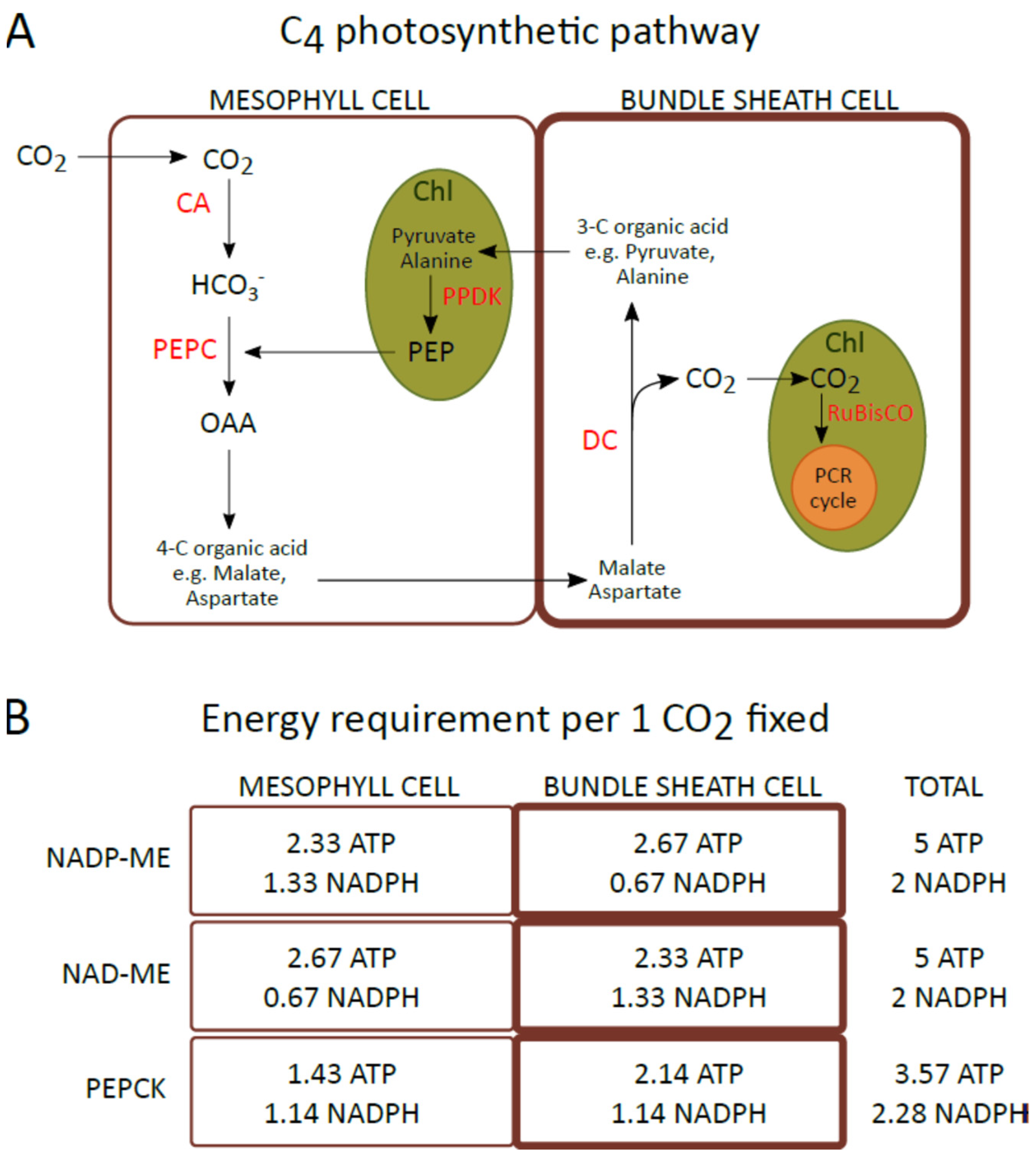
Most C4 plants that naturally occur in tropical or subtropical climates, in high light environments, had to evolve a series of adaptations of photosynthesis that allowed them to grow under these conditions. Some mechanisms that function under changing light conditions, particularly in high light intensity, are universal and are also found in C3 plants. However, some are modified in C4 plants to provide more efficient CO2 assimilation. The close relationship between the light phase of photosynthesis and the enzymatic reactions in chloroplasts, and the associated demand for ATP and NADPH, results that in C4 plants the linear and cyclic electron transport operate in a different ratio in the chloroplasts of mesophyll (M) and bundle sheath (BS) cells. In addition, differences in the intensity of light reaching M and BS chloroplasts and in the thylakoid structure (granal and agranal) will affect the processes of the redistribution of excitation energy between photosystems and the dissipation of its excess. Therefore, it can be assumed that, in the M chloroplasts, because of increased incoming light energy, the mechanisms related to the dissipation of excess energy must function better than in BS chloroplasts to prevent photosystems from photoinhibition and, in consequence, from a decrease in ATP and NADPH. On the other hand, BS chloroplasts, which receive less light energy, must have better functioning mechanisms that allow for its efficient use.
- bundle sheath chloroplasts
- C4 photosynthesis
- mesophyll chloroplasts
1. Diversity of C4 Photosynthesis
2. Ways of Light Energy Utilization: Balanced Distribution between Photosystems and Emission of Excess Energy as a Heat
Environmental factors, especially light intensity and quality during growth, can cause changes in pigment–protein complexes involved in the light reactions of photosynthesis. In some cases, light can cause photoinhibition, which is associated with the overproduction of reactive oxygen species (ROS) and damage of thylakoid membrane components [28]. The plant response may be more complex when there are several environmental factors, which in the case of C4 plants is quite natural due to their ability to grow in hot, and dry conditions with high light intensity [29][30]. The light energy that has been absorbed by chlorophyll molecules can be used in three ways: it can be used for photochemical reactions, its excess can be dissipated as heat, or it can be transferred as light, i.e., fluorescence (Figure 2).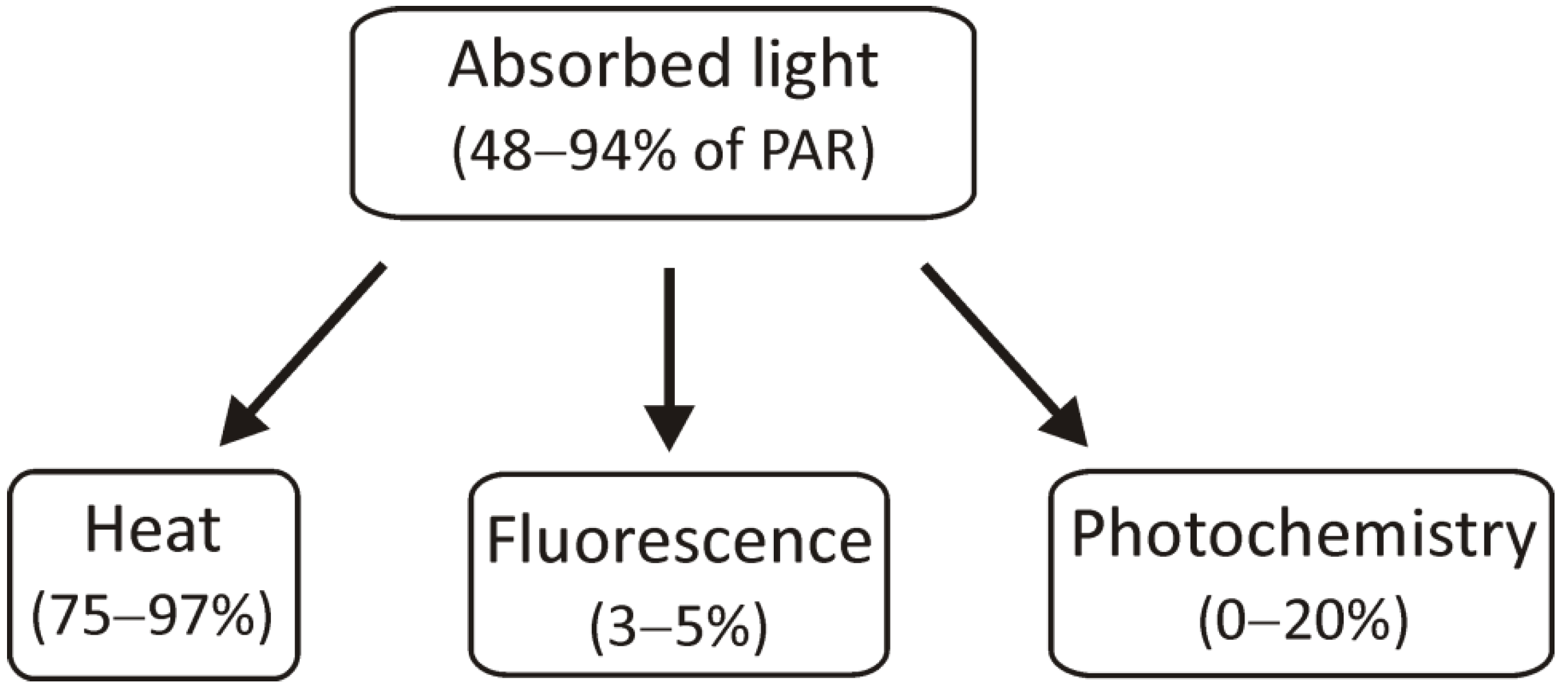
2.1. Elevation of Cyclic Electron Transport Components
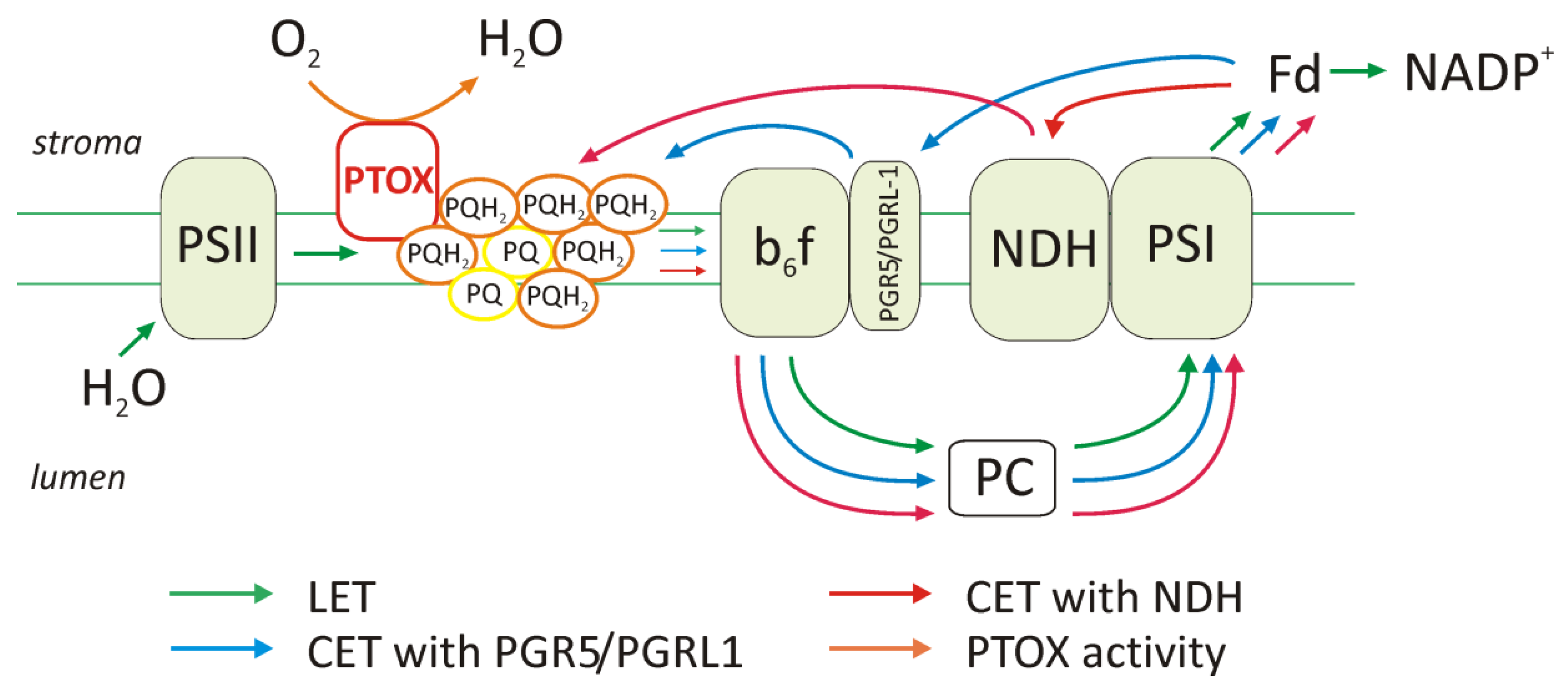
2.2. Function of PTOX Protein and Chlororespiration
2.3. Changes in the Amount of Thylakoid Complexes
The LHCII antenna system is one of the complexes in thylakoid membranes whose content changes under conditions of varying light intensities [36]. Adaptation to high light intensities in maize is a tightly coordinated regulation of the components/activity of the light reaction in both mesophyll and bundle sheath chloroplasts [37]. Under different light intensities, both the content of individual proteins and the arrangement of the complexes can change. Low light intensity promotes the development of antenna systems to capture as much light energy as possible, in contrast to high light when LHC systems decrease. When the intensity of light increases during growth, the levels of the PSII and PSI reaction centers, as well as the cytochrome b6f complex, increase [37]. It has also been shown that the content of LHCII antenna systems increases under conditions of low light, even in maize BS chloroplasts where the PSII content is reduced. Light, both its quantity and quality, as well as other environmental factors (e.g., temperature, CO2 and O2 concentration, drought, and also phosphate availability) affect the expression of chloroplast genes, which is dependent on the redox state of the chloroplasts. The PQH2/PQ ratio influences the transcription genes that encode the proteins of the PSI and PSII reaction centers [38], allowing the photosynthetic apparatus to be adjusted to the actual conditions. Maintaining the oxidized pool of plastoquinone (PQ) by exposure to PSI excitation light or by inhibiting electron transport in PSII was found to activate transcription of the psbA gene encoding D1—the core protein of PSII. In contrast, when the pool of plastoquinone is in a reduced state (PQH2), during exposure to PSII excitation light, the transcription of the psaA and psaB genes encoding the PSI reaction center proteins is activated. Variable light conditions also influence the rate of the Calvin cycle. Therefore, if the demand for ATP and NADPH and their production in light reactions of photosynthesis changes, the degree of reduction of the plastoquinone pool will also change. This has an impact on the expression of genes encoding core proteins of the photosystems, and this, in turn, may change the proportion of cyclic or linear electron transport under given conditions.2.4. Photoinhibition and Role of D1 Protein Phosphorylation
Photoinhibition is a phenomenon that leads to a decrease in photosynthetic activity and a reduction in CO2 assimilation. It is defined as the light-induced inhibition of photosystem II activity [39] when photosystem II degradation dominates over its repair [40]. The classic model of photoinhibition assumed the generation of reactive oxygen species by excessive reduction of the plastoquinone pool. The formed reactive oxygen species are responsible for the damage to the PSII reaction center. Currently, many authorscholars indicate that PSII repair processes are more sensitive to environmental stresses [28][41]. Photosystem II is considered to be the most damage-sensitive complex of the thylakoid membrane, which does not mean that PSI is not affected by photoinhibition as well. PSI photoinhibition occurs when the supply of electrons from PSII exceeds the acceptor capacity of PSI [42], but PSI is effectively protected against damage, for example, by photoinhibition of a certain pool of PSII. Repair of damaged PSII reaction centers requires the degradation of the D1 protein destroyed during photoinhibition, its de novo synthesis, and reconstruction of the PSII complex. D1 degradation is a multistage process regulated by protein phosphorylation and dephosphorylation, and also by the level of ATP in chloroplasts. The D1 protein, one of the most easily degraded, is phosphorylated under the influence of medium and high intensity of light in the granum of thylakoids by membrane-bound serine threonine kinase. This modification affects both intact and damaged reaction centers, protects against proteolytic degradation, and has no effect on the electron transport rate in PSII [43]. When irreversible damage of D1 is caused by photoinhibition, this D1 is directed to the thylakoid stroma, where it is dephosphorylated and then degraded [44].2.5. State Transitions and Phosphorylation of LHCII
At different light intensities, the migration of LHCII between photosystems is observed in the process called state transitions. The LHCII antenna, and especially the Lhcb2 protein, undergoes reversible phosphorylation, which is crucial for the switching of LHCII between photosystems. The levels of LHCII phosphorylation are lower at high light compared with those under low light conditions. State 1 is traditionally defined as the condition when PSI is preferentially excited and all LHCIIs become associated with PSII. Illumination conditions, which lead to an excess excitation of photosystem II (PSII), compared to photosystem I (PSI), induce a transition to state 2, in which the more absorbed excitation energy is diverted to PSI because the phosphorylated LHCII antennas are associated with PSI [45]. State transitions act as a mechanism to balance the excitation of the two photosystems under changing light regimes [46]. In C4 plants where there are differences in the organization of thylakoid membranes in the M and BS chloroplasts, the process may be quite different. Thylakoid membranes are heterogeneous, and while PSII with the LHCII antenna is located in the stacked regions of the grana, the PSI occurs in the stroma lamellae and marginal grana regions. Thus, the number of grana in a given chloroplast type in each metabolic subtype will determine the LHCII content and the amount of PSI to which these antennas can potentially be attached.2.6. Xanthophyll Cycle and Heat Dissipation
Among several mechanisms in chloroplasts that allow them to function under stress conditions, preventing the generation of ROS is extremely important. One of the protection mechanisms is the quenching of excess energy as a thermal dissipation. This process involves the xanthophyll cycle, related to the conversion of violaxanthin to zeaxanthin [47] and the protonation of the PsbS protein. Generally, when plants are exposed to high light intensities, violaxanthin is oxidized by violaxanthin de-epoxidase (VDE). This leads to the formation of antheraxanthin, followed by zeaxanthin. Zeaxanthin creates a barrier that prevents overexcitation of the PSII reaction center. The energy from LHCII is dissipated and is not directed to the reaction center. At low light intensities, zeaxanthin epoxidation catalyzed by zeaxanthin epoxidase (ZE) occurs. The content of carotenoids, including xanthophylls participating in the xanthophyll cycle, in M and BS chloroplasts was investigated by the group of Romanowska et al. [48]. An increase in zeaxanthin content was also observed in Sorghum bicolor (NADP-ME plant), after exposure to the same light intensity as described above. The amount of zeaxanthin, antheraxanthin, and violaxanthin participating in the xanthophyll cycle was twice as high compared to the control [49], which may indicate that regardless of the species, the functioning of the xanthophyll cycle is an important element of protection of the photosynthetic apparatus and dissipation of excess energy. The participation of the LHCII antenna in energy quenching was confirmed in the mesophyll chloroplasts of maize (NADP-ME), where after the high intensity of far-red light, the LHCII were dephosphorylated, detached from the PSI in the stroma lamellae, moved to the grana, and either bound to PSII or formed aggregates which in consequence, lead to induction of the qE parameter [46]. In M chloroplasts, light is not a factor that limits the production of ATP and NADPH, so the excess light energy is dissipated as heat.References
- Yamori, W.; Hikosaka, K.; Way, D.A. Temperature response of photosynthesis in C3, C4, and CAM plants: Temperature acclimation and temperature adaptation. Photosynth. Res. 2014, 119, 101–117.
- Hatch, M.D.; Slack, C.R. Photosynthesis by sugar-cane leaves. A new carboxylation reaction and the pathway of sugar formation. Biochem. J. 1966, 101, 103–111.
- Slack, C.R.; Hatch, M.D. Comparative studies on the activity of carboxylases and other enzymes in relation to the new pathway of photosynthetic carbon dioxide fixation in tropical grasses. Biochem. J. 1967, 103, 660.
- Osmond, C.B. Beta-carboxylation during photosynthesis in Atriplex. Biochim. Biophys. Acta 1967, 141, 197–199.
- Hatch, M.D. C4 photosynthesis: A unique elend of modified biochemistry, anatomy and ultrastructure. Biochim. Biophys. Acta Rev. Bioenerg. 1987, 895, 81–106.
- Gutierrez, M.; Gracen, V.E.; Edwards, G.E. Biochemical and cytological relationships in C4 plants. Planta 1974, 119, 279–300.
- Hatch, M.; Kagawa, T.; Craig, S. Subdivision of C4-Pathway Species Based on Differing C4 Acid Decarboxylating Systems and Ultrastructural Features. Funct. Plant Biol. 1975, 2, 111–128.
- Furbank, R.T. Evolution of the C4 photosynthetic mechanism: Are there really three C4 acid decarboxylation types? J. Exp. Bot. 2011, 62, 3103–3108.
- Edwards, G.E.; Voznesenskaya, E.V. Chapter 4 C4 Photosynthesis: Kranz Forms and Single-Cell C4 in Terrestrial Plants. In C4 Photosynthesis and Related CO2 Concentrating Mechanisms; Springer: Dordrecht, The Netherlands, 2011; pp. 29–61.
- Walker, B.J.; Kramer, D.M.; Fisher, N.; Fu, X. Flexibility in the Energy Balancing Network of Photosynthesis Enables Safe Operation under Changing Environmental Conditions. Plants 2020, 9, 301.
- Hatch, M.D. C4 Photosynthesis: An Unlikely Process Full of Surprises. Plant Cell Physiol. 1992, 33, 333–342.
- Hatch, M.D.; Osmond, C.B. 4. Compartmentation and Transport in C4 Photosynthesis. In Encyclopedia of Plant Physiology, New Series Vol 3, Transport in Plants III; Stocking, C.R., Heber, U., Eds.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1976; pp. 144–184.
- Yoshimura, Y.; Kubota, F.; Ueno, O. Structural and biochemical bases of photorespiration in C4 plants: Quantification of organelles and glycine decarboxylase. Planta 2004, 220, 307–317.
- Fisher, D.D.; Schenk, H.J.; Thorsch, J.A.; Ferren, W.R. Leaf anatomy and subgeneric affiliations of C3 and C4 species of Suaeda (Chenopodiaceae) in North America. Am. J. Bot. 1997, 84, 1198–1210.
- Voznesenskaya, E.V.; Franceschi, V.R.; Pyankov, V.I.; Edwards, G.E. Anatomy, chloroplast structure and compartmentation of enzymes relative to photosynthetic mechanisms in leaves and cotyledons of species in the tribe Salsoleae (Chenopodiaceae). J. Exp. Bot. 1999, 50, 1779–1795.
- Voznesenskaya, E.V.; Franceschi, V.R.; Chuong, S.D.X.; Edwards, G.E. Functional characterization of phosphoenolpyruvate carboxykinase-type C4 leaf anatomy: Immuno-, cytochemical and ultrastructural analyses. Ann. Bot. 2006, 98, 77–91.
- Furbank, R.; Hatch, M. Mechanism of C 4 Photosynthesis: The Size and Composition of the Inorganic Carbon Pool in Bundle Sheath Cells. Plant Physiol. 1987, 85, 958–95864.
- Burnell, J.N.; Hatch, M.D. Low Bundle Sheath Carbonic Anhydrase Is Apparently Essential for Effective C4 Pathway Operation. Plant Physiol. 1988, 86, 1252–1256.
- Sage, R.F.; Christin, P.-A.; Edwards, E.J. The C4 plant lineages of planet Earth. J. Exp. Bot. 2011, 62, 3155–3169.
- Sage, R.F.; Stata, M. Photosynthetic diversity meets biodiversity: The C4 plant example. J. Plant Physiol. 2015, 172, 104–119.
- Brown, R.H. A Difference in N Use Efficiency in C3 and C4 Plants and its Implications in Adaptation and Evolution. Crop Sci. 1978, 18, 93–98.
- Sage, R.F.; Pearcy, R.W.; Seemann, J.R. The Nitrogen Use Efficiency of C3 and C4 Plants: III. Leaf Nitrogen Effects on the Activity of Carboxylating Enzymes in Chenopodium album (L.) and Amaranthus retroflexus (L.). Plant Physiol. 1987, 85, 355.
- Jordan, D.B.; Ogren, W.L. The CO2/O2 specificity of ribulose 1,5-bisphosphate carboxylase/oxygenase. Planta 1984, 161, 308–313.
- Sage, R.F. A portrait of the C4 photosynthetic family on the 50th anniversary of its discovery: Species number, evolutionary lineages, and Hall of Fame. J. Exp. Bot. 2017, 68, e11–e28.
- Bianconi, M.E.; Dunning, L.T.; Moreno-Villena, J.J.; Osborne, C.P.; Christin, P.A. Gene duplication and dosage effects during the early emergence of C4 photosynthesis in the grass genus Alloteropsis. J. Exp. Bot. 2018, 69, 1967–1980.
- Williams, B.P.; Aubry, S.; Hibberd, J.M. Molecular evolution of genes recruited into C4 photosynthesis. Trends Plant Sci. 2012, 17, 213–220.
- von Caemmerer, S.; Furbank, R.T. Strategies for improving C4 photosynthesis. Curr. Opin. Plant Biol. 2016, 31, 125–134.
- Nishiyama, Y.; Murata, N. Revised scheme for the mechanism of photoinhibition and its application to enhance the abiotic stress tolerance of the photosynthetic machinery. Appl. Microbiol. Biotechnol. 2014, 98, 8777–8796.
- Ghannoum, O. C4 photosynthesis and water stress. Ann. Bot. 2009, 103, 635–644.
- Osborne, C.P.; Freckleton, R.P. Ecological selection pressures for C4 photosynthesis in the grasses. Proc. R. Soc. B Biol. Sci. 2009, 276, 1753–1760.
- Ishikawa, N.; Takabayashi, A.; Noguchi, K.; Tazoe, Y.; Yamamoto, H.; Von Caemmerer, S.; Sato, F.; Endo, T. NDH-Mediated Cyclic Electron Flow Around Photosystem I is Crucial for C4 Photosynthesis. Plant Cell Physiol. 2016, 57, 2020–2028.
- Shikanai, T. Chloroplast NDH: A different enzyme with a structure similar to that of respiratory NADH dehydrogenase. Biochim. Biophys. Acta Bioenerg. 2016, 1857, 1015–1022.
- Krieger-Liszkay, A.; Feilke, K. The Dual Role of the Plastid Terminal Oxidase PTOX: Between a Protective and a Pro-oxidant Function. Front. Plant Sci. 2016, 6, 1147:1–1147:3.
- Lennon, A.M.; Prommeenate, P.; Nixon, P.J. Location, expression and orientation of the putative chlororespiratory enzymes, Ndh and IMMUTANS, in higher-plant plastids. Planta 2003, 218, 254–260.
- Essemine, J.; Lyu, M.J.A.; Qu, M.; Perveen, S.; Khan, N.; Song, Q.; Chen, G.; Zhu, X.G. Contrasting Responses of Plastid Terminal Oxidase Activity Under Salt Stress in Two C4 Species With Different Salt Tolerance. Front. Plant Sci. 2020, 11, 1009.
- Leong, T.Y.; Anderson, J.M. Adaptation of the thylakoid membranes of pea chloroplasts to light intensities. I. Study on the distribution of chlorophyll-protein complexes. Photosynth. Res. 1984, 5, 105–115.
- Drozak, A.; Romanowska, E. Acclimation of mesophyll and bundle sheath chloroplasts of maize to different irradiances during growth. Biochim. Biophys. Acta Bioenerg. 2006, 1757, 1539–1546.
- Pfannschmidt, T.; Nilsson, A.; Allen, J.F. Photosynthetic control of chloroplast gene expression. Nature 1999, 397, 625–628.
- Murata, N.; Takahashi, S.; Nishiyama, Y.; Allakhverdiev, S.I. Photoinhibition of photosystem II under environmental stress. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 414–421.
- Demmig-Adams, B.; Cohu, C.M.; Muller, O.; Adams, W.W. Modulation of photosynthetic energy conversion efficiency in nature: From seconds to seasons. Photosynth. Res. 2012, 113, 75–88.
- Kangasjärvi, S.; Neukermans, J.; Li, S.; Aro, E.M.; Noctor, G. Photosynthesis, photorespiration, and light signalling in defence responses. J. Exp. Bot. 2012, 63, 1619–1636.
- Tikkanen, M.; Grebe, S. Switching off photoprotection of photosystem I—A novel tool for gradual PSI photoinhibition. Physiol. Plant. 2018, 162, 156–161.
- Pokorska, B.; Zienkiewicz, M.; Powikrowska, M.; Drozak, A.; Romanowska, E. Differential turnover of the photosystem II reaction centre D1 protein in mesophyll and bundle sheath chloroplasts of maize. Biochim. Biophys. Acta Bioenerg. 2009, 1787, 1161–1169.
- Rintamäki, E.; Kettunen, R.; Aro, E.M. Differential D1 Dephosphorylation in Functional and Photodamaged Photosystem II Centers: Dephosphorylation is a prerequisite for degradation of damaged D1. J. Biol. Chem. 1996, 271, 14870–14875.
- Tikkanen, M.; Grieco, M.; Kangasjärvi, S.; Aro, E.M. Thylakoid Protein Phosphorylation in Higher Plant Chloroplasts Optimizes Electron Transfer under Fluctuating Light. Plant Physiol. 2010, 152, 723–735.
- Rogowski, P.; Wasilewska-Dębowska, W.; Urban, A.; Romanowska, E. Maize bundle sheath chloroplasts—A unique model of permanent State 2. Environ. Exp. Bot. 2018, 155, 321–331.
- Jahns, P.; Holzwarth, A.R. The role of the xanthophyll cycle and of lutein in photoprotection of photosystem II. Biochim. Biophys. Acta 2012, 1817, 182–193.
- Zienkiewicz, M.; Krupnik, T.; Drozak, A.; Golke, A.; Romanowska, E. Chloramphenicol acetyltransferase- a new selectable marker in stable nuclear transformation of the red alga Cyanidioschyzon merolae. Protoplasma 2017, 254, 587–596.
- Sharma, P.K.; Hall, D.O. Changes in Carotenoid Composition and Photosynthesis in Sorghum under High Light and Salt Stresses. J. Plant Physiol. 1992, 140, 661–666.
