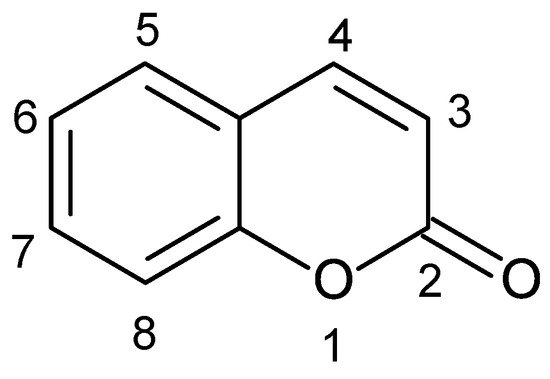
Coumarins are a structurally varied set of 2H-chromen-2-one compounds categorized also as members of the benzopyrone group of secondary metabolites. Coumarin derivatives attract interest owing to their wide practical application and the unique reactivity of fused benzene and pyrone ring systems in molecular structure. Coumarins have their own specific fingerprints as antiviral, antimicrobial, antioxidant, anti-inflammatory, antiadipogenic, cytotoxic, apoptosis, antitumor, antitubercular, and cytotoxicity agents. Natural products have played an essential role in filling the pharmaceutical pipeline for thousands of years. Biological effects of natural coumarins have laid the basis of low-toxic and highly effective drugs.
- natural compounds
- coumarins
- secondary metabolites
- biosynthesis
- fungi
1. Introduction
1.1. Overview on Fungal Coumarins
1.2. Novelty Statement
2. Coumarins Hold a Special Place among Natural Products
References
- Suryanarayanan, T.S.; Thirunavukkarasu, N.; Govindarajulu, M.B.; Sasse, F.; Jansen, R.; Murali, T.S. Fungal endophytes and bioprospecting. Fungal Biol. Rev. 2009, 23, 9–19.
- Kijpornyongpan, T.; Schwartz, A.; Yaguchi, A.; Salvachúa, D. Systems biology-guided understanding of white-rot fungi for biotechnological applications: A review. iScience 2022, 25, 104640.
- Razavi, S.M. Plant counnarins as allelopathic agents. Int. J. Biol. Chem. 2011, 5, 86–90.
- Reddy, D.S.; Kongot, M.; Kumar, A. Coumarin hybrid derivatives as promising leads to treat tuberculosis: Recent developments and critical aspects of structural design to exhibit anti-tubercular activity. Tuberculosis 2021, 127, 102050.
- Iranshahi, M.; Askari, M.; Sahebkar, A.; Hadjipavlou-Litina, D. Evaluation of antioxidant, anti-inflammatory and lipoxygenase inhibitory activities of the prenylated coumarin umbelliprenin. DARU J. Pharm. Sci. 2009, 17, 99–103.
- Ismael, R.N.; Mustafa, Y.F.; Al-Qazaz, H.K. Coumarin-based products: Their biodiversity and pharmacology. Iraqi J. Pharm. 2022, 18, 162–179.
- Braga, R.M.; Padilla, G.; Araújo, W.L. The biotechnological potential of Epicoccum spp.: Diversity of secondary metabolites. Crit. Rev. Microbiol. 2018, 44, 759–778.
- Hussain, M.I.; Syed, Q.A.; Khattak, M.N.K.; Hafez, B.; Reigosa, M.J.; El-Keblawy, A. Natural product coumarins: Biological and pharmacological perspectives. Biologia 2019, 74, 863–888.
- Chandrashekar, S.; Govindappa, M.; Shashank, M.P.; Ramith, R.; Bhargav, S.; Chandan, D.; Shiva, P.K.; Asad, S.; Chandan, S. Plants and endophytes—A partnership for the coumarin production through the microbial systems. Mycology 2022, 13, 2–15.
- Cheke, R.S.; Patel, H.M.; Patil, V.M.; Ansari, I.A.; Ambhore, J.P.; Shinde, S.D.; Kadri, A.; Snoussi, M.; Adnan, M.; Kharkar, P.S.; et al. Molecular insights into coumarin analogues as antimicrobial agents: Recent developments in drug discovery. Antibiotics 2022, 11, 566.
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A Natural, Privileged and Versatile Scaffold for Bioactive Compounds. Molecules 2018, 23, 250.
- Srikrishna, D.; Godugu, C.; Dubey, P.K. A review on pharmacological properties of coumarins. Mini Rev. Med. Chem. 2018, 18, 113–141.
- Zhu, J.J.; Jiang, J.G. Pharmacological and nutritional effects of natural coumarins and their structure–activity relationships. Mol. Nutr. Food Res. 2018, 62, 1701073.
- Hu, Y.Q.; Xu, Z.; Zhang, S.; Wu, X.; Ding, J.W.; Lv, Z.S.; Feng, L.S. Recent developments of coumarin-containing derivatives and their anti-tubercular activity. Eur. J. Med. Chem. 2017, 136, 122–130.
- Fan, Y.L.; Wu, J.B.; Cheng, X.W.; Zhang, F.Z.; Feng, L.S. Fluoroquinolone derivatives and their anti-tubercular activities. Eur. J. Med. Chem. 2018, 146, 554–563.
- Mishra, S.; Pandey, A.; Manvati, S. Coumarin: An emerging antiviral agent. Heliyon 2020, 6, e03217.
- Umashankar, T.; Govindappa, M.; Ramachandra, Y.L.; Padmalatha Rai, S.; Channabasava, R. Isolation and characterization of coumarin isolated from endophyte, Alternaria species-1 of Crotalaria pallida and its apoptotic action on HeLa cancer cell line. Metabolomics 2015, 5, 1000158.
- Kuriakose, G.C.; Singh, S.; Rajvanshi, P.K.; Surin, W.R.; Jayabaskaran, C. In vitro cytotoxicity and apoptosis induction in human cancer cells by culture extract of an endophytic Fusarium solani strain isolated from Datura metel L. Pharm. Anal. Acta 2014, 5, 1000293.
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. BioMed Res. Int. 2013, 2013, 963248.
- Chebrolu, K.K.; Jayaprakasha, G.K.; Jifon, J.; Patil, B.S. Purification of coumarins, including meranzin and pranferin, from grapefruit by solvent partitioning and a hyphenated chromatography. Sep. Purif. Technol. 2013, 116, 137–144.
- Singh, A.; Singh, D.K.; Kharwar, R.N.; White, J.F.; Gond, S.K. Fungal endophytes as efficient sources of plant-derived bioactive compounds and their prospective applications in natural product drug discovery: Insights, avenues, and challenges. Microorganisms 2021, 9, 197.
- Patel, G.; Banerjee, S. Review on synthesis of bio-active coumarin-fused heterocyclic molecules. Curr. Org. Chem. 2020, 24, 2566–2587.
- Nitiema, L.W.; Savadogo, A.; Simpore, J.; Dianou, D.; Traore, A.S. In vitro antimicrobial activity of some phenolic compounds (coumarin and quercetin) against gastroenteritis bacterial strains. Int. J. Microbiol. Res. 2012, 3, 183–187.
- Witaicenis, A.; Seito, L.N.; Di Stasi, L.C. Intestinal anti-inflammatory activity of esculetin and 4-methylesculetin in the trinitrobenzenesulphonic acid model of rat colitis. Chem. Biol. Interact. 2010, 186, 211–218.
- Witaicenis, A.; Seito, L.N.; da Silveira Chagas, A.; de Almeida Junior, L.D.; Luchini, A.C.; Rodrigues-Orsi, P.; Cestari, S.H.; Di Stasi, L.C. Antioxidant and intestinal anti-infammatory effects of plant-derived coumarin derivatives. Phytomedicine 2014, 21, 240–246.
- Kwon, O.S.; Choi, J.S.; Islam, M.N.; Kim, Y.S.; Kim, H.P. Inhibition of 5-lipoxygenase and skin inflammation by the aerial parts of Artemisia capillaris and its constituents. Arch. Pharmacal Res. 2011, 34, 1561–1569.
- Shin, E.; Choi, K.M.; Yoo, H.S.; Lee, C.K.; Hwang, B.Y.; Lee, M.K. Inhibitory effects of coumarins from the stem barks of Fraxinus rhynchophylla on adipocyte differentiation in 3T3-L1 cells. Biol. Pharm. Bull. 2010, 33, 1610–1614.
- Mahmoodi, M.; Aliabadi, A.; Emami, S.; Safavi, M.; Rajabalian, S.; Mohagheghi, M.A.; Khoshzaban, A.; Samzadeh-Kermani, A.; Lamei, N.; Shafiee, A.; et al. Synthesis and in-vitro cytotoxicity of poly-functionalized 4-(2-arylthiazol-4-yl)-4H-chromenes. Arch. Pharm. 2010, 343, 411–416.
- Luo, K.W.; Sun, J.G.; Chan, J.Y.W.; Yang, L.; Wu, S.H.; Fung, K.P.; Liu, F.Y. Anticancer effects of imperatorin isolated from Angelica dahurica: Induction of apoptosis in HepG2 cells through both death-receptor-and mitochondria-mediated pathways. Chemotherapy 2011, 57, 449–459.
- Yun, E.S.; Park, S.S.; Shin, H.C.; Choi, Y.H.; Kim, W.J.; Moon, S.K. p38 MAPK activation is required for esculetin-induced inhibition of vascular smooth muscle cells proliferation. Toxicol. In Vitro 2011, 25, 1335–1342.
- Chiang, C.C.; Cheng, M.J.; Peng, C.F.; Huang, H.Y.; Chen, I.S. A novel dimeric coumarin analog and antimycobacterial constituents from Fatoua pilosa. Chem. Biodivers. 2010, 7, 1728–1736.
- Gonçalves, G.A.; Spillere, A.R.; das Neves, G.M.; Kagami, L.P.; von Poser, G.L.; Canto, R.F.S.; Eifler-Lima, V.L. Natural and synthetic coumarins as antileishmanial agents: A review. Eur. J. Med. Chem. 2020, 203, 112514.
- Hassan, M.Z.; Osman, H.; Ali, M.A.; Ahsan, M.J. Therapeutic potential of coumarins as antiviral agents. Eur. J. Med. Chem. 2016, 123, 236–255.
- Huang, L.; Zhe-Ling, F.E.N.G.; Yi-Tao, W.A.N.G.; Li-Gen, L.I.N. Anticancer carbazole alkaloids and coumarins from Clausena plants: A review. Chin. J. Nat. Med. 2017, 15, 881–888.
- Umashankar, T.; Govindappa, M.; Ramachandra, Y.L.; Chandrappa, C.P.; Padmalatha Rai, S.; Channabasava, R. Isolation, purification and in vitro cytotoxicity activities of coumarin isolated from endophytic fungi, Alternaria species of Crotalaria pallida. Indo Am. J. Pharm. Res. 2015, 5, 926–936.
