Chronic obstructive pulmonary disease (COPD), asthma and cystic fibrosis (CF) are distinct respiratory diseases that share features such as the obstruction of small airways and disease flare-ups that are called exacerbations and are often caused by infections. Along the airway epithelium, immunoglobulin (Ig) A contributes to first line mucosal protection against inhaled particles and pathogens. Dimeric IgA produced by mucosal plasma cells is transported towards the apical pole of airway epithelial cells by the polymeric Ig receptor (pIgR), where it is released as secretory IgA. Secretory IgA mediates immune exclusion and promotes the clearance of pathogens from the airway surface by inhibiting their adherence to the epithelium.
- immunoglobulin A
- mucosal immunity
- COPD
1. Introduction
2. The Mucosal S-IgA System in Homeostasis
The mucosal barrier system is constitutively made of several components. The epithelial layer and the intercellular junctions, with the mucus layer on top, and the lamina propria underneath [3][4][13,14], form a physical barrier against exogenous antigens and pathogens. Mucosal antimicrobial molecules, cellular innate immunity and mucosal adaptative immunity are complementary lines of defence against aggressions [5][15]. In the bronchi, the epithelium is pseudostratified, and composed by numerous cell types whose proportions are tightly controlled [6][7][16,17]. Most of the bronchial airway epithelium consists of ciliated and goblet cells that together constitute the mucociliary elevator clearing particulates and other irritants out of the airspaces. As well as these cell types, the airway epithelium also comprises basal, club and neuroendocrine cells, along with rare ionocytes [8][18]. Ciliated cells are prominent and represent more than 50% of all airway epithelial cells. They possess around 300 cilia whose synchronized beating pushes the mucus layer towards the trachea and the larynx [9][19]. Goblet cells, accounting for 5 to 15% of airway epithelial cells in health, produce mucus and are virtually not present in small airways [9][19]. Basal cells are multipotent stem cells that both anchor the epithelium to the underlying lamina propria and drive epithelial homeostasis and orderly regeneration after injury [10][11][20,21]. They represent 5 to 30% of epithelial cells, their proportion decreasing from the trachea down to the respiratory bronchioles [12][22]. Club cells are dome-shaped cells involved in host defence, and represent 20% of epithelial cells in small airways, where they also behave as progenitor cells [13][14][23,24]. Neuroendocrine cells are rare, innervated cells (< 1% of airway epithelial cells) [15][25] that are thought to be involved in oxygen sensing, smooth muscle tonus and immune responses [16][17][26,27]. Finally, recently discovered ionocytes seem to control the airway surface liquid and mucus viscosity [18][28]. Among these cell types, ciliated, goblet and club cells have been robustly shown to express the polymeric immunoglobulin receptor (pIgR) and therefore participate in the epithelial transcytosis of dimeric IgA (d-IgA) towards the apical mucus layer (see below) [19][20][29,30], while recent single cell transcriptomic data and the emergence of the Human Lung Atlas suggest that ionocytes (but not basal and neuroendocrine cells) also express the pIgR [21][31].2.1. Production and Structure of S-IgA and pIgR
IgA constitutes the most prevalent Ig isotype at mucosal sites and the second most prevalent in serum after IgG. Therefore, IgA is the most abundantly produced Ig in the human body, with an average production rate of 66 mg/kg/day [22][23][32,33]. IgA largely mediates the adaptive humoral immune defence at mucosal surfaces [24][25][34,35], while its role in serum remains relatively unexplored. IgA is produced by B cells both in the systemic and mucosal immune systems, the latter being referred to as mucosa-associated lymphoid tissue (MALT) which includes mucus layers, epithelial cells, lymphoid tissues and immune molecules of the mucosal lamina propria [26][36]. Structurally, IgA is found as monomers (m-IgA) or polymers, mainly consisting of dimers (d-IgA), although some larger forms also exist. M-IgA is composed of two light chains κ and λ, common to all types of Ig’s, covalently linked to two specific heavy chains α [27][37]. In serum, IgA represents 6 to 15% of total immunoglobulins [28][38], and predominates as monomers (85% to 90%). Serum IgA mainly originates from the bone marrow, although the spleen and peripheral lymph nodes could contribute to a lower extent [28][38]. In humans, two IgA subclasses exist, namely IgA1 and IgA2. Each of these IgA subtypes can be found in a monomeric, dimeric, or secretory form [29][40]. In bronchial secretions, IgA2 accounts for 30% of total IgA, compared to 10% in plasma [29][40]. B cells that secrete IgA in the airways are antibody-secreting plasma cells that are generated following cognate interactions between T cells and dendritic cells that have taken up and processed a specific antigen [30][31][42,43].2.2. Transcytosis of d-IgA and Functions of S-IgA
pIgR-mediated polymeric Ig transcytosis requires four well-orchestrated steps [32][51]. First, d-IgA (or pentameric IgM) binds to pIgR through a non-covalent interaction between Ig’s J chain and the extracellular part of pIgR. Second, pIgR is internalized in the clathrin-mediated endocytosis process and delivered in basolateral early endosomes, then in common endosomes. Endocytosed pIgR is then either recycled back to the basolateral membrane, or delivered to the apical surface [33][50]. Third, the extracellular part of the receptor (SC, bound to the transported Ig), undergoes endoproteolytic cleavage by a host serine proteinase. Lastly, S-IgA consisting of d-IgA and SC, crosses the mucus layer by diffusion. Of note is that, unbound pIgR may also undergo epithelial transcytosis and proteolytic cleavage, thereby releasing free SC that may be found in mucosal secretions [32][33][50,51]. At mucosal surfaces, S-IgA functions include neutralization, as well as other biological effects through interactions with, and regulation of, immune cells (via specific Fcα receptors) and microbiota components (Figure 1). The first-line mucosal defence is primarily exerted by S-IgA through its binding of soluble or particulate antigens, preventing their adherence to the epithelium. This non-specific immunity process called ‘immune exclusion’ consists of several sub-steps, including agglutination and entrapment as well as clearance at mucosal surfaces [2][34][2,53].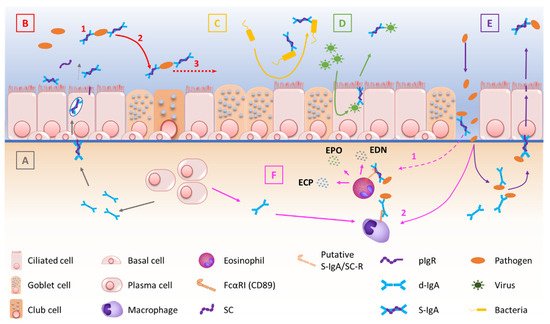
2.3. Regulation of S-IgA Production
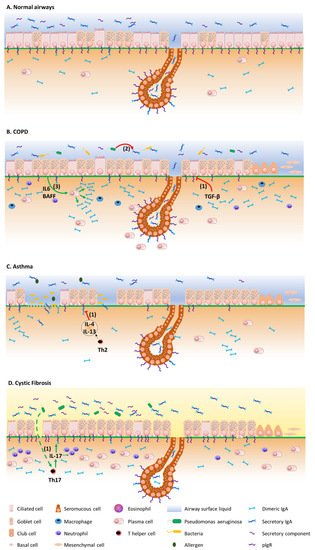
3. The Mucosal S-IgA System in Airway Disease
Figure 2 summarizes the mechanisms by which the IgA/pIgR system is altered in chronic respiratory diseases. Although resulting from complex physiopathological mechanisms in these diseases, respiratory tract colonization by pathogens and trespassing of the mucosal barrier have been shown to trigger these diseases, demonstrating their contribution to the development of such diseases [43][44][75,76].