Neurotropic viruses target the brain and contribute to neurologic diseases. Caspase recruitment domain containing family member 9 (CARD9) controls is an adaptor protective immunity in a variety of infectious disorders. To investigate the effect of CARD9 in neurotropic virus infection, CARD9−/− and corresponding C57BL/6 wildin that delivers signals through immunoreceptor tyrosine-based activation motif (ITAM)-coupled or hemITAM-bearing C-type control mice were infected with Theiler’s murine encephalomyelitis virus (TMEV). Brain tissue was analyzed by histology, immunohistochemistry and molecular analyses, and spleens by flow cytometry. To determine the impact of CARD9 deficiency on T cell responses in vitro, antigen presentation assays were utilized. Genetic ablation of CARD9 enhanced early pro-inflammatory cytokinlectin receptors (CLRs), including Dectin-1, Dectin-2 and Mincle. The Syk/CARD9 signaling axis triggered by engagement of Dectin-1, Dectin-2, or Mincle was recently shown to contribute to early innate responses and accelerated infiltration of T and B cells in the brain, together with a transient increase in TMEV-infected cells in the hippocampus. CARD9−/− mice showed an increased loss of neuronal nuclear protein+ mature neurongainst certain viruses, but also against bacteria, parasites and fungal pathogens. Viruses, such as vesicular stomatitis virus (VSV), encephalomyocarditis virus and doublecortin+ neuronal precursor cells and an increase in β-amyloid precursor protein+ damaged axons in the hippocampus. No effect of CARD9 deficiency was found on the initiation of CD8+ T cell responses by flow cytometry and co-culture experiments using virus-exposed dendritic cells or microglia-enriched glial cell mixtures, respectively. The present study indicates that vaccinia virus, which are sensed by the cytosolic nucleic sensors RIG-I, MDA5 and RAD50, respectively, were shown to engage CARD9 for inflammatory cytokine production. However, in contrast to the predominant protective effect observed in fungal diseases, CARD9 is dispensable for the initiation of early signaling seems to play an ambivalent role in viral disorders, with beneficial effects by promoting antiviral responses and TMEV elimination but may contribute to the modulation of neuroinflammation, thereby reducing hippocampal injury following neurotropic virus infection(e.g., VSV, vaccinia virus) and detrimental effects by triggering virus-induced immunopathology (e.g., influenza virus, coxsackievirus) being described in animal experiments.
- Theiler’s murine encephalomyelitis virus
- neurotropic virus
- CARD9
- hippocampal damage
1. Introduction
2. CARD9 Deficiency Increases Hippocampal Injury Following Acute Neurotropic Picornavirus Infection but Does Not Affect Pathogen Elimination
2.1. Loss of CARD9 Promotes Hippocampal Damage Following Virus Infection
2. Loss of CARD9 Promotes Hippocampal Damage Following Virus Infection
TMEV primarily targets the hippocampus of mice and causes polioencephalitis in C57BL/6 mice during acute infection [15]. Histology of infected mice revealed the presence of inflammatory responses within the hippocampus characterized by mononuclear infiltrates and gliosis associated with neuronal loss of the stratum pyramidale. A significantly increased severity of hippocampal damage, as determined by semiquantitative scoring, was found in CARD9−/− mice compared to WT mice at 7 dpi (p = 0.03; Figure 1A–C). Accordingly, an increased loss of NeuN+ mature neurons in the stratum pyramidale was found in CARD9−/− mice compared to WT mice at 7 dpi by densitometry (p = 0.05; Figure 1D–F). To detect more subtle brain lesions, labeling of DCX+ neuronal progenitor cells and β-APP, as a marker for damaged axons, was performed. Here, a significant reduction in DCX+ cells were observed in the hippocampal dentate gyrus of CARD9−/− mice compared to WT mice at 7 dpi (p < 0.01) and 14 dpi (p = 0.01; Figure 1G–I), indicative of disturbed neurogenesis. Moreover, a significant accumulation of swollen β-APP+ axons preferentially in the cornu ammonis region CA1 of CARD9−/− mice compared to WT mice was detected at 7 dpi (p = 0.03) and 14 dpi (p = 0.03; Figure 1J–L).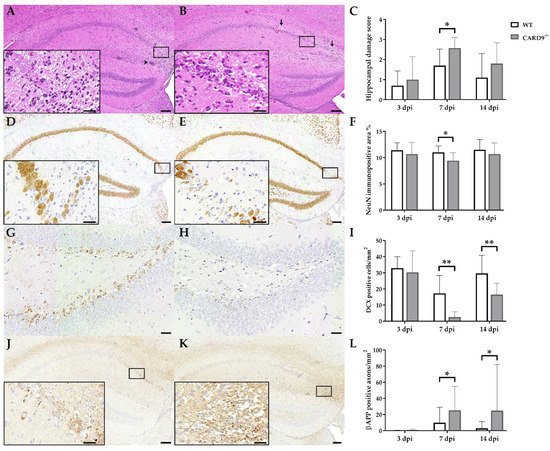
2.2. CARD9 Deficiency Transiently Increases the Viral Burden but Does Not Prevent Viral Clearance
3. CARD9 Deficiency Transiently Increases the Viral Burden but Does Not Prevent Viral Clearance
TMEV antigen was found predominantly in hippocampal pyramidal neurons of the CA1 regions in both CARD9−/− and WT mice following infection, as demonstrated by immunohistochemistry. Quantification revealed a significant increase in TMEV-infected cells in the hippocampus of CARD9−/− mice compared to WT mice at 7 dpi (p = 0.05). At 14 dpi, only a few TMEV-infected cells were detected in the forebrain in CARD9−/− mice and WT mice, indicating virus elimination in both groups (Figure 2A–C). Few TMEV-infected cells were found in the brain stem of infected mice without significant differences observed between CARD9−/− and WT mice (Figure 2D–F). In addition, only a few TMEV-infected cells were found in the spinal cord of animals (Figure S2) without differences observed between both groups. No infected cells were found in the cerebellum. Using RT-qPCR, an increase in TMEV-RNA levels were only detected by trend (p = 0.07) in the brain of CARD9−/− mice at 3 dpi, and no differences of viral RNA levels in the brain of CARD9−/− and WT mice were detected at 7 and 14 dpi (Figure S3).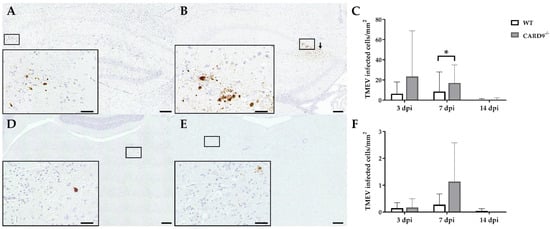
2.3. Hippocampal Damage in CARD9
4. Hippocampal Damage in CARD9
−/−
Mice Is Associated with an Increased T Cell, B Cell, Regulatory T Cell and M2-Type Myeloid Cell Infiltration
Immunohistochemistry was performed to phenotype immune cell sequestration in the hippocampus. Infected CARD9−/− mice showed a significantly enhanced recruitment of CD3+ T cells in the hippocampus at 14 dpi (p = 0.02) compared to infected WT mice (Figure 3A–C).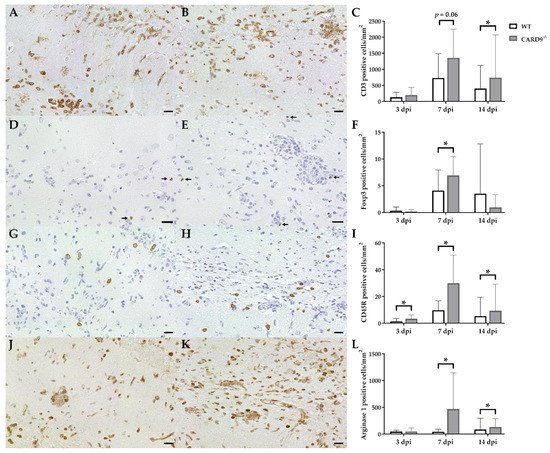
2.4. CARD9
5. CARD9
−/−
Mice Develop Early Pro-Inflammatory Cytokine Responses in the Brain
Cytokine expression analyses were performed to characterize inflammatory responses in the brain in more detail. RT-qPCR revealed a significant increase in IL-1β (p = 0.02) and IFN-γ (p = 0.03) in the brain of CARD9−/− mice compared to WT mice following TMEV infection at 3 dpi (Figure 4A,B). At 7 dpi, a significantly increased transcription of IL-5 (p = 0.05) was found in the brain of infected CARD9−/− mice compared to WT controls (Figure 4C). Gene expression of IL-1α, IL-4, IL-6, IL-10, TGF-β1 and TNF-α did not differ between groups at any investigated time point post infection (Figure S4). Between non-infected CARD9−/− mice and WT mice, no differences were found in the brain for all investigated cytokines (Figure S6).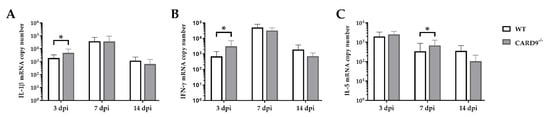
2.5. CARD9 Deficiency Has a Limited Effect on Initial CD8
6. CARD9 Deficiency Has a Limited Effect on Initial CD8
+
T Cell Responses
Timely peripheral activation and priming of naïve T cells in lymphoid organs during the early phase was shown to be crucial for mounting robust antiviral responses in the TMEV model [40,41][24][25]. To determine whether CARD9 deficiency affects peripheral T cell activation, flow cytometric analysis of spleen tissue has been performed. In agreement with the observed limited effect of CARD9 on the viral brain load, analyses revealed no variations of T cell activation marker expression between CARD9−/− mice and WT mice at 3 and 7 dpi (Figure 5A,B). Increased frequencies of splenic CD8+ T cells, CD4+CD62Llow and CD8+CD62low T cells were detected in CARD9−/− mice compared to WT mice at 14 dpi (Figure 5C), probably associated with hippocampal damage and the prolonged CD3+ T cell responses in the brain determined by immunohistochemistry. Non-infected CARD9−/− mice showed increased frequencies of splenic CD4+ and CD8+ cells compared to WT mice, while no differences in CD4+CD62Llow and CD8+CD62low T cell frequencies were found between both groups under non-infectious conditions (Figure S7).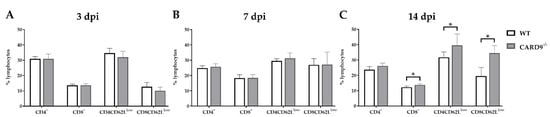
2.6. CARD9 Deficiency Does Not Affect Antigen Presenting Cells Ability to Prime CD8
7. CARD9 Deficiency Does Not Affect Antigen Presenting Cells Ability to Prime CD8
+
T Cells Following Theiler’s Murine Encephalomyelitis Virus Infection In Vitro
To support the finding of a limited effect of CARD9 deficiency on T cell priming and to test the direct effect of CARD9 deficiency upon antigen presentation, BMDC as professional antigen presenting cells (APC) were utilized in an in vitro stimulation assay. WT and CARD9−/− BMDC were incubated with live TMEV DA at a MOI 200, which had no visible effect on BMDC viability, but is necessary to induce adequate APC-specific effector functions. The presence of the virus led to BMDC activation, indicated by an upregulation of the surface marker CD86 and MHC I and an increased release of inflammatory cytokines IL-6 and IFN-β (Figure S5). No differences in activation marker expression and cytokine production were observed between WT and CARD9−/− BMDCs following activation with TMEV. To analyze the impact of CARD9 deficiency on early TMEV-mediated T cell responses in vitro, a BMDC/T cell and MEG/T cell co-cultivation system was used. OT-I T cells that specifically recognize the OVA257–264 peptide presented via the MHC I molecule H2-Kb were co-cultured with BMDCs or MEGs that had been exposed to TMEV-OVA. In this setting, non-OVA-expressing TMEV DA was utilized as a negative control. To evaluate CD8+ T cell activation, expression of the T cell activation marker CD69 was analyzed by flow cytometry (Figure 6A,D), and cytokine release was measured by ELISA (Figure 6B,C,E,F). Both co-culture systems revealed a TMEV-OVA-mediated increase in CD8+ T cell activation compared to non-OVA expressing TMEV DA exposed cells, indicated by upregulated levels of CD69 as well as an elevated release of IL-2 and IFN-γ (Figure 6B,C,E,F). However, no difference in CD8+ T cell responses between the WT and CARD9−/− group was observed, neither when BMDCs nor when MEGs were used in the co-culture system. Furthermore, the overall CD8+ T cell response following TMEV-OVA incubation of MEGs was comparably low, suggesting that the potential of antigen processing and presentation of microglia is limited compared to BMDCs in vitro.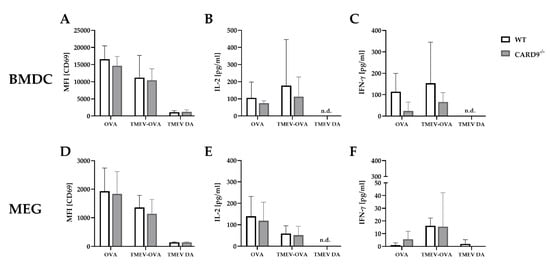
References
- Ludlow, M.; Kortekaas, J.; Herden, C.; Hoffmann, B.; Tappe, D.; Trebst, C.; Griffin, D.E.; Brindle, H.E.; Solomon, T.; Brown, A.S.; et al. Neurotropic virus infections as the cause of immediate and delayed neuropathology. Acta Neuropathol. 2016, 131, 159–184.
- Bröer, S.; Hage, E.; Käufer, C.; Gerhauser, I.; Anjum, M.; Li, L.; Baumgärtner, W.; Schulz, T.F.; Löscher, W. Viral mouse models of multiple sclerosis and epilepsy: Marked differences in neuropathogenesis following infection with two naturally occurring variants of Theiler’s virus BeAn strain. Neurobiol. Dis. 2017, 99, 121–132.
- Carson, P.J.; Konewko, P.; Wold, K.S.; Mariani, P.; Goli, S.; Bergloff, P.; Crosby, R.D. Long-term clinical and neuropsychological outcomes of West Nile virus infection. Clin. Infect. Dis. 2006, 43, 723–730.
- Fowler, Å.; Stödberg, T.; Eriksson, M.; Wickström, R. Long-term outcomes of acute encephalitis in childhood. Pediatrics 2010, 126, e828–e835.
- Vezzani, A.; Fujinami, R.S.; White, H.S.; Preux, P.M.; Blumcke, I.; Sander, J.W.; Loscher, W. Infections, inflammation and epilepsy. Acta Neuropathol. 2016, 131, 211–234.
- Betourne, A.; Szelechowski, M.; Thouard, A.; Abrial, E.; Jean, A.; Zaidi, F.; Foret, C.; Bonnaud, E.M.; Charlier, C.M.; Suberbielle, E.; et al. Hippocampal expression of a virus-derived protein impairs memory in mice. Proc. Natl. Acad. Sci. USA 2018, 115, 1611–1616.
- De Chiara, G.; Piacentini, R.; Fabiani, M.; Mastrodonato, A.; Marcocci, M.E.; Limongi, D.; Napoletani, G.; Protto, V.; Coluccio, P.; Celestino, I.; et al. Recurrent herpes simplex virus-1 infection induces hallmarks of neurodegeneration and cognitive deficits in mice. PLoS Pathog. 2019, 15, e1007617.
- Cornelius, A.D.A.; Hosseini, S.; Schreier, S.; Fritzsch, D.; Weichert, L.; Michaelsen-Preusse, K.; Fendt, M.; Kroger, A. Langat virus infection affects hippocampal neuron morphology and function in mice without disease signs. J. Neuroinflamm. 2020, 17, 278.
- Theiler, M. Spontaneous encephalomyelitis of mice—A new virus disease. Science 1934, 80, 122.
- Libbey, J.E.; Kirkman, N.J.; Smith, M.C.; Tanaka, T.; Wilcox, K.S.; White, H.S.; Fujinami, R.S. Seizures following picornavirus infection. Epilepsia 2008, 49, 1066–1074.
- Stewart, K.-A.A.; Wilcox, K.S.; Fujinami, R.S.; White, H.S. Development of Post-infection Epilepsy Following Theiler Virus Infection of C57BL/6 Mice. J. Neuropathol. Exp. Neurol. 2010, 69, 1210–1219.
- Buenz, E.J.; Rodriguez, M.; Howe, C.L. Disrupted spatial memory is a consequence of picornavirus infection. Neurobiol. Dis. 2006, 24, 266–273.
- Libbey, J.E.; Fujinami, R.S. Neurotropic viral infections leading to epilepsy: Focus on Theiler’s murine encephalomyelitis virus. Future Virol. 2011, 6, 1339–1350.
- Umpierre, A.D.; Remigio, G.J.; Dahle, E.J.; Bradford, K.; Alex, A.B.; Smith, M.D.; West, P.J.; White, H.S.; Wilcox, K.S. Impaired cognitive ability and anxiety-like behavior following acute seizures in the Theiler’s virus model of temporal lobe epilepsy. Neurobiol. Dis. 2014, 64, 98–106.
- Gerhauser, I.; Hansmann, F.; Ciurkiewicz, M.; Löscher, W.; Beineke, A. Facets of Theiler’s Murine Encephalomyelitis Virus-Induced Diseases: An Update. Int. J. Mol. Sci. 2019, 20, 448.
- Bowen, J.L.; Olson, J.K. IFNgamma influences type I interferon response and susceptibility to Theiler’s virus-induced demyelinating disease. Viral Immunol. 2013, 26, 223–238.
- Li, L.; Ulrich, R.; Baumgartner, W.; Gerhauser, I. Interferon-stimulated genes-essential antiviral effectors implicated in resistance to Theiler’s virus-induced demyelinating disease. J. Neuroinflamm. 2015, 12, 242.
- Drappier, M.; Jha, B.K.; Stone, S.; Elliott, R.; Zhang, R.; Vertommen, D.; Weiss, S.R.; Silverman, R.H.; Michiels, T. A novel mechanism of RNase L inhibition: Theiler’s virus L* protein prevents 2–5A from binding to RNase, L. PLoS Pathog. 2018, 14, e1006989.
- Kirkman, N.J.; Libbey, J.E.; Wilcox, K.S.; White, H.S.; Fujinami, R.S. Innate but not adaptive immune responses contribute to behavioral seizures following viral infection. Epilepsia 2010, 51, 454–464.
- Howe, C.L.; LaFrance-Corey, R.G.; Sundsbak, R.S.; LaFrance, S.J. Inflammatory monocytes damage the hippocampus during acute picornavirus infection of the brain. J. Neuroinflamm. 2012, 9, 1–13.
- Howe, C.L.; Lafrance-Corey, R.G.; Sundsbak, R.S.; Sauer, B.M.; Lafrance, S.J.; Buenz, E.J.; Schmalstieg, W.F. Hippocampal protection in mice with an attenuated inflammatory monocyte response to acute CNS picornavirus infection. Sci. Rep. 2012, 2, 545.
- Cusick, M.F.; Libbey, J.E.; Patel, D.C.; Doty, D.J.; Fujinami, R.S. Infiltrating macrophages are key to the development of seizures following virus infection. J. Virol. 2013, 87, 1849–1860.
- Waltl, I.; Kaufer, C.; Gerhauser, I.; Chhatbar, C.; Ghita, L.; Kalinke, U.; Loscher, W. Microglia have a protective role in viral encephalitis-induced seizure development and hippocampal damage. Brain Behav. Immun. 2018, 74, 186–204.
- Chang, J.R.; Zaczynska, E.; Katsetos, C.D.; Platsoucas, C.D.; Oleszak, E.L. Differential expression of TGF-beta, IL-2, and other cytokines in the CNS of Theiler’s murine encephalomyelitis virus-infected susceptible and resistant strains of mice. Virology 2000, 278, 346–360.
- Oleszak, E.L.; Chang, J.R.; Friedman, H.; Katsetos, C.D.; Platsoucas, C.D. Theiler’s virus infection: A model for multiple sclerosis. Clin. Microbiol. Rev. 2004, 17, 174–207.
