AThe phenomenon of cridine, an alkaloid from anthracene, also known by the names of 10-azaanthracene, dibenzopyorrosion threatens metallic components, human safety, and the economy. Despite being eco-friendly and promising as a corrosion inhibitor, acridine, 2,3,5,6-dibenzopyridine, and its derivatives or analogs, has not been explored to its full potential. In this review, we have discussed multiple biological activities that acridines have a great potential to be an efficient class of excellentbeen found to show in a bid to prove that they are environmentally benign and much less toxic than many inhibitor. The phenomenon of corrosion threatens metallic components, human safety, and the economy. Despite being eco-friendly and promising as as. Some synthetic routes to acridines and substituted acridines have also been discussed. Thereafter, a multitude of acridines and substituted acridines as corrosion inhibitor, acridine has not been explored to its full potentials of different metals and alloys in various corrosive media have been highlighted.
1. Introduction
The assault of corrosion on metallic structures has become an extremely widespread and persistent dilemma across the globe. In general, corrosion has been defined as the deterioration of metallic structures by reaction with the surrounding environment
[1][2][1,2]. However, in a more technical sense, corrosion entails the movement of mass and charge across a metal/solution interface
[3]. The phenomenon of corrosion ensues in the presence of four things: an anode, a cathode, a metallic path, and an electrolyte. Upon contact with an appropriate electrolyte, the anode part loses free electrons that travel through the electrolyte to reach the cathode. Those free electrons are consumed to produce molecular hydrogen in acidic media and hydroxyl ions in basic/neutral conditions contaminated with oxygen. There are acids (H
2SO
4, HCl, and HNO
3) and bases (NaOH, CaCO
3, NaHCO
3) that commonly attack metallic structures. Besides, there is moisture/water (H
2O), salts (NaCl), gases (formaldehyde, ammonia, sulfur-containing gases, etc.), and aggressive metal polishes as well that have also been found to be aggravating to metallic structures
[4]. Metals have tremendous implications in the construction industry for building pipework, structural components, cladding materials, etc., owing to their outstanding strength and durability. Because of the ubiquitous nature of metallic elements, it is not surprising that they are always in contact with different corrosive scenarios, making corrosion damage prevalent and demanding so much attention
[5].
Corrosion profoundly impacts human health and safety, environment, materials’ life span, and the economy. It has been reported that any country in the world spends around 1–5% of its gross national product (GNP) on corrosion
[6]. The global cost of corrosion is so huge that a study done by the National Association of Corrosion Engineers (NACE) revealed that the global cost of corrosion for the year 2013 was 3.4% (equivalent to USD 2.5 trillion) of the global gross domestic product (GDP)
[7]. A more detailed account of how corrosion impacted the economy and claimed human lives in the past can be found in the previously published literature
[8]. Corrosion inhibition
[9][10][11][9,10,11], anodic protection
[12], cathodic protection
[13][14][13,14], alloying
[15][16][15,16], and coatings
[17][18][17,18] are some of the most widely used approaches to combat corrosion. However, corrosion inhibitors (CIs) have been found to be very popular and effective for protecting metallic units in petroleum refining, oil and gas production, exploration, the chemical industry, water treatment plants, etc. CIs are chemical substances added to corrosive environments to reduce or impede corrosion rates
[5]. CIs form a protective layer on the surface of the metal to keep aggressive electrolytes from coming into contact with the metal surface. As inorganic inhibitors have already been denoted to be environmentally harmful, organic inhibitors have recently gained widespread popularity
[4][19][4,19]. These inhibitors get adsorbed onto the metal surface by either an electrostatic attraction (physical adsorption or physisorption) or a coordinate covalent bond (chemical adsorption or chemisorption) when inhibitor molecules containing lone pairs of electrons donate electrons to the vacant
d-orbitals of the metal. On the other hand, the metal can sometimes donate electrons (retro-donation) to the inhibitor molecules to form that protective layer. As a consequence, organic compounds containing heteroatoms (O, N, P, and S), polar functional groups (−COOH, −OH, −NH
2, −CN, −C=O, −NO
2), conjugated bonds, aromatic rings, π-systems, etc., are known to provide the best corrosion protection.
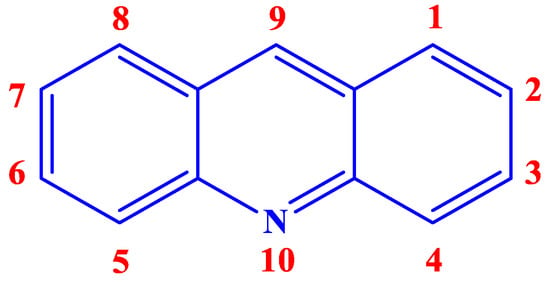
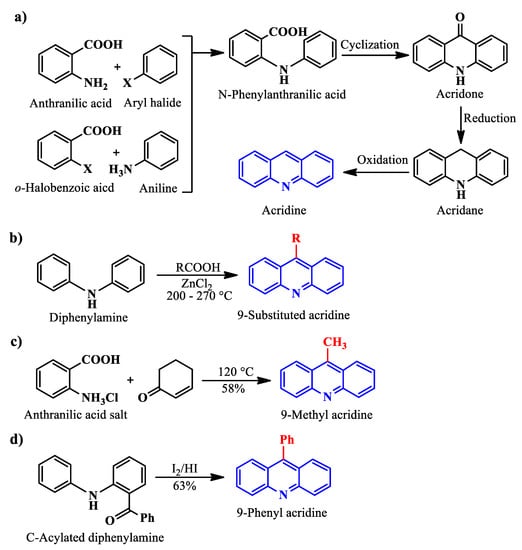
Acridine (Figure 1), an alkaloid from anthracene, also known by the names of 10-azaanthracene, dibenzopyridine, 2,3,5,6-dibenzopyridine, and its derivatives or analogs, have a great potential to be an efficient class of excellent inhibitor. Aromatic rings, π-systems, and a nitrogen atom with an available lone pair of electrons already present in the chemical structure of acridine hint at the possibility of it acting as a suitable inhibitor. Additionally, incorporating different functional groups that can undergo chemisorption or physisorption with the vacant d-orbitals of the metal in the acridine can make it even better as a CI. Furthermore, acridine and its analogs have been reported to have shown loads of biological activities, rendering it environmentally safe as well.
Figure 1.
Chemical structure and numeration of acridine.
2. Biological Properties of Acridines
Inorganic salts and salts of heavy metals had been used historically as CIs, including molybdates, nitrates, phosphates, polyphosphates, phosphonates, sodium chromates, silicates, hydroxides, etc.
[20]. However, even after showing practical efficiencies, these types of inhibitors have been restricted in their use, having been identified as toxic to human life and the environment. As a consequence, several tough regulations, including the US Occupational Safety and Health Administration (OSHA) in 1993, the Emergency Planning and Community Right to Know Act of 1986, the Chemical Hazard Assessment and Risk Management (CHARM) model adopted in the UK and other European countries, caused researchers to look for non-toxic yet efficient corrosion inhibitors. Acridine and its derivatives are found in natural plants and marine organisms, forming an important class of
N-containing heterocycles. Due to their unique chemical and physical properties and biological activities, acridine derivatives are used across different industries. Pharmaceutically, acridine derivatives have been found to show bio-activities, such as anticancer, antitubercular, antiviral, antimalarial, antimicrobial, anti-inflammatory, antiparasitic, and fungicidal activities
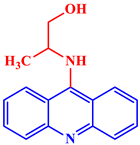
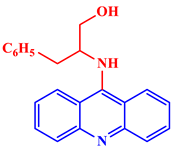
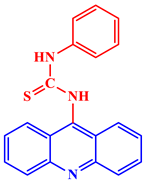
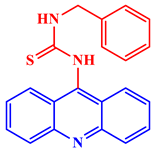
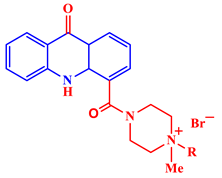
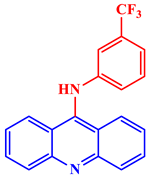
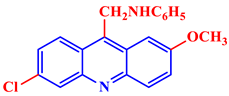
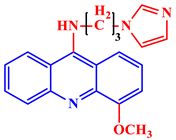
[21]. Some acridine derivatives, their structures, and relevant bio-activity have been included in
Table 1 to show that acridine and its derivatives are being explored as environmentally friendly and efficient CIs.