1. General Presentation of Lateral Flow ImmunoIAssay
1.1. Components and Principle
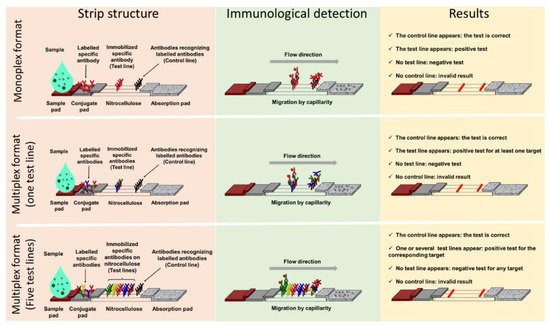
Lateral Flow ImmunoAssay (LFIA) IA tests generally consist of a strip supporting different porous compounds on which liquids migrate by capillarity. The sample pad (SP), which is usually made of cellulose, receives the sample that successively drains onto the conjugate pad (CP). Made of glass fiber, the CP is the storage area for the conjugate, a labelled molecule that generates the signal on the strip. This signal is located on a nitrocellulose membrane printed with different compounds in order to form one test line and a control line (Figure 1). The test line captures the targets of interest while the control line serves as an inner control for confirmation of correct flow and compatible test conditions. Finally, the wicking pad acts as a pump as well as a reservoir for the liquid dispensed on the SP, and its capacity influences the volume of sample that can be analyzed. All components overlap one over the other and are generally enclosed in a plastic cassette. This cassette provides pressure points to maintain close contacts, providing efficient flow of the reagents through the strip, protection, easy handling, a localized sample dispensing area, and a reading frame.
Figure 1. Lateral flow assay formats: components and principle. Presentation of three lateral flow immunoassay formats with, from left to right, their structure (pink panel), immunological detection principle (green panel), and results interpretation (yellow panel). The monoplex format able to detect only one target is presented in the upper panel, a multiplex detection format with one test line and no possible identification of the target is in the central panel, and a multiplex detection format with spatial separation of the test lines and identification of the target(s) detected is presented in the lower panel.
Migration begins once the sample has been loaded on the SP that, depending on the nature of the samples, can be pre-treated in order to reduce matrix effects. The sample solution resuspends the conjugate that forms complexes with the analyte if present. Capillary pressure transports the complexes along the nitrocellulose membrane, and they accumulate on the test line and the excess of conjugate on the control line.
Most of the time, the test line and the conjugate involve antibodies specific for the target being detected, and two formats of LFIA are available. The first, known as non-competitive or sandwich immunoassay format, is for large molecular weight analytes such as proteins that provide several antigenic sites. In this case, a colored test line represents a positive result. The second format, known as competitive or inhibition immunoassay format, is dedicated to small molecular weight antigens, and an attenuated or absent test line indicates a positive result.
1.2. Advantages of Lateral Flow ImmunoAssay
1.2. Advantages of LFIA
LFIA are one-step assays that require no washing and only a small sample volume. The time to results, following an easy sample handling, is short (15–30 min), which positions LFIA as a good candidate for primary screening at PoC or PoN. They are inexpensive and do not have specific storage constraints, such as refrigeration, making them accessible to third-world countries
[1][2][22,23]. The qualitative interpretation of the result can be visual, and no particular skills are required. Moreover, depending on the parameters of the test, results can also be semi-quantitative or even quantitative through the use of a reader
[3][4][24,25]. In addition, if the reagents are available, the development time to market of an LFIA is relatively short, which can be useful to respond to an urgent sanitary crisis, as illustrated with the recent COVID-19 pandemic. The LFIA, due to its configuration, allows the detection of multiple analytes such as proteins, haptens, or nucleic acids
[5][20].
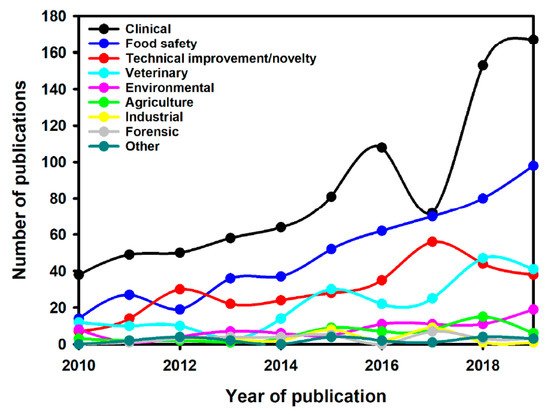
Interest in LFIAs is best illustrated by the increase in publications describing their use in different fields of application over the last 10 years, especially in the clinical domain (
Figure 2).The search query used was (Scopus format): TITLE-ABS-KEY (lateral AND flow AND immunoassay) OR TITLE (lateral AND flow AND assay) OR TITLE (immunochromatographic) AND PUBYEAR > 2009 AND PUBYEAR < 2020 AND (LIMIT-TO (DOCTYPE, “ar”)) AND (LIMIT-TO (LANGUAGE, “English”)). The research has been done in October 2020. Then, the articles were selected following the PRISMA guideline for systematic reviews
[1][22]. It is likely that the ease of use, speed, and specificity of these tests are at the origin of their exponential use, including in the field of AMR.
Figure 2. Number of publications per year for different LFIA applications [1]. Number of publications per year for different LFIA applications [22].
2. Classical Lateral Flow Assays and Antimicrobial MResistant
Bacteria possess four main mechanisms that can confer resistance to antibiotics: (i) expression or overexpression of efflux pumps reducing antibiotic concentration within the bacteria; (ii) decreased permeability of the membrane or cell wall leading to ineffective drug concentration in the bacteria; (iii) changed target structure with affinity loss for antibiotics; and (iv) antibiotic degradation mediated by hydrolysis
[6][26]. Any protein involved in a mechanism of AMR can then become a privileged target for LFIA. As a result, a number of LFIA tests have been developed, and some are commercially available.
2.1. Monoplex Lateral Flow ImmunoAssay to Address Antimicrobial Resistant (AMR) Detection
2.1. Monoplex LFIA to Address AMR Detection
As the number of antibiotic-resistant isolates has increased, LFIA tests have been developed to target enzyme-mediated resistance traits in the clinically most important bacteria, with the primary objective of detecting enzymes involved in the resistance of most prevalent pathogens. A LFIA test detecting the expression of the
Pseudomonas aeruginosa 6′-N-acetyltransferase AAC(6′)-Iae, which confers resistance to aminoglycosides, was described in 2010 and has a sensitivity of 10
5 cfu/test
[7][27]. Another LFIA targets ArmA 16S rRNA methylase, which is one of the most prevalent 16S rRNA methylase reported and leads to pan-aminoglycoside resistance in Gram-negative bacteria (GNB) such as
Acinetobacter baumannii and
Escherichia coli [8][28].
Methicillin-resistant
Staphylococcus aureus (MRSA) is another major pathogen responsible for severe morbidity and mortality in many hospitals worldwide, which also has an efficient capacity for spreading in the community
[9][29]. The early detection of methicillin resistance, which confers resistance to all ß-lactams, is essential. It relies on the detection of the penicillin-binding-protein 2a (PBP2a), which has a reduced affinity for beta-lactam antibiotics. Antibody-based techniques for the detection of MRSA are challenging as
S. aureus protein A binds to mammalian immunoglobulins. Nevertheless, the use of IgY anti-PBP2a antibodies has been described by Yamada et al.
[10][30], and another study described an optimized LFIA with a detection limit of 10
4 cfu/mL
[11][31].
Vancomycin-resistant enterococcus (VRE) is one of the most important nosocomial pathogens worldwide
[12][13][14][32,33,34]. The vancomycin resistance mechanism in
Enterococcus faecium and
Enterococcus faecalis is mostly acquired and linked to the production of ligases. In Europe, the most prevalent are VanA and VanB
[12][15][32,35]. In this context, a LFIA for the identification of VanA-VRE isolates was described with 100% sensitivity and 100% specificity and a limit of detection of 6.3 × 10
6 cfu and 4.9 × 10
5 cfu per test when the growth was performed on MH or ChromID
® VRE plates, respectively
[16][36]. Another LFIA for the detection of VanB-VRE isolates with a lower sensitivity and a mandatory pre-culture on vancomycin-containing media for induction of VanB ligase has also been reported
[17][37].
Beta-lactams represent the major family of antibiotics to treat infections due to Gram-negative bacteria, but their use is currently challenged by the spread of beta-lactamases
[18][38]. In particular, the spread of extended-spectrum beta-lactamases (ESBLs) among Enterobacterales represents a major threat as these enzymes are able to inactivate most beta-lactam molecules (including 3rd and 4th generation cephalosporins and aztreonam), sparing only carbapenems
[19][39]. The most common family of the ESBL, the CTX-M, represented by five sub-groups: CTX-M-1, CTX-M-2, CTX-M-8, CTX-M-9, and CTX-M-25, has disseminated worldwide
[20][40]. The increase in the prevalence of ESBL-producing Enterobacterales has led to an increased use of carbapenems, a last resort antibiotic, to treat infections with ESBL-producers. This has led to the selection and subsequent increase in bacteria resistant to these antibiotics. Carbapenem-resistant Enterobacterales (CREs) are usually resistant to most, if not all antibiotics, thus posing serious therapeutic issues in clinical practice. Among CREs, carbapenemase-producing isolates are the most worrisome, as they are capable of efficiently hydrolyzing carbapenems, and their genes are carried by plasmids that may be exchanged between bacteria. Early identification of carriers is essential in order to implement reinforced infection control measures, among which isolation of the patient is a prerequisite. There are five major carbapenemases, KPC, NDM, OXA-48, VIM, and IMP. LFIAs to detect KPC and OXA-48-like enzymes
[21][41] (Coris Bioconcept, Gembloux Belgium), IMP (the most prevalent metallo-β-lactamases in Japan
[22][23][42,43]), and NDM
[24][25][44,45] have been developed, all with 100% sensitivity and specificity when used with isolated colony from agar plates.
Faced with infections resistant to carbapenems, the paucity of therapeutic options has led to the use of polymyxins such as colistin as last resort antibiotics
[26][46]. Inexorably, bacteria acquired colistin resistance. While most colistin resistance is due to chromosomic mutations, plasmid-encoded mechanisms, such as MCR-1, have also been described. The latter are considered particularly threatening, as the mcr genes are plasmid encoded, and the resistance phenotype is difficult to detect. MCR-1, initially described in 2015, mediates the modification of the lipopolysaccharide by a phosphoethanolamine transferase activity
[27][47]. More than eight different MCR-alleles have now been described
[28][48]. Soon after the first description of MCR-1, a LFIA directed against this allele was commercialized (NG-Test MCR-1). This assay has been evaluated in a multicentric study against a collection of human and animal enterobacterial isolates. The results revealed 100% sensitivity for MCR-1 expressing isolates, but some MCR-2 carriers were missed, and the assay did not detect MCR-3, MCR-4, and MCR-5 carriers
[29][49].
2.2. Multiplex Lateral Flow ImmunoAssay in the Antimicrobial Resistant Field
2.2. Multiplex LFIA in the AMR Field
Monoplex LFIAs have proven efficacy for the detection and identification of resistance-determining markers. When targeting mechanisms involving a multitude of variants, such as the CTX-M family, a wider specificity can be relevant. Multiplex LFIAs, able to detect CTX-M enzymes but without discriminating the variant or subgroup to which they belong, have been developed. For example, the NG-Test CTX-M MULTI (NG Biotech, Guipry, France) is commercially available and relies on a cocktail of anti-CTX-M mouse antibodies, immobilized on a unique test line (
Figure 1, central panel). This test allows the detection of the five CTX-M-subgroups. A recent study showed that the NG-Test CTX-M MULTI could detect 98% of ESBL-producers from a French clinical setting, either from colonies or positive blood cultures, missing only two SHV-ESBL producers
[30][50]. A study, conducted in Italy, reported that the NG-Test CTX-M MULTI was a reliable assay for the detection of CTX-M-like ESBLs from bacterial pellets from blood culture broth, showing excellent sensitivity and specificity
[31][51]. A further study recently described the detection of CTX-M-group-1, -2, and -9 producers using a monoclonal rabbit anti-CTX-M antibody, and showed 100% sensitivity and specificity with clinical isolates grown on agar plates
[32][52]. Most resistance genes are carried by mobile genetic elements
[33][53] and a single strain can harbor more than one resistance determinant. In this scenario, the detection and identification of more than one mechanism of resistance in a single test is relevant. To do so, several test lines are printed on the same strip (
Figure 1, lower panel) and target identification is made through a spatial repartition
[34][54]. Several multiplex assays have been described, so far only for the identification of carbapenemase-producing strains. Several versions of the RESIST LFIA test exist (from Coris Bioconcept), which are differentiated by the number of carbapenemases that can be detected. RESIST-3
[35][55] can detect NDM, KPC, and OXA-48 enzymes. This test has since been upgraded with the additional detection of VIM (RESIST-4
[36][56]), and IMP or OXA-163 (RESIST-5
[37][38][39][57,58,59]). All these multiplex assays consist of two-independent cassettes that are used in parallel with the same bacterial extract. Another assay, named NG CARBA-5
[40][60] (from NG Biotech, Guipry, France), also targets NDM, IMP, VIM, OXA-48, and KPC carpabenemases, but a major difference compared to the RESIST-LFIA test is that the sample has to be loaded onto one unique cassette. The NG Carba-5 has been evaluated by the Antimicrobial Resistance and Healthcare Associated Infections (AMRHAI) in London, with isolates covering the diversity of the carbapenemases. It showed 97.31% sensitivity and 99.75% specificity, missing IMP-13- and IMP-14-like enzymes
[41][61], also not identified during a previous evaluation by the same team
[42][62]. Missing such enzymes could be an issue in countries with a high IMP prevalence, and since the AMRHAI study, a new version has become available with improved detection of IMP variants
[43][63]. In Europe, NG CARBA-5 shows high sensitivity (97.3% to 100%) and specificity (96.1% to 100%) according to recent studies
[41][44][45][46][47][61,64,65,66,67]. NG CARBA-5 has also received U.S. Food and Drug Administration clearance and evaluation at three medical centers in the USA confirmed its accuracy for detecting and identifying the five most common carbapenemases
[48][68]. NG Carba-5 is often compared to the molecular Xpert Carba-R test (Cepheid) and shows a very high correlation with Carba-R
[48][49][50][51][68,69,70,71], but with the advantages of time efficiency and lower cost. LFIAs targeting carbapenemases using the CIM method and its various versions (mCIM, zCIM, and eCIM) make detection of most common and rare variants rapid, simple, and inexpensive
[52][53][72,73]. Most studies have described the use of NG Carba-5 with Enterobacterales isolates, but in France, it is also valuable for the detection of carbapenemases produced by
Pseudomonas spp., 89.4% detected compared with only 12.9% of carbapenemase-producing
Acinetobacter spp. Indeed, the most prevalent carbapenemases in this organism are not targeted by this test
[44][64].
2.3. Limitations of LFIA in the Context of AMR
Despite the many possibilities that LFIA offer, there are some limitations. For example, variations in sample volume loaded on the device can both reduce the accuracy of the result and impact the sensitivity of the test. Moreover, LFIA performance relies mostly on antibody affinity and specificity. However, even if the latter is determinant,
reswe
archers can only detect what
researcherswe are looking for; thus, the specificity is a limitation in respect of the diversity of enzymes involved in bacterial resistance mechanisms. Implementation of an LFIA test in the clinical setting has to take into account the local epidemiological context in terms of prevalence of resistance mechanisms. The user should keep in mind that any new enzyme variant harboring a mutation in the epitope recognized by any of the antibodies involved in the test may give a reduced signal or a false negative result. Moreover, isolates with an AMR profile but which harbor a mechanism not targeted by the assay will also give a negative result. As for all diagnostic tests, interpretation of results must be made in light of the clinical data and viewed critically. Most LFIAs have been evaluated on colonies grown on agar-containing plates. The ability to use directly from clinical samples would therefore be an important improvement as it would increase turn-around time. For this to be achieved, the sensitivity of the assays has to be improved (on average 10
5–10
6 cfu), and interferences with the different biological matrixes needs to be evaluated. The sample or the matrix analyzed may require an additional sample pre-treatment to avoid interferences. Indeed, sample viscosity may prevent efficient migration on the nitrocellulose membrane leading to invalid results, or the matrix may generate interferences leading to false positive or negative results. Current LFIA systems must therefore be improved before they can be used directly with clinical samples
[5][54][20,74]. Finally, LFIAs only detect the enzymes for which they have been developed. This is the case for the NG-Test CTX-M MULTI that detects the main ESBLs and CTX-Ms, but misses minor ESBLs, and plasmid-encoded cephalosporinase. Combining it with an LFIA that detects hydrolytic activity instead of the enzyme itself would therefore be a major improvement.