Obsessive–compulsive disorder (OCD) is a debilitating mental health disorder characterized by intrusive thoughts (obsessions) and repetitive behaviors (compulsions). Dysbiosis, an imbalance in the gut microbial composition, has been associated with various health conditions, including mental health disorders, autism, and inflammatory diseases. While the exact mechanisms underlying OCD remain unclear, researchers present a growing body of evidence suggesting a potential link between dysbiosis and the multifaceted etiology of OCD, interacting with genetic, neurobiological, immunological, and environmental factors.
- OCD
- obsessive–compulsive disorder
- microbiota
- gut
- gut–brain axis
- probiotics
- fecal transplants
1. Introduction
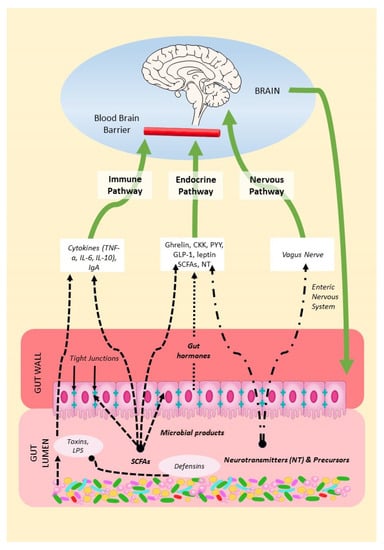
2. Mechanisms of the Microbiota–Gut–Brain Axis (MGBA)
3. Microbial Reprogramming Strategies
3.1. Prebiotics, Probiotics, and Postbiotics
3.2. Fecal Microbiota Transplants
This entry is adapted from the peer-reviewed paper 10.3390/ijms241511978
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5™, 5th ed.; American Psychiatric Publishing, Inc.: Arlington, VA, USA, 2013; p. 947.
- Oren, E.; Dar, R.; Liberman, N. Obsessive-compulsive tendencies are related to a maximization strategy in making decisions. Front. Psychol. 2018, 9, 778.
- Nestadt, G.; Kamath, V.; Maher, B.S.; Krasnow, J.; Nestadt, P.; Wang, Y.; Bakker, A.; Samuels, J. Doubt and the decision-making process in obsessive-compulsive disorder. Med. Hypotheses 2016, 96, 1–4.
- Pushkarskaya, H.; Tolin, D.; Ruderman, L.; Kirshenbaum, A.; Kelly, J.M.; Pittenger, C.; Levy, I. Decision-making under uncertainty in obsessive-compulsive disorder. J. Psychiatr. Res. 2015, 69, 166–173.
- Murphy, D.L.; Timpano, K.R.; Wheaton, M.G.; Greenberg, B.D.; Miguel, E.C. Obsessive-compulsive disorder and its related disorders: A reappraisal of obsessive-compulsive spectrum concepts. Dialogues Clin. Neurosci. 2010, 12, 131–148.
- Jalal, B.; Chamberlain, S.R.; Sahakian, B.J. Obsessive-compulsive disorder: Etiology, neuropathology, and cognitive dysfunction. Brain Behav. 2023, 13, e3000.
- Johnson, P.M.; Kenny, P.J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010, 13, 635–641.
- Mattheisen, M.; Samuels, J.F.; Wang, Y.; Greenberg, B.D.; Fyer, A.J.; McCracken, J.T.; Geller, D.A.; Murphy, D.L.; Knowles, J.A.; Grados, M.A.; et al. Genome-wide association study in obsessive-compulsive disorder: Results from the OCGAS. Mol. Psychiatry 2015, 20, 337–344.
- Frick, L.; Pittenger, C. Microglial Dysregulation in OCD, Tourette Syndrome, and PANDAS. J. Immunol. Res. 2016, 2016, 8606057.
- Mahjani, B.; Bey, K.; Boberg, J.; Burton, C. Genetics of obsessive-compulsive disorder. Psychol. Med. 2021, 51, 2247–2259.
- Purty, A.; Nestadt, G.; Samuels, J.F.; Viswanath, B. Genetics of obsessive-compulsive disorder. Indian J. Psychiatry 2019, 61, S37–S42.
- Fawcett, E.J.; Power, H.; Fawcett, J.M. Women are at greater risk of OCD than men: A meta-analytic review of OCD prevalence worldwide. J. Clin. Psychiatry 2020, 81, 13075.
- Grassi, G.; Cecchelli, C.; Vignozzi, L.; Pacini, S. Investigational and experimental drugs to treat obsessive-compulsive disorder. J. Exp. Pharmacol. 2020, 12, 695–706.
- Foa, E.B. Cognitive behavioral therapy of obsessive-compulsive disorder. Dialogues Clin. Neurosci. 2010, 12, 199–207.
- Kellner, M. Drug treatment of obsessive-compulsive disorder. Dialogues Clin. Neurosci. 2010, 12, 187–197.
- Fineberg, N.A.; Gale, T.M.; Sivakumaran, T. A review of antipsychotics in the treatment of obsessive compulsive disorder. J. Psychopharmacol. 2006, 20, 97–103.
- Eisen, J.L.; Goodman, W.K.; Keller, M.B.; Warshaw, M.G.; DeMarco, L.M.; Luce, D.D.; Rasmussen, S.A. Patterns of remission and relapse in obsessive-compulsive disorder: A 2-year prospective study. J. Clin. Psychiatry 1999, 60, 346–351.
- Ferguson, J.M. SSRI antidepressant medications: Adverse effects and tolerability. Prim Care Companion J. Clin. Psychiatry 2001, 3, 22–27.
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836.
- Benjamin, J.L.; Hedin, C.R.; Koutsoumpas, A.; Ng, S.C.; McCarthy, N.E.; Prescott, N.J.; Pessoa-Lopes, P.; Mathew, C.G.; Sanderson, J.; Hart, A.L.; et al. Smokers with active Crohn’s disease have a clinically relevant dysbiosis of the gastrointestinal microbiota. Inflamm. Bowel Dis. 2012, 18, 1092–1100.
- Kang, S.; Denman, S.E.; Morrison, M.; Yu, Z.; Dore, J.; Leclerc, M.; McSweeney, C.S. Dysbiosis of fecal microbiota in Crohn’s disease patients as revealed by a custom phylogenetic microarray. Inflamm. Bowel Dis. 2010, 16, 2034–2042.
- Hou, M.; Xu, G.; Ran, M.; Luo, W.; Wang, H. APOE-ε4 carrier status and gut microbiota dysbiosis in patients with Alzheimer disease. Front. Neurosci. 2021, 15, 619051.
- Pistollato, F.; Sumalla Cano, S.; Elio, I.; Masias Vergara, M.; Giampieri, F.; Battino, M. Role of gut microbiota and nutrients in amyloid formation and pathogenesis of Alzheimer disease. Nutr. Rev. 2016, 74, 624–634.
- Shabbir, U.; Arshad, M.S.; Sameen, A.; Oh, D.H. Crosstalk between gut and brain in Alzheimer’s disease: The role of gut microbiota modulation strategies. Nutrients 2021, 13, 690.
- Scheperjans, F.; Aho, V.; Pereira, P.A.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov. Disord. 2015, 30, 350–358.
- Madore, C.; Leyrolle, Q.; Lacabanne, C.; Benmamar-Badel, A.; Joffre, C.; Nadjar, A.; Layé, S. Neuroinflammation in Autism: Plausible Role of Maternal Inflammation, Dietary Omega 3, and Microbiota. Neural. Plast. 2016, 2016, 3597209.
- De Angelis, M.; Francavilla, R.; Piccolo, M.; De Giacomo, A.; Gobbetti, M. Autism spectrum disorders and intestinal microbiota. Gut Microbes 2015, 6, 207–213.
- Li, Q.; Zhou, J.M. The microbiota-gut-brain axis and its potential therapeutic role in autism spectrum disorder. Neuroscience 2016, 324, 131–139.
- Ding, H.T.; Taur, Y.; Walkup, J.T. Gut microbiota and autism: Key concepts and findings. J. Autism. Dev. Disord. 2017, 47, 480–489.
- Zhu, F.; Guo, R.; Wang, W.; Ju, Y.; Wang, Q.; Ma, Q.; Sun, Q.; Fan, Y.; Xie, Y.; Yang, Z.; et al. Transplantation of microbiota from drug-free patients with schizophrenia causes schizophrenia-like abnormal behaviors and dysregulated kynurenine metabolism in mice. Mol. Psychiatry 2020, 25, 2905–2918.
- Frazier, T.H.; DiBaise, J.K.; McClain, C.J. Gut microbiota, intestinal permeability, obesity-induced inflammation, and liver injury. JPEN J. Parenter. Enteral. Nutr. 2011, 35, 14S–20S.
- Musso, G.; Gambino, R.; Cassader, M. Obesity, diabetes, and gut microbiota: The hygiene hypothesis expanded? Diabetes Care 2010, 33, 2277–2284.
- Jess, T. Microbiota, antibiotics, and obesity. N. Engl. J. Med. 2014, 371, 2526–2528.
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut microbiota’s effect on mental health: The gut-brain axis. Clin. Pract. 2017, 7, 987.
- Iannone, L.F.; Preda, A.; Blottière, H.M.; Clarke, G.; Albani, D.; Belcastro, V.; Carotenuto, M.; Cattaneo, A.; Citraro, R.; Ferraris, C.; et al. Microbiota-gut brain axis involvement in neuropsychiatric disorders. Expert Rev. Neurother. 2019, 19, 1037–1050.
- Sommer, F.; Bäckhed, F. The gut microbiota--masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238.
- Sudo, N. Microbiome, HPA axis and production of endocrine hormones in the gut. Adv. Exp. Med. Biol. 2014, 817, 177–194.
- de Weerth, C. Do bacteria shape our development? Crosstalk between intestinal microbiota and HPA axis. Neurosci. Biobehav. Rev. 2017, 83, 458–471.
- Sudo, N. Role of microbiome in regulating the HPA axis and its relevance to allergy. Chem. Immunol. Allergy 2012, 98, 163–175.
- Skott, E.; Yang, L.L.; Stiernborg, M.; Söderström, Å.; Rȕegg, J.; Schalling, M.; Forsell, Y.; Giacobini, M.; Lavebratt, C. Effects of a synbiotic on symptoms, and daily functioning in attention deficit hyperactivity disorder—A double-blind randomized controlled trial. Brain Behav. Immun. 2020, 89, 9–19.
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156.
- Wu, Z.; Xu, Q.; Wang, Q.; Chen, Y.; Lv, L.; Zheng, B.; Yan, R.; Jiang, H.; Shen, J.; Wang, S.; et al. The impact of dietary fibers on Clostridioides difficile infection in a mouse model. Front. Cell. Infect. Microbiol. 2022, 12, 1028267.
- Chen, K.; Chen, H.; Faas, M.M.; de Haan, B.J.; Li, J.; Xiao, P.; Zhang, H.; Diana, J.; de Vos, P.; Sun, J. Specific inulin-type fructan fibers protect against autoimmune diabetes by modulating gut immunity, barrier function, and microbiota homeostasis. Mol. Nutr. Food Res. 2017, 61, 1601006.
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071.
- Wang, L.; Christophersen, C.T.; Sorich, M.J.; Gerber, J.P.; Angley, M.T.; Conlon, M.A. Low relative abundances of the mucolytic bacterium Akkermansia muciniphila and Bifidobacterium spp. in feces of children with autism. Appl. Environ. Microbiol. 2011, 77, 6718–6721.
- Demirci, M.; Tokman, H.B.; Uysal, H.K.; Demiryas, S.; Karakullukcu, A.; Saribas, S.; Cokugras, H.; Kocazeybek, B.S. Reduced Akkermansia muciniphila and Faecalibacterium prausnitzii levels in the gut microbiota of children with allergic asthma. Allergol. Immunopathol. 2019, 47, 365–371.
- Klaenhammer, T.R.; Altermann, E.; Pfeiler, E.; Buck, B.L.; Goh, Y.J.; O’Flaherty, S.; Barrangou, R.; Duong, T. Functional genomics of probiotic Lactobacilli. J. Clin. Gastroenterol. 2008, 42 Pt 2 (Suppl. 3), S160–S162.
- Maassen, C.B.; Claassen, E. Strain-dependent effects of probiotic lactobacilli on EAE autoimmunity. Vaccine 2008, 26, 2056–2057.
- Martín, R.; Delgado, S.; Maldonado, A.; Jiménez, E.; Olivares, M.; Fernández, L.; Sobrino, O.J.; Rodríguez, J.M. Isolation of lactobacilli from sow milk and evaluation of their probiotic potential. J. Dairy Res. 2009, 76, 418–425.
- Moorthy, G.; Murali, M.R.; Devaraj, S.N. Lactobacilli facilitate maintenance of intestinal membrane integrity during Shigella dysenteriae 1 infection in rats. Nutrition 2009, 25, 350–358.
- Kantak, P.A.; Bobrow, D.N.; Nyby, J.G. Obsessive-compulsive-like behaviors in house mice are attenuated by a probiotic (Lactobacillus rhamnosus GG). Behav. Pharmacol. 2014, 25, 71–79.
- Tabouy, L.; Getselter, D.; Ziv, O.; Karpuj, M.; Tabouy, T.; Lukic, I.; Maayouf, R.; Werbner, N.; Ben-Amram, H.; Nuriel-Ohayon, M.; et al. Dysbiosis of microbiome and probiotic treatment in a genetic model of autism spectrum disorers. Brain Behav. Immun. 2018, 73, 310–319.
- Szklany, K.; Wopereis, H.; de Waard, C.; van Wageningen, T.; An, R.; van Limpt, K.; Knol, J.; Garssen, J.; Knippels, L.M.J.; Belzer, C.; et al. Supplementation of dietary non-digestible oligosaccharides from birth onwards improve social and reduce anxiety-like behaviour in male BALB/c mice. Nutr. Neurosci. 2020, 23, 896–910.
- Sanikhani, N.S.; Modarressi, M.H.; Jafari, P.; Vousooghi, N.; Shafei, S.; Akbariqomi, M.; Heidari, R.; Lavasani, P.S.; Yazarlou, F.; Motevaseli, E.; et al. The Effect of Lactobacillus casei consumption in improvement of obsessive-compulsive disorder: An animal study. Probiotics Antimicrob. Proteins 2020, 12, 1409–1419.
- Sunand, K.; Mohan, G.K.; Bakshi, V. Enrichment of gut ecosystem by daily supplementation of selective probiotic strains and probiotic complex in dysbiosis condition of autism. Int. J. Pharm. Res. 2021, 13, 4614–4628.
- Alghamdi, M.A.; Al-Ayadhi, L.; Hassan, W.M.; Bhat, R.S.; Alonazi, M.A.; El-Ansary, A. Bee Pollen and Probiotics May Alter Brain Neuropeptide Levels in a Rodent Model of Autism Spectrum Disorders. Metabolites 2022, 12, 562.
- Pochakom, A.; Mu, C.; Rho, J.M.; Tompkins, T.A.; Mayengbam, S.; Shearer, J. Selective probiotic treatment positively modulates the microbiota-gut-brain axis in the BTBR mouse model of autism. Brain Sci 2022, 12, 781.
- Sen, P.; Sherwin, E.; Sandhu, K.; Bastiaanssen, T.F.S.; Moloney, G.M.; Golubeva, A.; Fitzgerald, P.; Paula Ventura Da Silva, A.; Chruścicka-Smaga, B.; Olavarría-Ramírez, L.; et al. The live biotherapeutic Blautia stercoris MRx0006 attenuates social deficits, repetitive behaviour, and anxiety-like behaviour in a mouse model relevant to autism. Brain Behav. Immun. 2022, 106, 115–126.
- Kobliner, V.; Mumper, E.; Baker, S.M. Reduction in Obsessive Compulsive Disorder and self-injurious behavior with Saccharomyces boulardii in a child with autism: A case report. Integr. Med. 2018, 17, 38–41.
- Al-Ali, D.; Ahmed, A.; Shafiq, A.; McVeigh, C.; Chaari, A.; Zakaria, D.; Bendriss, G. Fecal microbiota transplants: A review of emerging clinical data on applications, efficacy, and risks (2015–2020). Qatar Med. J. 2021, 2021, 5.
- Kang, D.W.; Adams, J.B.; Coleman, D.M.; Pollard, E.L.; Maldonado, J.; McDonough-Means, S.; Caporaso, J.G.; Krajmalnik-Brown, R. Long-term benefit of microbiota transfer therapy on autism symptoms and gut microbiota. Sci. Rep. 2019, 9, 5821.
- Ananthaswamy, A. Faecal transplant eases symptoms of Parkinson’s disease. New Sci. 2011, 209, 8–9.
- Ianiro, G.; Segal, J.P.; Mullish, B.H.; Quraishi, M.N.; Porcari, S.; Fabiani, G.; Gasbarrini, A.; Cammarota, G. Fecal microbiota transplantation in gastrointestinal and extraintestinal disorders. Future Microbiol. 2020, 15, 1173–1183.
- Li, N.; Chen, H.; Cheng, Y.; Xu, F.; Ruan, G.; Ying, S.; Tang, W.; Chen, L.; Chen, M.; Lv, L.; et al. Fecal microbiota transplantation relieves gastrointestinal and autism symptoms by improving the gut microbiota in an open-label study. Front. Cell. Infect. Microbiol. 2021, 11, 759435.
- Wang, J.; Liu, X.; Li, Q. Interventional strategies for ischemic stroke based on the modulation of the gut microbiota. Front. Neurosci. 2023, 17, 1158057.
- Khanna, S.; Vazquez-Baeza, Y.; González, A.; Weiss, S.; Schmidt, B.; Muñiz-Pedrogo, D.A.; Rainey, J.F., 3rd; Kammer, P.; Nelson, H.; Sadowsky, M.; et al. Changes in microbial ecology after fecal microbiota transplantation for recurrent C. difficile infection affected by underlying inflammatory bowel disease. Microbiome 2017, 5, 55.
- Kang, D.W.; Adams, J.B.; Gregory, A.C.; Borody, T.; Chittick, L.; Fasano, A.; Khoruts, A.; Geis, E.; Maldonado, J.; McDonough-Means, S.; et al. Microbiota Transfer Therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: An open-label study. Microbiome 2017, 5, 10.
- Kilinçarslan, S.; Evrensel, A. The effect of fecal microbiota transplantation on psychiatric symptoms among patients with inflammatory bowel disease: An experimental study. Actas Esp. Psiquiatr. 2020, 48, 1–7. Available online: https://www.actaspsiquiatria.es/repositorio//22/123/ENG/22-123-ENG-1-7-952049.pdf (accessed on 9 July 2023).
- Wang, J.; Cao, Y.; Hou, W.; Bi, D.; Yin, F.; Gao, Y.; Huang, D.; Li, Y.; Cao, Z.; Yan, Y.; et al. Fecal microbiota transplantation improves VPA-induced ASD mice by modulating the serotonergic and glutamatergic synapse signaling pathways. Transl. Psychiatry 2023, 13, 17.
