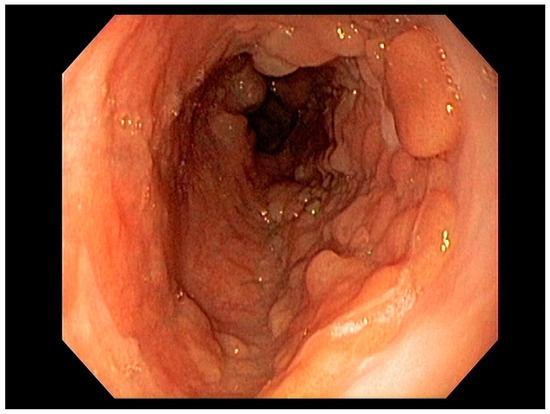
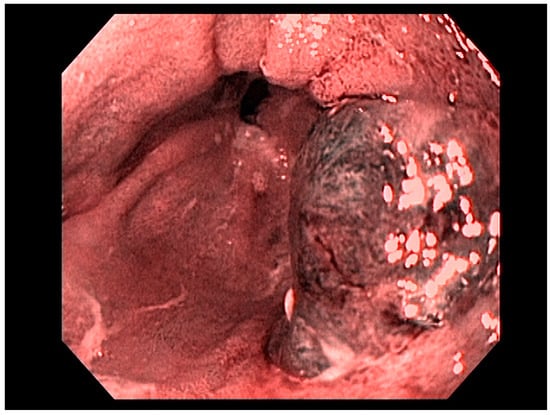
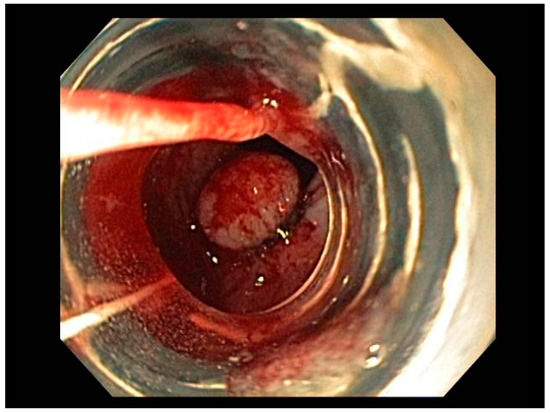
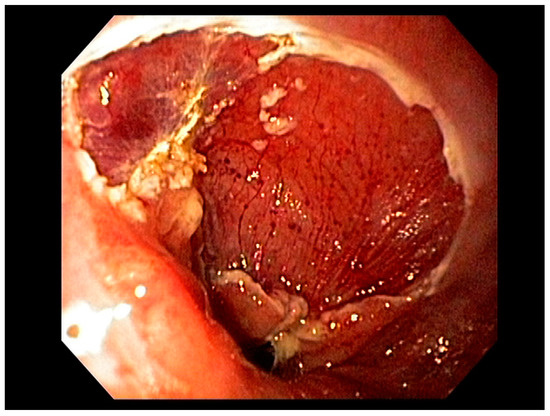
3.1. Endoscopic Therapeutic Options for Barrett’s Esophagus Associated Dysplasia
Therapeutic options are based upon the stage of the disease at diagnosis.
Barrett’s esophagus with low- or high-grade dysplasia (HGD) without visible lesions can be treated with radio-frequency ablation (RFA) or cryoablation therapies [
34,
44]. A randomized, controlled trial demonstrated that (RFA) for Barrett’s esophagus with HGD was superior to surveillance with respect to the outcome of progression to esophageal adenocarcinoma but did not find any difference in progression to cancer among patients with low-grade dysplasia (LGD) [
34]. A European, multicenter, randomized, controlled trial in a large group of patients with Barrett’s esophagus and LGD proved that RFA prevents progression to cancer in patients with LGD [
45]. After RFA for BE dysplasia, there is >30% chance of recurrence within 5 years, but most recurrences are responsive to further endoscopic therapy [
46].
Visible, discrete mucosal lesions within the Barrett’s segment, including high-grade dysplasia, T1a adenocarcinoma, and T1b adenocarcinoma with favorable histological features [
47], can be approached with endoscopic resection techniques. EMR is a widely utilized endoscopic resection technique, performed using either the “Cap-assisted EMR (Cap-EMR)” technique or multiband mucosectomy (MBM) techniques with the primary goal of assessing the depth of invasion (pathological T-stage) of a visible lesion that is suspicious for intramucosal adenocarcinoma or high-grade dysplasia; see . EMR and ESD are performed with intent of “staging” but can indeed be curative if the resection margins are “negative” or free of cancer, but the primary goal in Barrett’s esophagus is to obtain an accurate pathological T-stage of the lesion. This concept of “Staging-EMR” is demonstrated by studies that have shown a change in pathological diagnosis and/or T-staging noted in 20–30% of patients, both upstaging and downstaging, based on the pathology from the EMR specimen [
48], see .
Figure 5. Band-assisted mucosectomy.
Figure 6. Endoscopic Mucosal Rejection (EMR) T1 esophageal cancer; resection site.
3.2. Endoscopic Resection Techniques for Early-Stage Esophageal Cancer (T1a and T1b)
Endoscopic resection techniques applied to T1a and T1b mucosal cancer with favorable histological features include EMR or ESD. As mentioned earlier, EMR is primarily a “staging procedure” that provides the most accurate T-stage for mucosal lesions that are likely T1 based on EUS and/or optical chromoendoscopy [
49,
50]. EMR is a technique that involves submucosal injection of saline or a colloidal solution mixed with a dye such as methylene blue to create a “submucosal cushion”, followed by resection using a snare, and may be considered for lesions less than 20 mm. Techniques such as Cap-EMR, MBM, and ESD may be employed depending on anatomical location [
51,
52].
Cap-assisted EMR involves submucosal injection to lift the target lesion from the mucosa, isolating the lesion within a transparent “cap” mounted on the tip of the endoscope, gentle suction, followed by resection of the lesion using the pre-deployed snare within the cap using electrocautery [
53]. MBM involves suction of target tissue into an endoscopic band-ligation device and deployment of a band over the target lesion followed by resection of the tissue with electrocautery snaring [
54]. EMR can be either stepwise/complete (sEMR) or focal (fEMR), with focal EMR often combined with RFA. Piecemeal EMR has a high local recurrence and therefore is combined with RFA to ablate the residual BE epithelium [
55]. In theory, ESD is an en-bloc resection technique with the goal being an R0 resection and is therefore a more appealing endoscopic therapeutic option.
ESD is a more complex technique that may be considered for lesions less than 20 mm. Larger lesions may be amenable to this technique depending on availability of advanced expertise and resources [
56]. Specifically, this technique involves the creation of a submucosal tract to dissect lesions restricted to the mucosa (T1a or selected T1b), for complete resection. Factors including lesion size less than approximately 20 mm, anatomical stage (T1a), and middle thoracic and lower thoracic esophageal locations are favorable variables for successful ESD, demonstrating curative resection in some studies [
20,
56]. Additionally, T1a m2, T1a m3, and select T1b lesions with favorable histological features [
57] are amenable to ESD, with comparable outcomes to esophagectomy [
58,
59]. For these types of lesions, current evidence shows that EMR and ESD are comparable in terms of complication rates, referral to surgery, positive margins, lymph node positivity, local recurrence, and metachronous cancer. When compared to piecemeal EMR resection, ESD may offer some advantages, but data are limited [
60,
61]. Meta-analyses comparing both of these therapies revealed comparable outcomes in terms of Barrett’s eradication, low rates of recurrence, and have shown non-inferiority of EMR to ESD for therapy of Barrett’s related cancer or GEJ neoplasia, with similar complication rates [
15,
62].
3.3. Multimodality Therapy for Locally Advanced Esophageal Cancer
For locally advanced tumors (T2 or with nodal involvement), a combination of neoadjuvant chemoradiation therapy and surgical therapy can be used, with systemic chemotherapy being reserved for metastatic disease (Stage IV) with possible consolidation chemoradiation to sites of disease involvement if the patient responds well [
63].
Based on current guidelines, esophagectomy for operable non-metastatic patients with T1b or greater primary lesions and/or any nodal disease should be performed with one of several techniques including: transhiatal, transthoracic, three field, and, increasingly, minimally invasive approaches [
64]. Meta-analyses of minimally invasive esophagectomy, which includes laparoscopic/thoracoscopic and robotic approaches, have shown no difference in survival and improved or no difference in complications. Robotic-assisted esophagectomy is becoming increasingly popular, with demonstrated reduction in cardiac and pulmonary complications when compared with open esophagectomy. Technologic advances in the minimally invasive and robotic platforms are also rapidly developing and may allow for further innovation with these techniques [
65,
66]. While surgery alone can be considered for early-stage, low-risk adenocarcinoma (<3 cm and well differentiated) and early-stage squamous cell carcinoma, trimodality therapy with neoadjuvant chemoradiation followed by surgery is now a preferred treatment pathway for more advanced disease and requires multidisciplinary management [
67]. Less commonly, postoperative chemoradiation for pathologic T3 and/or node positive disease may be indicated if a GEJ cancer was treated with preoperative chemotherapy, while palliative radiotherapy may be indicated for treatment of significant dysphagia or bleeding. In definitive treatment of inoperable patients or those who decline esophagectomy, concurrent chemoradiation should be prioritized, followed by imaging and endoscopic surveillance to determine clinical complete response [
68]. While clinical outcomes and morbidity for these patients are still suboptimal, there are a growing number of long-term survivors after definitive chemoradiation and investigations are being made into radiotherapy dose escalation, proton therapy, and inclusion of targeted therapy [
69,
70,
71,
72].
Three-dimensional conformal planning for radiation therapy can be utilized; however, more institutions have implemented intensity-modulated radiation therapy (IMRT) to reduce cardiac and lung dosing, which allows for a simultaneous integrated boost (SIB) or sequential boost techniques [
73,
74,
75]. This approach is important as cardiac dose is increasingly seen as an independent risk factor for reduced survival [
76] and has been correlated with excess G3+ cardiac toxicity [
77,
78].
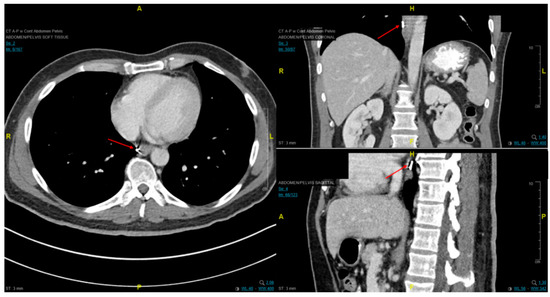
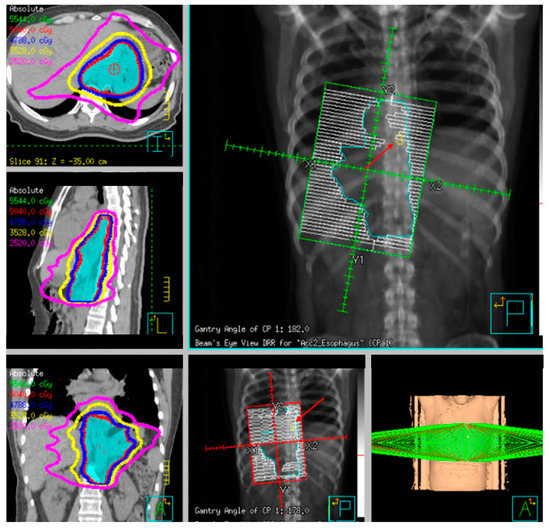
Fiducial markers can be placed endoscopically to delineate the extent of disease with high technical success with a small risk of migration [
79,
80,
81]. Stable fiducial markers improve the reliability of target volume delineation and assessment of respiratory tumor motion with four-dimensional CT (4D-CT) simulation as a direct visual correlate of tumor extent. Combined with fused PET/CT, fiducials reduce the margins for treatment planning due to improved confidence of accurately defined gross tumor volumes (GTVs) [
82] and facilitate daily image-guided radiation therapy (IGRT) during treatment [
83], see and . Moreover, 4D-CT imaging at simulation has a greater benefit in GEJ and gastric tumors due to the propensity of respiratory motion [
84] and aids in internal target volume construction and planning target volume expansions during treatment planning. Radiation treatments generally are conventionally fractionated (1.8–2.0 Gy daily); however, previously mentioned SIB techniques treating at 2.2–2.25 Gy daily can push dose to gross disease past 60 Gy. If given sequential neoadjuvant chemotherapy, the extent of original disease is often included in the clinical target volume receiving 4500–5040 cGy and boost is only directed to residual gross disease. Clinical target volumes extend 3–4 cm craniocaudal and 1 cm radially and are edited off anatomical structures that are clinically uninvolved such as vertebral bodies, trachea, aorta, lung, and pericardium. For middle and upper thoracic tumors, the at-risk periesophageal and adjacent mediastinal lymph nodes and, for distal esophageal and GEJ tumors, the celiac lymph nodes are covered. Multi-institutional consensus contouring guidelines for IMRT in esophageal and gastroesophageal tumors have been published [
85].
Figure 7. Axial, coronal, and sagittal CT images demonstrating EUS-placed fiducial (red arrow) proximal to esophageal tumor.
Figure 8. Example radiotherapy plan for image-guided treatment with fiducial contoured (red arrow) and within the planning target volume (PTV).
3.4. Neoadjuvant Radiation Therapy Alone and Adjuvant Therapy
Neoadjuvant treatment carries the theoretical gains of tumor downstaging, margin attenuation, and improved control of regional disease. However, limited benefit has been observed across several randomized trials investigating the role of radiotherapy alone in preoperative or adjuvant treatment of esophageal cancer. Early studies on patients who received 4000 cGy followed by surgery 1–4 weeks after found no difference in resectability or survival compared to surgery alone [
86,
87]. A trial from the European Organization for Research and Treatment of Cancer (EORTC) also demonstrated no difference in survival, but noted a lower rate of local failure with the addition of radiotherapy to 46% from 67% [
88]. More recent studies that reported improved survival included patients that received chemotherapy but had an imbalance of lower stage tumors conflicting the interpretation of the results [
89]. These findings have been confirmed in meta-analyses that demonstrate no statistical difference in survival, or at best, the suggestion of a very modest clinical benefit of <4% with neoadjuvant radiotherapy alone [
90,
91].
Recommendations for postoperative or adjuvant treatment are generally driven by adverse pathological features such as positive lymph nodes, positive margins, or locally advanced disease found at time of surgery. While the goal of local therapy is to reduce the risk of local recurrence, often, toxicity is greater due to larger treatment volumes, efficacy is reduced due to a hypoxic tumor bed, and dose is limited by the surgical anastomosis. In two historic series, adjuvant radiotherapy alone showed no survival benefit compared to observation [
92,
93]. However, similar to preoperative treatment, there are trends towards improved local control after delivery of 4500–5500 cGy [
93] after subtotal resection [
92]. Retrospective data and population-level analyses show a benefit of adjuvant chemoradiation in pathologically node positive [
94] but not node negative disease [
95]. The benefit of the addition of chemotherapy to adjuvant radiation is currently being investigated in a phase II/III protocol seeking to add paclitaxel and cisplatin/nedaplatin to 50.4–54.0 Gy of radiotherapy for pathological stage IIB-III disease [
96].
3.5. Preoperative Chemoradiation
For locally advanced resectable disease, the CROSS trial (Chemoradiotherapy for Oesophageal Cancer Followed by Surgery Study) established a new paradigm towards total neoadjuvant treatment with chemoradiation followed by surgical resection in esophageal cancer [
97]. Other comparisons to neoadjuvant chemotherapy alone in the German POET trial [
98] and the NeoRes trial [
99] demonstrated improvements, with pathological downstaging with the addition of 30 and 40 Gy of chemoradiation, but did not translate to statistically significant survival differences, likely limited by increased toxicity in the experimental arms with concurrent cisplatin/flurouracil. Within CROSS, radiation therapy was delivered to 4140 in 1.8 daily fractions with concurrent paclitaxel/carboplatin and was compared to surgery alone. Neoadjuvant chemoradiation was associated with a 25-month improvement in median overall survival (49 vs. 24 months) and was notable for a 47% 5-year survival rate and 29% overall pCR rate that was significantly different between histologies (28% adenocarcinoma vs. 49% squamous) [
100]. Additionally, preoperative chemoradiation reduced hematogenous metastases (35% vs. 29%) and peritoneal carcinomatosis (14% vs. 4%). There was no difference in operative mortality between the arms or long-term quality of life, with all endpoints declining after surgery and subsequently improved and stabilized between 6 and 12 months afterwards [
101].
The CROSS trial excluded patients with tumor diameter greater than 8 cm and patients who had lost more than 10% of their original body weight. In these patients, upfront chemotherapy alone can be considered as they are at high risk of clinical decline during chemoradiation. Therapeutic response to chemotherapy may enable a reduction in irradiation volumes to mitigate pulmonary toxicity with concurrent taxols or motivate one to switch to an alternative regimen before or during chemoradiation. This strategy may also be considered in patients with advanced primary tumor (T4) or regional nodal burden (bulky nodes or cN2+ disease) as they are at higher risk of distant progression and theoretically may derive greater benefit from intensification of systemic therapy. The outcomes from the CROSS trial remain some of the best to date with this strategy, and similar patterns were noted in the Chinese multicenter NEOCRTEC5010 trial that demonstrated improved median survival (66 vs. 100 months), disease-free survival, and R0 resection rate with neoadjuvant vinorelbine/cisplatin with 40 Gy compared to surgery alone [
102]. Future strategies seek to compare FOLFOX to paclitaxel-carboplatin with concurrent neoadjuvant radiation for both squamous and adenocarcinoma in the PROTECT-1402 trial (Preoperative Chemoradiation for Resectable Esophageal and Junctional Cancer) [
103]. Given the advances in surgical and radiotherapy techniques and corresponding declines in perioperative morbidity and mortality, trimodality therapy is now a preferred approach.
3.6. Definitive Radiation and Chemoradiation
Historically, definitively treated patients generally carry significant comorbidities precluding surgery or are opposed to the morbidity of resection. Alternatively, the surgical options for patients diagnosed with cervical esophageal cancer are often limited and these patients are treated within the paradigm of head and neck cancer with definitive chemoradiation. The use of radiation alone in the modern era is generally considered to be palliative, with 5-year survival of <10% [
104,
105], while the importance of concurrent chemotherapy has been demonstrated historically by RTOG 8501 where, at 5 years, no survival was noted with dose escalated 64 Gy radiotherapy alone versus 27% survival with 50 Gy and concurrent four cycles of fluorouracil and cisplatin [
106]. Initial forays of radiotherapy dose escalation in the definitive treatment of esophageal cancer showed disappointing clinical results. INT 0123 investigated chemoradiation with cisplatin/fluorouracil and compared 5040 cGy to dose escalation to 6480 cGy [
107]. A total of 236 patients were enrolled with T1-4, N0-1 disease with 85% squamous cell carcinoma. Overall, the target volumes for this trial were relatively large, with 5-cm superior/inferior and 2-cm radial esophageal expansion as well as a 2-cm isometric expansion from tumor for the boost to gross disease. The dose escalation arm experienced a 10% treatment-related G5 toxicity rate; however, 64% of the mortality occurred prior to surpassing the dose of the control arm, potentially implicating the toxic combination of cisplatin and fluorouracil. Median survival approximated 15 months; however, 2-year locoregional failure was greater than 50%. RTOG 9207 was a phase I/II study that utilized the same control arm as INT 0123 but dose-escalated through the use of predominantly high dose rate (HDR) brachytherapy with iridium-192 to 15 Gy in three weekly fractions [
108]. Unfortunately, toxicity from this strategy was high, with 12% fistula rates, 24% G4 toxicity, and 10% treatment-related deaths. Subsequently, the brachytherapy dose was dropped to 10 Gy in two weekly fractions and no fistulas were noted in the 10 patients treated with the intermediate dose. However, overall clinical outcomes remained poor, with 1-year survival of 50% and 37% local control.
With a goal of improving on the near 50% local failure rate seen in previous studies as well as overcoming high rates of persistent disease [
107,
108,
109], dose escalation in the modern era appears to be more tolerable but still carries an elevated risk of toxicity as well as minimal evidence of clinical benefit. In preliminary results, both a sequential boost to 60 Gy with cisplatin/docetaxel [
110] and simultaneous integrated boost to 61.2 Gy with carboplatin/paclitaxel [
111] failed to show improvement in overall or progression free survival, despite the latter regimen carrying a 13% rate of G4 toxicity and 10% treatment-related toxicity in the high dose arm. MD Anderson has published results with their SIB technique of a high dose gross tumor volume (GTV) plus 3 mm and a planning target volume (PTV) margin of 5 mm with concurrent chemotherapy, demonstrating encouraging 66% local control at 2 years, acute G3 toxicity in 23%, and only stricture-related late G3 toxicity in 7% [
112].
Newer strategies include proton therapy, which carries dosimetric benefits and higher relative biological effectiveness than photons due to the physical nature of the particle. Early retrospective data have not demonstrated an oncological benefit in comparison to photon therapy [
113]; however, there is some evidence of a decrease in treatment-related toxicity which, in larger prospective studies, may carry clinical significance [
114]. More work remains to further elucidate the benefits from these techniques.
3.7. Therapy for Metastatic Esophageal Cancer
Palliative radiotherapy is commonly delivered for esophageal obstruction in metastatic patients as a significant improvement in patient quality of life. Other indications are severe pain, chronic blood loss, or nausea due to tumor mass effect. While external beam radiotherapy and self-expanding metal stents (SEMS) to palliate dysphagia remain more common palliative techniques, growing literature supports the safe use of intraluminal brachytherapy for durable palliation with caution for fistulation or stenosis [
115].
A meta-analysis of 53 studies (mostly RCTs) on palliation of dysphagia in inoperable esophageal cancer concluded that SEMS insertion is safe, effective, and quicker in palliating dysphagia compared to other modalities. The authors added that, “Brachytherapy might be a suitable alternative to SEMS in providing a survival advantage and possibly a better quality of life” [
116].
The goal of chemotherapy in patients with stage IV metastatic esophageal cancer is to improve survival and quality of life; several chemotherapeutic agents have been tested and used in the past several decades and proven to be effective in achieving this goal. Unfortunately, survival has remained poor and rarely surpasses one year.
Combination of 5 fluorouracil (5 fu) and platinum agents is an acceptable first-line treatment option and is considered the standard of care in this setting; other regimens including paclitaxel with platinum regimen, irinotecan plus 5-fu are recommended as well. Three drug combinations such as modified DCF (Docetaxel, Cisplatin, 5 fu) are usually given to patients with high volume disease, young age, and good performance status who might benefit from a higher response rate. Single-agent treatment is recommended for patients with low volume disease and or poor performance status.
Several targeted therapies have been tested as front-line and in combination without significant improvement in overall survival until the ToGA trial was conducted. The ToGA trial was a randomized phase III trial where patients with Her-2 expressing tumors were randomized to chemotherapy with or without trastuzumab (Her-2 monoclonal antibody); patients who received the combination had better overall survival, leading to the approval of this combination in this patient population [
117].
Recently, at the European Society of Medical oncology meeting in 2020 (ESMO 2020), three important trials were presented incorporating immune checkpoint inhibitors in the front-line treatment of esophageal and gastric cancer. The CheckMate 649 trial evaluated nivolumab plus chemotherapy versus chemotherapy alone as first-line treatment in patients with non-HER-2-positive advanced gastric and GE junction adenocarcinoma. The addition of Nivolumab improved overall survival and progression-free survival in patients with PD-L1 combined positive score (CPS) ≥5 tumors. Improvements were also observed in patients with PD-L1 CPS ≥1 tumors and in the overall patient population. In another study, the ATTRACTION 4 trial, which was performed only in Asian patients and the primary endpoints were designed for all-comers, rather than a specific CPS value, first-line treatment with nivolumab plus chemotherapy improved the co-primary progression-free survival endpoint, but not overall survival [
118].
The third trial, presented at the same meeting, the KEYNOTE 590 trial, examined first-line chemotherapy with or without pembrolizumab in patients with squamous cell carcinoma of the esophagus, adenocarcinoma of the esophagus, or Siewert-type GE junction adenocarcinoma. It demonstrated that pembrolizumab plus chemotherapy improved overall survival in patients with squamous cell carcinoma of the esophagus with PD-L1 CPS ≥10 tumors, all squamous cell carcinomas, all patients with CPS ≥10, and the study population as a whole. Progression-free survival was also improved. It is expected that these trials will change the landscape of the treatment of esophageal cancer worldwide [
118].
Several chemotherapy and targeted agents have been studied in the second-line treatment of esophageal and gastric cancer and beyond. Chemotherapy improves survival compared to placebo, single-agent paclitaxel with or without ramucirumab, a monoclonal antibody targeting the VEGF pathway, which is a commonly used second-line regimen with overall survival approaching 9 months. Single-agent ramucirumab, irinotecan, and immune checkpoint inhibitors are all approved for the second-line treatment as well, with modest benefits. Patients with microsatellite instable disease should be treated with immune check point inhibitors at any point during the course of their disease [
118].
Several ongoing trials are testing other targeted therapies and are beyond the scope of this review.
Patients with inoperable esophageal cancer and with high-grade dysphagia were randomized to receive a self-expandable metal stent (SEMS) alone (Group I), versus a combination of SEMS followed by external beam radiation (over 2 weeks) (Group II). Dysphagia scores improved significantly in both groups following stent insertion. However, dysphagia relief was more sustained in Group II than in Group I (7 vs. 3 months,
p = 0.002), and overall median survival was significantly higher in Group II than in Group I [
119]. A recent meta-analysis of several RCTs evaluating the efficacy of SEMS alone vs. SEMS combined with radiotherapy concluded that the combination of SEMS and radiation significantly improves the overall survival as well as leading to improvements in quality of life scores [
120].
In summary, the current trend in the literature shows that the best oncological outcomes are associated with trimodality therapy with neoadjuvant chemoradiation. This has been supported by recent meta-analyses that conclude that compared with neoadjuvant chemotherapy, neoadjuvant chemoradiotherapy should be recommended, with a significant long-term survival benefit in patients with cancer of the esophagus or the GEJ [
121,
122]. While treatments and outcomes are improving, a large proportion of patients fail locally, and addition of biologically targeted agents or local therapy intensification may prove to be beneficial.
For patients who are considered to be surgical candidates, neoadjuvant chemotherapy with weekly carboplatin and paclitaxel in combination with radiation followed by surgical resection is the standard of care. This treatment is based on the CROSS trial, where neoadjuvant therapy resulted in improved overall survival compared to esophagectomy alone [
97]. Definitive chemotherapy and radiation is a reasonable option for patients who are not surgical candidates or with cervical/mid-esophageal SCC. On rare occasions, patients who are thought to have T1N0 disease are upstaged during esophagectomy; the role of adjuvant therapy in this setting is not clear and carries significant toxicity. The addition of Her-2 targeted therapy with trastuzumab (Her-2 antibody) to the neoadjuvant therapy did not result in improved outcome [
123]. Several ongoing trials are currently evaluating the addition of immune checkpoint inhibitors in both the neoadjuvant and the adjuvant settings. These results are highly anticipated.
As previously described, neoadjuvant radiation therapy in addition to chemotherapy plays an important role in the multimodality treatment of locally advanced esophageal cancer. Fiducial markers have been integrated into the management of multiple malignancies to guide more precise delivery of radiation therapy (RT).