microbiol. Suppl. 2004, 293, 86–92.
- Hildebrandt, A.; Krämer, A.; Sachse, S.; Straube, E. Detection of Rickettsia spp. and Anaplasma phagocytophilum in Ixodes ricinus ticks in a region of Middle Germany (Thuringia). Ticks Tick-Borne Dis. 2010, 1, 52–56.
- May, K.; Strube, C. Prevalence of Rickettsiales (Anaplasma phagocytophilum and Rickettsia spp.) in hard ticks (Ixodes ricinus) in the city of Hamburg, Germany. J. Parasitol. Res. 2014, 113, 2169–2175.
- Chirek, A.; Silaghi, C.; Pfister, K.; Kohn, B. Granulocytic anaplasmosis in 63 dogs: Clinical signs, laboratory results, therapy and course of disease. J. Small Anim. Pract. 2018, 59, 112–120.
- Silaghi, C.; Fröhlich, J.; Reindl, H.; Hamel, D.; Rehbein, S. Anaplasma phagocytophilum and Babesia species of Sympatric Roe Deer (Capreolus capreolus), Fallow Deer (Dama dama), Sika Deer (Cervus nippon) and Red Deer (Cervus elaphus) in Germany. Pathogens 2020, 9, 968.
- Langenwalder, D.B.; Schmidt, S.; Gilli, U.; Pantchev, N.; Ganter, M.; Silaghi, C.; Aardema, M.L.; von Loewenich, F.D. Genetic characterization of Anaplasma phagocytophilum strains from goats (Capra aegagrus hircus) and water buffalo (Bubalus bubalis) by 16S rRNA gene, ankA gene and multilocus sequence typing. Ticks Tick Borne Dis. 2019, 10, 101267.
- Wolf, A.; Prüfer, T.L.; Schoneberg, C.; Campe, A.; Runge, M.; Ganter, M.; Bauer, B.U. Prevalence of Coxiella burnetii in German sheep flocks and evaluation of a novel approach to detect an infection via preputial swabs at herd-level. Epidemiol. Infect. 2020, 148, e75.
This entry is offline, you can click here to edit this entry!
