Natural bioactive compounds have recently emerged as a current strategy for Alzheimer’s disease treatment. Carotenoids, including astaxanthin, lycopene, lutein, fucoxanthin, crocin and others are natural pigments and antioxidants, and can be used to treat a variety of diseases, including Alzheimer’s disease. However, carotenoids, as oil-soluble substances with additional unsaturated groups, suffer from low solubility, poor stability and poor bioavailability. Therefore, the preparation of various nano-drug delivery systems from carotenoids is a current measure to achieve efficient application of carotenoids. Different carotenoid delivery systems can improve the solubility, stability, permeability and bioavailability of carotenoids to a certain extent to achieve Alzheimer’s disease efficacy.
- carotenoids
- Alzheimer’s disease
- nanosized drug delivery systems
1. Introduction
2. Carotenoids in Alzheimer’s Disease Treatment
2.1. Pathogenesis of Alzheimer’s Disease
The etiology of Alzheimer’s disease remains unclear due to the multi-factorial nature of the disease’s process. The formation of amyloid plaques is a key factor in the development of AD. These plaques can aggregate into senile plaques on the outer surface of blood vessels and brain neurons, and in intracellular aggregations of neurofibrillary tangles generated from hyperphosphorylated tau [7][8][9][10][32]. Naturally, oxidative stress (OS) is a major feature of AD. The reasons why neurons are extremely sensitive to OS include the following: (1) the energy generated by oxidative phosphorylation in neuronal mitochondria is extremely important [33][34]; (2) about 20% of the oxygen generated by respiration is used by neurons, 1–2% of which is converted into reactive oxygen species to cause OS [35][36][37][38]; (3) metal ions in neurons accumulate and catalyze ROS production in the brain as the aging process progresses [39]; (4) polyunsaturated fatty acids in neurons are susceptible to oxidation [40]; and (5) neurons have relatively low levels of antioxidants and related enzymes [41][42].2.2. Overview of Carotenoids
Carotenoids (Figure 1) are the most prevalent class of isoprenoid yellow-orange pigments that can be synthesized by photosynthetic organisms and fungal microorganisms and bacteria [43][44]. It can be categorized into two main groups: (1) nonpolar carotenes, such as β-carotene and lycopene, which are the hydrocarbon compound that carry no functional groups [45][46]; (2) polar xanthophylls, for instance, astaxanthin, lutein, and canthaxanthin, the structures of which contain hydrogen, carbon, and oxygen [17]. Carotenoids can also be divided into pro- and non-pro vitamin A, which cannot be converted into retinoids [45]. Carotenoids are known to be efficient compounds due to their antioxidant properties and nontoxic nature, which can minimize the risk of age-related muscular disorders [47].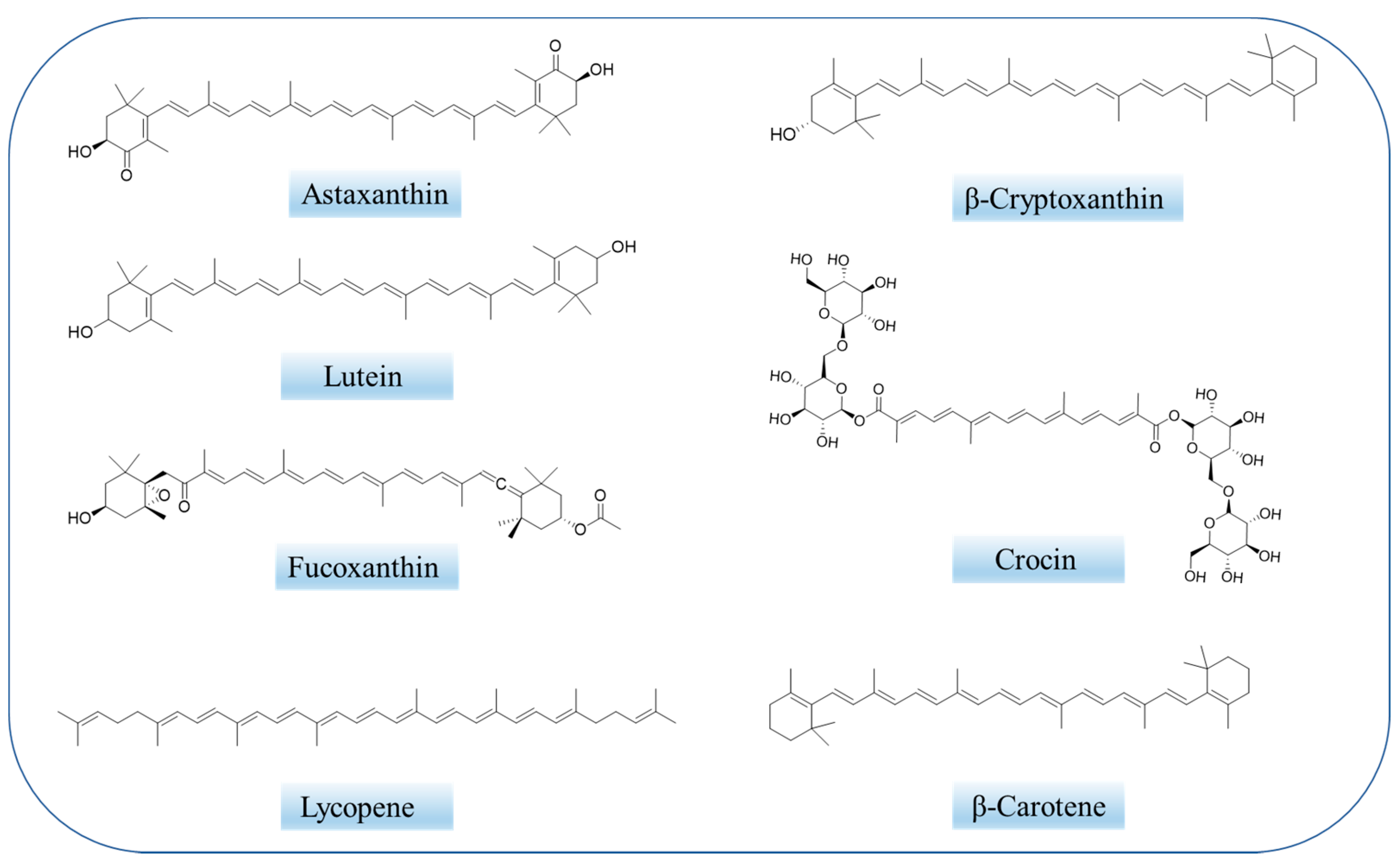
2.3. Therapeutic Mechanisms of Carotenoids on Alzheimer’s Disease
The interest in carotenoids has increased dramatically over the last decade due to their newly discovered activities, in particular, their neuroprotective properties. Neuroprotective mechanisms of carotenoids include antioxidant, anti-inflammatory, and anti-apoptotic activities, as well as the potential to promote neural plasticity. Although the exact molecular mechanisms of neurodegenerative diseases are still being elucidated, aging is considered as a primary risk factor for their development, including development of Alzheimer’s disease [48]. It is usually accepted that increased inflammation and oxidative stress within the brain contribute to neurodegeneration. The brain is usually susceptible to higher oxidative stress due to its high metabolic activity and the presence of various oxidized compounds. Oxidative stress can harm biomolecules (peptides, lipids and so on) and lead to neuronal dysfunction over time. The antioxidant activity of carotenoids is perhaps the known property responsible for its health benefit in prevention of neurodegenerative diseases. Carotenoids are known to act as singlet oxygen quenchers and free radical scavengers and are used to combat oxidative stress in organisms. Singlet oxygen quenching, important in photosynthesis, relies on the energy transfer between electrophilic singlet oxygen and the carotenoid skeleton. The scavenging rate increases with the conjugation length. There are three main types of carotenoid radical scavenging reactions: (1) electron transfer between free radicals and carotenoids resulting in the formation of carotenoid radical cations or carotenoid radical anions; (2) formation of free radical adducts; and (3) hydrogen atom transfer to form neutral carotenoid group [19].3. Carotenoid-Loaded Nanocarriers for Alzheimer’s Disease Therapy
The brain is a special organ that is protected by two major barriers, the blood–brain barrier (BBB) with its 20 m2 surface area, and the blood–cerebrospinal-fluid barrier (BCSFB) [49]. Because of the large molecular weight of carotenoids, they do not penetrate easily to the brain, which presents one of the most important challenges in the development of drugs for the central nervous system [50]. Recently, the development of nano-based drug delivery systems has created exciting opportunities for the prevention and treatment of AD. Heretofore-poorly-distributed drugs are now prepared using nano-drug delivery systems. The drug delivery system has an excellent interaction with endothelial microvascular cells at the blood–brain barrier and is capable of producing elevated drug concentrations in the cerebral parenchyma. First, nanocarriers can pass through the blood–brain barrier passively (through the direct plasma membrane) or actively (endocytosis, pinocytosis, etc.) through transmembrane channels. Secondly, functional groups on the surface of nanocarriers (polysorbate surfactant layer or covalent binding of apolipoprotein, etc.) can enhance the efficiency of the carrier system in penetrating through the blood–brain barrier. The nanocarrier system is easily phagocytized by mononuclear phagocyte system, and then degraded or metabolized by lysosomes. The chemical groups on the surface of inorganic nanomaterials can be metabolized by enzymes or non-enzymes. Organic nanomaterials may first decompose and then metabolize into smaller particles. In the liver, if too large to pass through the pores between the transcellular hepatic sinusoidal endothelial cells, small-particle-sized materials can pass through the pores into the perisinusoidal space and then into the hepatocytes, where they are subsequently passed by monooxygenases, transferases, esters metabolism by enzymes, and epoxide hydrolases [51].3.1. Polymeric Nanocarriers
3.1.1. Polymeric Micelles
Micelles are core-shell structures formed spontaneously by amphiphilic molecules in water [52]. Proper micelles can be obtained by adjusting the fraction of monomers in the bulk copolymer so that most hydrophobic drugs can be easily incorporated into the core of the micelles. The function of the micellar shell is to protect the drug from interactions with serum proteins and non-target cells. In addition, targeted drug delivery can be achieved by end-functionalization of micelles with block copolymers of peptides, sugars, and additional components. Nanoscale micelles minimize the clearance of micelles from the body, prolong the action time of the drug, and improve the bioavailability of the drug.3.1.2. Polymeric Nanoparticles
Polymer nanoparticles and micelles have some similar characteristics, such as loading efficiency, versatility, stimulus response (including light, temperature, enzyme, pH, and other biological and chemical agents) and so on [53]. Amphiphilic polymers with different structures, lengths and charges can be used to prepare polymer nanoparticles. They vary in size, shape and stability and can be used to encapsulate hydrophilic and hydrophobic drug molecules, including macromolecules such as carotenoids [28][31][54]. Polymer nanoparticles are widely used as biodegradable materials in the medical field. Commonly used polymers include polylactides, polyglycolides, poly-ε-caprolactone, and polyethylene glycol. Although these materials have been approved by the FDA for use in the medical field, they are not considered ideal for the treatment of central nervous system disorders due to their poor solubility and degradation in acidic byproducts. Acrylic polymer nanoparticles, especially poly (butyl cyanoacrylate) (PBCA) nanoparticles, have been widely used in the delivery of drugs in the central nervous system [55]. PBCA can be rapidly degraded in vivo to reduce toxicity due to polymer accumulation in the central nervous system. Drugs used to treat diseases of the central nervous system through the PBCA nanoparticle delivery system include doxorubicin, temozolomide, methotrexate, etc. [56][57].3.1.3. Dendrimers
Dendrimers are highly branched molecules with a 3D structure consisting of repetitive monomeric units with highly branched structures [58]. The modifiability of its surface structure gives dendrimers versatility, and the presence of a hydrophobic core enables encapsulation of genes, nucleic acids, and other drug molecules through electrostatic interactions or conjugation for the treatment of central nervous system disorders such as AD [50][59][60]. Polymeric dendrites have been developed for the treatment of Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, ischemic stroke and other central nervous system disorders.3.2. Lipid-Based Nanocarriers
3.2.1. Liposomes
One of the drug delivery systems that has received increasing attention is the liposome, which is a spherical vesicle composed of a unilamellar or multilamellar phospholipid bilayer [61]. Liposomes have excellent biocompatibility and biodegradability, low toxicity, and enable the targeted delivery of lipophilic and hydrophilic drugs. The greatest advantage of liposomes for central nervous system (CNS) delivery is that they can be easily surface-modified to prepare advanced liposomes such as immunoliposomes for targeted delivery [61].3.2.2. Solid Lipid Nanoparticles (SLNs)
SLN is normally composed of a lipid matrix in the solid state at room temperature and dispersed in water or a solution composed of surfactants for stabilization at body temperature. Fatty acids, cholesterol, monostearin, etc. are commonly used lipid matrices for the preparation of SLNs [62][63]. Solid lipid nanoparticles are produced from natural materials or natural lipids, are biocompatible, and do not affect the internal and external environment of cells after degradation, which makes them less immunogenic. At the same time, solid lipid nanoparticles are modest in size, flow rapidly in the blood, and are not easily absorbed by macrophages, thus facilitating the continuous release of therapeutic drugs in the body. Because solid lipid nanoparticle delivery systems are able to bypass P-glycoprotein by cell-by-cell percolation, they facilitate the penetration of lipophilic drugs across the blood–brain barrier. In fact, SLN can also bind apolipoprotein to target brain tissues [64][65].3.2.3. Nanostructured Lipid Carriers (NLCs)
To improve the inherent shortcomings of SLNs for high load efficiency, nanostructured lipid carriers came into being [66]. Nanostructured lipid carriers are composed of liquid and solid lipids (inner layer) and water emulsifiers (outer layer). They are transformed forms of SLN. The difference between SLN and NLC is that in NLC, 5–40% of the solid phase is exchanged with the liquid phase [67], liquid lipids or oils including fatty acid esters or alcohols such as 2-octanol are mixed with solid lipids [68][69], and lipids with different chain lengths, such as mono-, di-, and triglycerides, are also used to increase the space of the delivery system [70]; note that some hydrophobic drugs have better solubility in liquid lipids, which makes nanostructured lipid carriers more loading-efficient [71][72].3.3. Inorganic Nanocarriers
Inorganic nanocarriers include cerium dioxide, iron oxide, gold, inorganic quantum dots and so on. Inorganic nano-delivery systems have been used for drug delivery due to their excellent physical and chemical properties, including size, shape, surface functionality, chemical structure, and high specific surface area, as well as imaging capabilities, so they can play their therapeutic and diagnostic roles at the same time.3.4. Hybrid Nanocarriers
Hybrid nanocarriers are composed of lipid, organic, and inorganic polymers [73][74][75]. In general, the organic polymer or inorganic substance acts as the core, and the lipid layer acts as the shell. The outer lipid can inhibit the diffusion of water to the inner layer, thus delaying the degradation of the inner polymer and ensuring that the loaded drug molecules are slowly and continuously released. Hybrid nanocarriers have controllable drug release capacity, high loading efficiency, biocompatibility and biodegradability, and are good carriers for the treatment of central nervous system diseases [73].4. Different Nano-Encapsulated Carotenoids in Alzheimer’s Disease Therapy
4.1. Crocin and Crocetin
Crocin (Figure 1) and crocetin are mainly derived from saffron and the fruits of gardenia. Crocin, a water-soluble carotenoid, is composed of a conjugated polyene skeleton and sugar substituents at both ends. When the substituents at both ends are hydrogen atoms, it is a crocetin, which is insoluble in water. Numerous studies have shown that crocin and crocetin have disparate pharmacological effects, such as anti-Alzheimer, antioxidant, anti-tumor, anti-inflammatory, memory enhancing, antidepressant, etc. [76][77].4.2. Astaxanthin
Astaxanthin (AST, 3,3′-dihydroxy-β, β-carotene-4,4′-dione, Figure 1) is a xanthophyll-type carotenoid [78]. It is synthesized by algae, bacteria or yeasts and can also accumulate in birds, fish and crustaceans through the food chain [79]. Unlike other carotenoids, astaxanthin contains two ionone rings at each end of the carbon chain with long-chain conjugated double bonds. Their unique molecular configuration and size allow astaxanthin molecules to be inserted vertically into the phospholipid bilayer of the cell membrane, allowing astaxanthin to prevent lipid peroxidation and protect the integrity of the cell membrane [80]. It is by far the most powerful natural antioxidant found in nature. Its strong antioxidant effect is due to the effective stability of the ionone rings and polyene skeletons against free radicals. The mechanism of antioxidant activity in AST involves the absorption of free radicals into the polyene chain, providing electrons or forming chemical bonds with the active material [81].4.3. Lycopene
Lycopene, a natural carotenoid, is widely found in fruits as diverse as pink guavas, tomatoes and red-skinned watermelons [82][83]. In the clinic, lycopene is the subject of numerous studies investigating its anti-cancer, cardiovascular disease prevention, liver protection, and other alternative effects.4.4. Lutein
Lutein, a dietary carotenoid, comes from foods such as egg yolk, corn, kiwi, persimmon, and green vegetables. Its structure is conjugated polyene skeleton and ketone at both ends. Due to the particularity of its structure, it is easily affected by environmental factors such as temperature, light, oxygen and so on [84]. Similar to other carotenoids, it has antioxidant, anti-inflammatory and anti-tumor activity, and the potential to protect against several diseases, including Alzheimer’s disease and age-related macular degeneration.4.5. Fucoxanthin
Fucoxanthin, an orange pigment and one of the most abundant carotenoids in nature [85], is found in the chloroplasts of brown algae [86]. Recently, fucoxanthin has been reported to have a variety of biological activities, including anti-cancer, antioxidant, anti-angiogenesis, anti-diabetes, anti-obesity, anti-inflammatory, and anti-malaria activities [87]. Fucoxanthin has previously been reported to have neuroprotective effects against Alzheimer’s disease [88][89][90][91][92].5. Conclusions and Perspectives
5. Conclusions and Perspectives
Nowadays, it is clear that carotenoids have many benefits for health and positive nutritional effects and can reduce the risk of many diseases. However, there are some critical points to be considered: (1) Most carotenoids play a synergistic role when combined with other compounds, and the single form of carotenoids may not be effective, but from another perspective if two or more carotenoids are put together inside the nanocarrier there may be competition for absorption, which leads to lower bioavailability. (2) Carotenoids are unstable, and easy to transform into different compounds; therefore, the safety of carotenoids needs additional research. (3) The therapeutic effect varies from person to person, thus the effective dose is an unknown problem.
Carotenoid-based nano-drug delivery systems are feasible for effective disease prevention and treatment. This entreviewy presents a series of examples of carotenoid nano-delivery systems for Alzheimer’s disease. Each technology has its own strengths and limitations. To some extent, nano-delivery systems can improve the loading capacity, bioavailability, bioactivity, stability and solubility of carotenoids. In the author’s opinion, polymeric micelles are more suitable for the delivery of carotenoids. First, the polymer micelles can be adjusted to a suitable size to accommodate carotenoids of different sizes. Second, the modifiability and ease of modification of the polymer surface increases the functional properties of carotenoids. Finally, carotenoid polymers can make it easier to pass through the blood–brain barrier by adjusting the hydrophilic–lipophilic balance. However, industrial production of nanomedicines is still in its early stages. Safety and health concerns need to be explored in depth before widespread consumption. First, each process or material must be formally approved by regulatory authorities. However, the regulatory framework for the inclusion of nano-carriers in pharmaceutical products is still in flux. State agencies are expected to add initiatives and some legislation to regulate and monitor the proper development and application of nanoparticles in food and drug formulations.
In modern medicine, the idea of “synergy” between drug and carrier has attracted increased attention, seeking to preserve and improve the health benefits of various drugs in prevention and the treatment of many diseases. In the case of carotenoids, drug delivery systems can assist these bioactive compounds in exerting greater biological activity and stability. On the other hand, some nano-delivery systems can also play more functional roles, including targeted delivery to the brain or other organs, or overcome the blood–brain-barrier. Thirdly, since most of the nanocarriers are natural protein or polysaccharide components, they can provide the body with some needed nutrients to a certain extent and improve the efficiency of disease prevention and treatment.
References
- Paris, D.; Beaulieu-Abdelahad, D.; Bachmeier, C.; Reed, J.; Ait-Ghezala, G.; Bishop, A.; Chao, J.; Mathura, V.; Crawford, F.; Mullan, M. Anatabine lowers Alzheimer’s A beta production in vitro and in vivo. Eur. J. Pharmacol. 2011, 670, 384–391.
- Fratiglioni, L.; Launer, L.J.; Andersen, K.; Breteler, M.M.B.; Copeland, J.R.M.; Dartigues, J.F.; Lobo, A.; Martinez-Lage, J.; Soininen, H.; Hofman, A.; et al. Incidence of dementia and major subtypes in Europe: A collaborative study of population-based cohorts. Neurology 2000, 54, S10–S15.
- Lobo, A.; Launer, L.J.; Fratiglioni, L.; Andersen, K.; Di Carlo, A.; Breteler, M.M.B.; Copeland, J.R.M.; Dartigues, J.F.; Jagger, C.; Martinez-Lage, J.; et al. Prevalence of dementia and major subtypes in Europe: A collaborative study of population-based cohorts. Neurology 2000, 54, S4–S9.
- Jonsson, L.; Jonhagen, M.E.; Kilander, L.; Soininen, H.; Hallikainen, M.; Waldemar, G.; Nygaard, H.; Andreasen, N.; Winblad, B.; Wimo, A. Determinants of costs of care for patients with Alzheimer’s disease. Int. J. Geriatr. Psychiatry 2006, 21, 449–459.
- Citron, M. Strategies for disease modification in Alzheimer’s disease. Nat. Rev. Neurosci. 2004, 5, 677–685.
- Chon, S.-H.; Yang, E.-J.; Lee, T.; Song, K.-S. Beta-Secretase (BACE1) inhibitory and neuroprotective effects of p-terphenyls from Polyozellus multiplex. Food Funct. 2016, 7, 3834–3842.
- Golde, T.E. The A beta hypothesis: Leading us to rationally-designed therapeutic strategies for the treatment or prevention of Alzheimer disease. Brain Pathol. 2005, 15, 84–87.
- Hardy, J.; Selkoe, D.J. Medicine-The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356.
- Kabir, M.T.; Uddin, M.S.; Setu, J.R.; Ashraf, G.M.; Bin-Jumah, M.N.; Abdel-Daim, M.M. Exploring the Role ofPSENMutations in the Pathogenesis of Alzheimer’s Disease. Neurotox. Res. 2020, 38, 833–849.
- Kabir, M.T.; Uddin, M.S.; Zaman, S.; Begum, Y.; Ashraf, G.M.; Bin-Jumah, M.N.; Bungau, S.G.; Mousa, S.A.; Abdel-Daim, M.M. Molecular Mechanisms of Metal Toxicity in the Pathogenesis of Alzheimer’s Disease. Mol. Neurobiol. 2021, 58, 1–20.
- Cores, A.; Abril, S.; Michalska, P.; Duarte, P.; Olives, A.I.; Martin, M.A.; Villacampa, M.; Leon, R.; Menendez, J.C. Bisavenathramide Analogues as Nrf2 Inductors and Neuroprotectors in In Vitro Models of Oxidative Stress and Hyperphosphorylation. Antioxidants 2021, 10, 941.
- Bucciantini, M.; Leri, M.; Nardiello, P.; Casamenti, F.; Stefani, M. Olive Polyphenols: Antioxidant and Anti-Inflammatory Properties. Antioxidants 2021, 10, 1044.
- Loi, M.; Paciolla, C. Plant Antioxidants for Food Safety and Quality: Exploring New Trends of Research. Antioxidants 2021, 10, 972.
- Rodriguez-Yoldi, M.J. Anti-Inflammatory and Antioxidant Properties of Plant Extracts. Antioxidants 2021, 10, 921.
- Bolhassani, A.; Khavari, A.; Bathaie, S.Z. Saffron and natural carotenoids: Biochemical activities and anti-tumor effects. Biochim. Biophys. Acta-Rev. Cancer 2014, 1845, 20–30.
- Cong, L.; Wang, C.; Chen, L.; Liu, H.; Yang, G.; He, G. Expression of phytoene synthase1 and Carotene Desaturase crtl Genes Result in an Increase in the Total Carotenoids Content in Transgenic Elite Wheat (Triticum aestivum L.). J. Agric. Food Chem. 2009, 57, 8652–8660.
- Prakash, D.; Gupta, C. Carotenoids: Chemistry and Health Benefits. Phytochem. Nutraceut. Importance 2014, 181–195.
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Carotenoids: Importance in Daily Life-Insight Gained from EPR and ENDOR. Appl. Magn. Reson. 2021, 52, 1093–1112.
- Fiedor, J.; Burda, K. Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients 2014, 6, 466–488.
- Widomska, J.; Zareba, M.; Subczynski, W.K. Can Xanthophyll-Membrane Interactions Explain Their Selective Presence in the Retina and Brain? Foods 2016, 5, 7.
- Lakey-Beitia, J.; Doens, D.; Kumar, D.J.; Murillo, E.; Fernandez, P.L.; Rao, K.S.; Durant-Archibold, A.A. Anti-amyloid aggregation activity of novel carotenoids: Implications for Alzheimer’s drug discovery. Clin. Interv. Aging 2017, 12, 815–822.
- Bohn, T. Carotenoids and Markers of Oxidative Stress in Human Observational Studies and Intervention Trials: Implications for Chronic Diseases. Antioxidants 2019, 8, 179.
- Iddir, M.; Yaruro, J.F.P.; Cocco, E.; Hardy, E.M.; Appenzeller, B.M.R.; Guignard, C.; Larondelle, Y.; Bohn, T. Impact of Protein-Enriched Plant Food Items on the Bioaccessibility and Cellular Uptake of Carotenoids. Antioxidants 2021, 10, 1005.
- Young, A.J.; Lowe, G.L. Carotenoids-Antioxidant Properties. Antioxidants 2018, 7, 28.
- Kim, S.H.; Kim, M.S.; Lee, B.Y.; Lee, P.C. Generation of structurally novel short carotenoids and study of their biological activity. Sci. Rep. 2016, 6, 21987.
- Polyakov, N.E.; Leshina, T.V.; Salakhutdinov, N.F.; Konovalova, T.A.; Kispert, L.D. Antioxidant and redox properties of supramolecular complexes of carotenoids with beta-glycyrrhizic acid. Free Radic. Biol. Med. 2006, 40, 1804–1809.
- Polyakov, N.E.; Leshina, T.V.; Salakhutdinov, N.F.; Kispert, L.D. Host-guest complexes of carotenoids with beta-glycyrrhizic acid. J. Phys. Chem. B 2006, 110, 6991–6998.
- Polyakov, N.E.; Leshina, T.V.; Meteleva, E.S.; Dushkin, A.V.; Konovalova, T.A.; Kispert, L.D. Water Soluble Complexes of Carotenoids with Arabinogalactan. J. Phys. Chem. B 2009, 113, 275–282.
- Polyakov, N.E.; Kispert, L.D. Water soluble biocompatible vesicles based on polysaccharides and oligosaccharides inclusion complexes for carotenoid delivery. Carbohydr. Polym. 2015, 128, 207–219.
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Supramolecular Carotenoid Complexes of Enhanced Solubility and Stability-The Way of Bioavailability Improvement. Molecules 2019, 24, 3947.
- Apanasenko, I.E.; Selyutina, O.Y.; Polyakov, N.E.; Suntsova, L.P.; Meteleva, E.S.; Dushkin, A.V.; Vachali, P.; Bernstein, P.S. Solubilization and stabilization of macular carotenoids by water soluble oligosaccharides and polysaccharides. Archives Biochem. Biophys. 2015, 572, 58–65.
- Neve, R.L.; Robakis, N.K. Alzheimer’s disease: A re-examination of the amyloid hypothesis. Trends Neurosci. 1998, 21, 15–19.
- Dominiak, K.; Jarmuszkiewicz, W. The Relationship between Mitochondrial Reactive Oxygen Species Production and Mitochondrial Energetics in Rat Tissues with Different Contents of Reduced Coenzyme Q. Antioxidants 2021, 10, 533.
- Kolodziej, F.; O’Halloran, K.D. Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World? Antioxidants 2021, 10, 609.
- Olowe, R.; Sandouka, S.; Saadi, A.; Shekh-Ahmad, T. Approaches for Reactive Oxygen Species and Oxidative Stress Quantification in Epilepsy. Antioxidants 2020, 9, 990.
- Picazo, C.; Molin, M. Impact of Hydrogen Peroxide on Protein Synthesis in Yeast. Antioxidants 2021, 10, 952.
- Ullah, H.; Di Minno, A.; Santarcangelo, C.; Khan, H.; Daglia, M. Improvement of Oxidative Stress and Mitochondrial Dysfunction by beta-Caryophyllene: A Focus on the Nervous System. Antioxidants 2021, 10, 546.
- Woo, J.; Cho, H.; Seol, Y.; Kim, S.H.; Park, C.; Yousefian-Jazi, A.; Hyeon, S.J.; Lee, J.; Ryu, H. Power Failure of Mitochondria and Oxidative Stress in Neurodegeneration and Its Computational Models. Antioxidants 2021, 10, 229.
- Kaur, I.; Behl, T.; Aleya, L.; Rahman, M.H.; Kumar, A.; Arora, S.; Akter, R. Role of metallic pollutants in neurodegeneration: Effects of aluminum, lead, mercury, and arsenic in mediating brain impairment events and autism spectrum disorder. Environ. Sci. Pollut. Res. 2021, 28, 8989–9001.
- Shichiri, M. The role of lipid peroxidation in neurological disorders. J. Clin. Biochem. Nutr. 2014, 54, 151–160.
- Lima, L.W.; Nardi, S.; Santoro, V.; Schiavon, M. The Relevance of Plant-Derived Se Compounds to Human Health in the SARS-CoV-2 (COVID-19) Pandemic Era. Antioxidants 2021, 10, 1031.
- Ofosu, F.K.; Mensah, D.-J.F.; Daliri, E.B.-M.; Oh, D.-H. Exploring Molecular Insights of Cereal Peptidic Antioxidants in Metabolic Syndrome Prevention. Antioxidants 2021, 10, 518.
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324.
- Huang, J.J.; Lin, S.; Xu, W.; Cheung, P.C.K. Occurrence and biosynthesis of carotenoids in phytoplankton. Biotechnol. Adv. 2017, 35, 597–618.
- Ribeiro, D.; Freitas, M.; Silva, A.M.S.; Carvalho, F.; Fernandes, E. Antioxidant and pro-oxidant activities of carotenoids and their oxidation products. Food Chem. Toxicol. 2018, 120, 681–699.
- González-Peña, M.A.; Ortega-Regules, A.E.; Anaya de Parrodi, C.; Lozada-Ramírez, J.D. Chemistry, Occurrence, Properties, Applications, and Encapsulation of Carotenoids—A Review. Plants 2023, 12, 313.
- Meyers, K.J.; Mares, J.A.; Igo, R.P., Jr.; Truitt, B.; Liu, Z.; Millen, A.E.; Klein, M.; Johnson, E.J.; Engelman, C.D.; Karki, C.K.; et al. Genetic Evidence for Role of Carotenoids in Age-Related Macular Degeneration in the Carotenoids in Age-Related Eye Disease Study (CAREDS). Investig. Ophthalmol. Vis. Sci. 2014, 55, 587–599.
- Arvanitakis, Z.; Fleischman, D.A.; Arfanakis, K.; Leurgans, S.E.; Barnes, L.L.; Bennett, D.A. Association of white matter hyperintensities and gray matter volume with cognition in older individuals without cognitive impairment. Brain Struct. Funct. 2016, 221, 2135–2146.
- Neuwelt, E.A.; Bauer, B.; Fahlke, C.; Fricker, G.; Iadecola, C.; Janigro, D.; Leybaert, L.; Molnar, Z.; O’Donnell, M.E.; Povlishock, J.T.; et al. Engaging neuroscience to advance translational research in brain barrier biology. Nat. Rev. Neurosci. 2011, 12, 169–182.
- Pardridge, W.M. The blood-brain barrier: Bottleneck in brain drug development. NeuroRx J. Am. Soc. Exp. NeuroTherapeutics 2005, 2, 3–14.
- Guiot, C.; Zullino, S.; Priano, L.; Cavalli, R. The physics of drug-delivery across the blood-brain barrier. Ther. Deliv. 2016, 7, 153–156.
- Kedar, U.; Phutane, P.; Shidhaye, S.; Kadam, V. Advances in polymeric micelles for drug delivery and tumor targeting. Nanomed.-Nanotechnol. Biol. Med. 2010, 6, 714–729.
- Wang, Y.C.; Shim, M.S.; Levinson, N.S.; Sung, H.W.; Xia, Y.N. Stimuli-Responsive Materials for Controlled Release of Theranostic Agents. Adv. Funct. Mater. 2014, 24, 4206–4220.
- Elsabahy, M.; Wooley, K.L. Design of polymeric nanoparticles for biomedical delivery applications. Chem. Soc. Rev. 2012, 41, 2545–2561.
- Rempe, R.; Cramer, S.; Huewel, S.; Galla, H.-J. Transport of Poly(n-butylcyano-acrylate) nanoparticles across the blood-brain barrier in vitro and their influence on barrier integrity. Biochem. Biophys. Res. Commun. 2011, 406, 64–69.
- Kim, H.R.; Andrieux, K.; Gil, S.; Taverna, M.; Chacun, H.; Desmaele, D.; Taran, F.; Georgin, D.; Couvreur, P. Translocation of poly(ethylene glycol-co-hexadecyl)cyanoacrylate nanoparticles into rat brain endothelial cells: Role of apolipoproteins in receptor-mediated endocytosis. Biomacromolecules 2007, 8, 793–799.
- Tian, X.-H.; Lin, X.-N.; Wei, F.; Feng, W.; Huang, Z.-C.; Wang, P.; Ren, L.; Diao, Y. Enhanced brain targeting of temozolomide in polysorbate-80 coated polybutylcyanoacrylate nanoparticles. Int. J. Nanomed. 2011, 6, 445–452.
- Svenson, S. Dendrimers as versatile platform in drug delivery applications. Eur. J. Pharm. Biopharm. 2009, 71, 445–462.
- He, H.; Li, Y.; Jia, X.-R.; Du, J.; Ying, X.; Lu, W.-L.; Lou, J.-N.; Wei, Y. PEGylated Poly(amidoamine) dendrimer-based dual-targeting carrier for treating brain tumors. Biomaterials 2011, 32, 478–487.
- Menjoge, A.R.; Kannan, R.M.; Tomalia, D.A. Dendrimer-based drug and imaging conjugates: Design considerations for nanomedical applications. Drug Discov. Today 2010, 15, 171–185.
- Webb, M.S.; Rebstein, P.; Lamson, W.; Bally, M.B. Liposomal drug delivery: Recent patents and emerging opportunities. Recent Pat. Drug Deliv. Formul. 2007, 1, 185–194.
- Zhang, Q.-Z.; Zha, L.-S.; Zhang, Y.; Jiang, W.-M.; Lu, W.; Shi, Z.-Q.; Jiang, X.-G.; Fu, S.-K. The brain targeting efficiency following nasally applied MPEG-PLA nanoparticles in rats. J. Drug Target. 2006, 14, 281–290.
- Wong, H.L.; Bendayan, R.; Rauth, A.M.; Li, Y.Q.; Wu, X.Y. Chemotherapy with anticancer drugs encapsulated in solid lipid nanoparticles. Adv. Drug Deliv. Rev. 2007, 59, 491–504.
- Goppert, T.M.; Muller, R.H. Polysorbate-stabilized solid lipid nanoparticles as colloidal carriers for intravenous targeting of drugs to the brain: Comparison of plasma protein adsorption patterns. J. Drug Target. 2005, 13, 179–187.
- Kerwin, B.A. Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: Structure and degradation pathways. J. Pharm. Sci. 2008, 97, 2924–2935.
- Muller, R.H.; Radtke, M.; Wissing, S.A. Nanostructured lipid matrices for improved microencapsulation of drugs. Int. J. Pharm. 2002, 242, 121–128.
- Katouzian, I.; Esfanjani, A.F.; Jafari, S.M.; Akhavan, S. Formulation and application of a new generation of lipid nano-carriers for the food bioactive ingredients. Trends Food Sci. Technol. 2017, 68, 14–25.
- Iqbal, M.A.; Md, S.; Sahni, J.K.; Baboota, S.; Dang, S.; Ali, J. Nanostructured lipid carriers system: Recent advances in drug delivery. J. Drug Target. 2012, 20, 813–830.
- Rizwanullah, M.; Ahmad, J.; Amin, S. Nanostructured Lipid Carriers: A Novel Platform for Chemotherapeutics. Curr. Drug Deliv. 2016, 13, 4–26.
- Naseri, N.; Valizadeh, H.; Zakeri-Milani, P. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers: Structure, Preparation and Application. Adv. Pharm. Bull. 2015, 5, 305–313.
- Alam, T.; Pandit, J.; Vohora, D.; Aqil, M.; Ali, A.; Sultana, Y. Optimization of nanostructured lipid carriers of lamotrigine for brain delivery: In vitro characterization and in vivo efficacy in epilepsy. Expert Opin. Drug Deliv. 2015, 12, 181–194.
- Li, F.; Wang, Y.; Liu, Z.; Lin, X.; He, H.; Tang, X. Formulation and characterization of bufadienolides-loaded nanostructured lipid carriers. Drug Dev. Ind. Pharm. 2010, 36, 508–517.
- Weber, S.; Zimmer, A.; Pardeike, J. Solid Lipid Nanoparticles (SLN) and Nanostructured Lipid Carriers (NLC) for pulmonary application: A review of the state of the art. Eur. J. Pharm. Biopharm. 2014, 86, 7–22.
- Burton, G.W.; Ingold, K.U. Beta-Carotene: An unusual type of lipid antioxidant. Science 1984, 224, 569–573.
- Palozza, P.; Serini, S.; Maggiano, N.; Angelini, M.; Boninsegna, A.; Di Nicuolo, F.; Ranelletti, F.O.; Calviello, G. Induction of cell cycle arrest and apoptosis in human colon adenocarcinoma cell lines by beta-carotene through down-regulation of cyclin A and Bcl-2 family proteins. Carcinogenesis 2002, 23, 11–18.
- Moratalla-Lopez, N.; Jose Bagur, M.; Lorenzo, C.; Martinez-Navarro, M.E.; Rosario Salinas, M.; Alonso, G.L. Bioactivity and Bioavailability of the Major Metabolites of Crocus sativus L. Flower. Molecules 2019, 24, 2827.
- Batarseh, Y.S.; Bharate, S.S.; Kumar, V.; Kumar, A.; Vishwakarma, R.A.; Bharate, S.B.; Kaddoumi, A. Crocus sativus Extract Tightens the Blood-Brain Barrier, Reduces Amyloid beta Load and Related Toxicity in 5XFAD Mice. Acs Chem. Neurosci. 2017, 8, 1756–1766.
- Donoso, A.; Gonzalez-Duran, J.; Agurto Munoz, A.; Gonzalez, P.A.; Agurto-Munoz, C. Therapeutic uses of natural astaxanthin: An evidence-based review focused on human clinical trials. Pharmacol. Res. 2021, 166, 105479.
- Routray, W.; Dave, D.; Cheema, S.K.; Ramakrishnan, V.V.; Pohling, J. Biorefinery approach and environment-friendly extraction for sustainable production of astaxanthin from marine wastes. Crit. Rev. Biotechnol. 2019, 39, 469–488.
- Kidd, P. Astaxanthin, Cell Membrane Nutrient with Diverse Clinical Benefits and Anti-Aging Potential. Altern. Med. Rev. 2011, 16, 355–364.
- Liu, X.; Shibata, T.; Hisaka, S.; Osawa, T. Astaxanthin inhibits reactive oxygen species-mediated cellular toxicity in dopaminergic SH-SY5Y cells via mitochondria-targeted protective mechanism. Brain Res. 2009, 1254, 18–27.
- Kong, K.W.; Ismail, A. Lycopene content and lipophilic antioxidant capacity of by-products from Psidium guajava fruits produced during puree production industry. Food Bioprod. Process. 2011, 89, 53–61.
- Perkins-Veazie, P.; Collins, J.K.; Pair, S.D.; Roberts, W. Lycopene content differs among red-fleshed watermelon cultivars. J. Sci. Food Agric. 2001, 81, 983–987.
- Chuacharoen, T.; Sabliov, C.M. Stability and controlled release of lutein loaded in zein nanoparticles with and without lecithin and pluronic F127 surfactants. Colloids Surf. A-Physicochem. Eng. Asp. 2016, 503, 11–18.
- Dembitsky, V.M.; Maoka, T. Allenic and cumulenic lipids. Prog. Lipid Res. 2007, 46, 328–375.
- Heo, S.-J.; Jeon, Y.-J. Protective effect of fucoxanthin isolated from Sargassum siliquastrum on UV-B induced cell damage. J. Photochem. Photobiol. B-Biol. 2009, 95, 101–107.
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a Marine Carotenoid Present in Brown Seaweeds and Diatoms: Metabolism and Bioactivities Relevant to Human Health. Mar. Drugs 2011, 9, 1806–1828.
- Yang, M.; Jin, L.; Wu, Z.; Xie, Y.; Zhang, P.; Wang, Q.; Yan, S.; Chen, B.; Liang, H.; Naman, C.B.; et al. PLGA-PEG Nanoparticles Facilitate In Vivo Anti-Alzheimer’s Effects of Fucoxanthin, a Marine Carotenoid Derived from Edible Brown Algae. J. Agric. Food Chem. 2021, 69, 9764–9777.
- Xiang, S.; Liu, F.; Lin, J.; Chen, H.; Huang, C.; Chen, L.; Zhou, Y.; Ye, L.; Zhang, K.; Jin, J.; et al. Fucoxanthin Inhibits beta-Amyloid Assembly and Attenuates beta-Amyloid Oligomer-Induced Cognitive Impairments. J. Agric. Food Chem. 2017, 65, 4092–4102.
- Oliyaei, N.; Moosavi-Nasab, M.; Tanideh, N.; Iraji, A. Multiple roles of fucoxanthin and astaxanthin against Alzheimer’s disease: Their pharmacological potential and therapeutic insights. Brain Res. Bull. 2022, 193, 11–21.
- Li, N.; Gao, X.; Zheng, L.; Huang, Q.; Zeng, F.; Chen, H.; Farag, M.A.; Zhao, C. Advances in fucoxanthin chemistry and management of neurodegenerative diseases. Phytomedicine 2022, 105, 154352.
- Lee, A.H.; Hong, S.-C.; Park, I.; Yoon, S.; Kim, Y.; Kim, J.; Yang, S.-H. Validation of Fucoxanthin from Microalgae Phaeodactylum tricornutum for the Detection of Amyloid Burden in Transgenic Mouse Models of Alzheimer’s Disease. Appl. Sci. 2021, 11, 5878.
