Chlorine first reacts with organic and inorganic matters before pathogens inactivation. The amount of chlorine consumed in this process is termed chlorine demand. The combined chlorine that forms together with any free available chlorine in the water is called the chlorine residual. This is the component of the added chlorine that disinfects the water. Free available chlorine is formed by differences in the concentrations of hypochlorous and hypochlorite ions, a process that depends on the pH of the water. Even though the chlorination procedure is well-researched, establishing an appropriate chlorine dose remains a difficult task for many field applications. Nevertheless, the effective chlorine dose should be sufficient to destroy pathogens and oxidize the organic contaminants as well as maintain sufficient free available chlorine in the water distribution system, post-chlorination.
1. Breakpoint Chlorination and Factors Influencing Disinfection
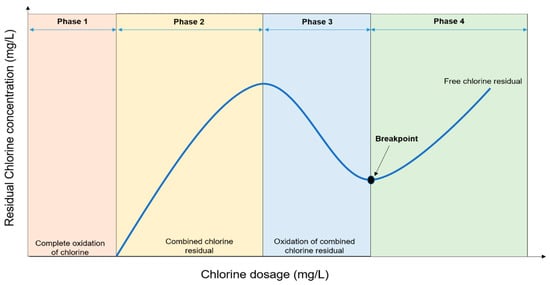
Breakpoint chlorination is the continuous addition of chlorine until the chlorine demand is satisfied, combined chlorine compounds are oxidized and only free chlorine remains. The four phases involved in breakpoint chlorination is depicted in Figure 12.
Figure 12.
Graphical representation of breakpoint chlorination.
Complete oxidation of chlorine occurs in the first phase due to low chlorine concentration and lack of free chlorine residual. The phase is also known as the initial chlorine demand. Combined chlorine residual is formed between Cl
− and NH
4+ in the second phase. The concentration of the added chlorine in this phase is proportional to the total concentration of chlorine residual. Continuous addition of chlorine in the third phase causes oxidation of the combined residues causing a reduced concentration of the residual chlorine up to “dip” or “breakpoint”. Beyond this point, the concentration of the residual chlorine increases
[1][22].
Temperature, pH, contact time, density and the inherent nature of microorganisms present determine the efficacy of the chlorination. Keeping all other factors constant, higher microbial load will demand higher chlorine dose and contact time. The physiological state of the microbes plays an important role in their chlorine sensitivity
[2][13]. Generally, autochthonous microbes in natural water environments are more resistant to chlorination than laboratory-grown strains.
2. Mechanisms of Chlorine Disinfection
The biocidal mechanism of chlorination remains poorly understood. Some investigators opined that HOCl and OCl
− formed when chlorine is added to water destroy microbial macromolecules. Further work on this supposition led to the “multiple hit” concept, which asserts that bacterial kill by chlorination is probably due to the attack and damage of microbial biomolecules
[3][23]. Venkobachar et al.
[4][24] and Haas
[5][25] reported leakage of cellular macromolecules when bacteria were treated with chlorine. Venkobachar
[4][24] also observed that chlorine significantly inhibits oxidative phosphorylation and the uptake of oxygen, and that, the inhibition of respiratory enzyme was responsible for the phosphorylation inhibition phenomenon, rather than a deficiency in phosphate uptake. In a different study, Chang
[6][26] suggested that the extensive destruction of bacterial enzymatic systems is responsible for the rapid destruction of pathogens during chlorination. Various other investigators have also recognized that chlorine destroys microorganisms through changes in membrane permeability, nucleic acid damage and leakage of intracellular biomolecules
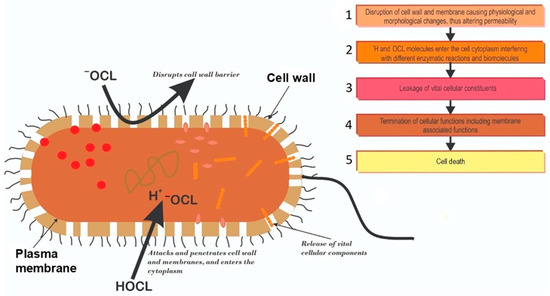
[7][8][27,28]. Generally, it appears that chlorination causes physiological and morphological changes to the bacterial cell wall, altering its permeability. The chlorine molecules subsequently enter the cytoplasm, interfering with different enzymatic reactions, in a possible event cascade depicted in
Figure 23.
Figure 23. Schematic presentation of the possible events cascade of chlorine disinfection leading to microbial cell death. 1: Dissociating chlorine molecules attack and destroy bacterial cell walls altering permeability; 2: Entry of chlorine molecules into the cytoplasm interfering with biomolecules and enzymatic reactions; 3: Altered permeability causes vital cellular components leakage; 4: The series of events lead to the loss of cellular constituents and functions; 5: The loss of cellular constituents and function lead to eventual cell death.
3. Incidences of Chlorine Tolerant Microorganisms from Treated Water Sources
Various international and local authorities have regulations to ensure the protection of water sources from chemical and microbiological contaminants. At a global level, for instance, the WHO recommends chlorine residual of 0.2 to 0.5 mg/L in water supplies
[9][29], while the South African Department of Water and Sanitation stipulates chlorine residual of 0.25 mg/L for discharged wastewater effluents released into surface water environment
[10][11][30,31]. Although some concerns have emerged in recent years about the limitations of chlorine usage in water treatments, there is no doubt, however, that chlorination remains one of the most significant advances in water purification and public health protection
[12][32]. Reports on chlorine surviving microorganisms from chlorinated water sources continue to emanate
[13][14][15][33,34,35]. Various investigators have proposed different mechanisms by which microorganisms may develop tolerance to disinfectants. These include (i) cell surface modifications or encapsulation
[16][36], (ii) attachment to surfaces or suspended particulate matter
[17][37], (iii) microbial aggregation in biofilms
[18][38], (iv) expression of multidrug efflux pump activity
[19][9] and (v) spore formation
[20][39]. These mechanisms conceivably favor the survival of opportunistic waterborne pathogens, thereby contributing to their persistence in water
[21][40].
Table 1 lists some published studies on chlorine-resistant microorganisms from different sources and their reported mechanism of resistance.
Table 1.
Summary of some reports of disinfectant-resistant microbes isolated from diverse aquatic sources.
| Source |
Microorganism |
Disinfectant Concentration/Time |
Mechanism(s) of Resistance |
Reference |
| Drinking water |
P. aeruginosa |
≤0.5 mg/L Cl− |
Natural resistance due to the permeability barrier caused by outer membrane lipopolysaccharides; biofilm formation |
[22][41] |
| Experimental isolates |
Acinetobacter
baumannii |
0.2–4 mg/L |
Increased expression of efflux pumps other antibiotic resistance genes |
[23][42] |
| Drinking water reservoir |
Acinetobater species, Serratia species |
2 mg/L |
Not determined |
[15][35] |
| Sewage |
Bacillus species |
0.1 mg/L NaOCl |
Probable spore formation |
[24][43] |
| Secondary effluent |
Citrobacter species |
0.5 mg/L Ca(OCl)2 for 30 min |
Not determined |
[25][44] |
| Drinking water |
Bacillus species, Actinomycete |
10 mg/L NaOCl for 2 min |
Cellular aggregation or adhesion to suspended particulate. Production of extracellular slime or capsular material |
[26][45] |
| Drinking water and experimental isolates |
Heterotrophic bacteria, faecal coliforms, E. coli, Salmonella typhimurium, Yersinia enterocolitica, Shigella sonnie |
2.0 mg/L free chlorine for 1 h |
Bacterial attachment to surface and production of extracellular slime layer |
[27][46] |
| Chlorine-demand–free buffer solution |
Coliform isolated from drinking water systems and Enteric bacterial from culture collections cocultured with protozoa (Ciliates and amoebae). |
2–4 mg/L free chlorine for 1–2 h |
Shielding of bacteria from chlorine by ingesting protozoans (cysts) and, thus, enhancing resistance |
[28][47] |
| Treated drinking water |
S. aureus, Micrococcus varians, Aeromonas hydrophila |
1–100 mg/L Ca(OCl)2 solution for 30 min |
Possible synthesis of unique proteins or aggregation of bacteria or encapsulation |
[13][33] |
| Environmental isolates (Wastewater clarifier effluent) suspended in phosphate buffer saline |
Enterococcus species |
0.5 mg/L Ca(OCl)2 for 30 min |
Not determined |
[29][48] |
| Environmental strains cultured in sterile phosphate buffer solution |
Legionella pneumophila from environmental water cocultured with Acanthamoeba species |
2–3 mg/L Cl2 for 1 h with a residual Cl2 of 1 mg/L after 1 h |
Possible phenotypic modification of Legionella pneumophila due to intra-cellular growth with Acanthamoeba sp. |
[30][49] |
| Environmental isolates from wastewater treatment plants suspended in saline |
Bacillus species, Citrobacter freundii, Enterobacter species, Kluyvera cryocrescens, Kluyvera intermedia |
0.5 mg/L NaOCl for 30 min |
Authors suggested the possible expression of certain stress factor genes which may reduce bacterial metabolism or change the permeability of cell membranes |
[31][50] |
| Environmental isolates (Wastewater clarifier effluent) suspended in phosphate buffer saline |
E. coli |
0.5 mg/L NaOCl for 30 min |
Not determined |
[14][34] |