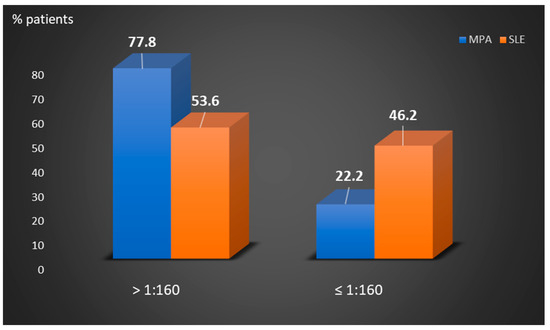
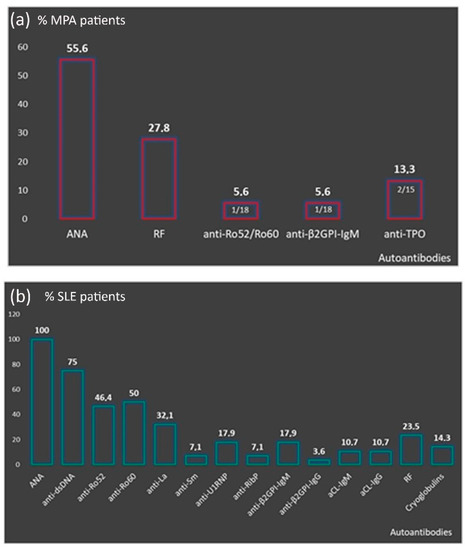
Perinuclear anti-neutrophilic cytoplasmic antibodies (P-ANCA) recognize heterogeneous antigens, including myeloperoxidase (MPO), lactoferrin, elastase, cathepsin-G and bactericidal/permeability-increasing protein. Although P-ANCA have diagnostic utility in vasculitides, they may also be found in patients with various other systemic autoimmune rheumatic diseases (SARDs). Nevertheless, the clinical significance and the targets recognized by P-ANCA in such patients remain unclear. For this purpose, herein we investigated the occurrence of ANCA-related antigenic specificities in 82 P-ANCA-positive sera by multiplex ELISA, as well as their association with other autoantibodies. The P-ANCA-positive sera corresponded to patients with vasculitides (n = 24), systemic lupus erythematosus (n = 28), antiphospholipid syndrome (n = 5), Sjögren’s syndrome (n = 7), rheumatoid arthritis (n = 3), systemic scleroderma (n = 1), sarcoidosis (n = 1) and Hashimoto′s thyroiditis (n = 13). In most P-ANCA-positive patients studied (51/82, 62.3%), these autoantibodies occurred in high titers (>1:160). The analysis of P-ANCA-positive sera revealed reactivity to MPO in only 50% of patients with vasculitides, whereas it was infrequent in the other disease groups studied. Reactivity to other P-ANCA-related autoantigens was also rarely detected. Our findings support that high P-ANCA titers occur in SARD. The P-ANCA-positive staining pattern is associated with MPO specificity in vasculitides, while in other autoimmune diseases, it mostly involves unknown autoantigens.