The detection of phospholipase A2 receptor (PLA2R) and thrombospondin domain containing 7A THSD7A among primary membranous glomerulonephritis (MGN) patients transformed the diagnosis, treatment monitoring, and prognosis. Anti-PLA2R can be detected in 70–90% of primary MGN patients while anti-THSD7A in 2–3% of anti-PLA2R negative primary MGN patients depending on the technique used. Serum and urine samples are less invasive and non-invasive, respectively, and thus can detect the presence of anti-PLA2R and anti-THSD7A with higher sensitivity and specificity, which is significant in patient monitoring and prognosis. It is better than exposing patients to a frequent biopsy, which is an invasive procedure. Different techniques of detection of PLA2R and THSD7A in patients’ urine and sera were reviewed to provide newer and alternative techniques. We proposed the use of biomarkers (PLA2R and THSD7A) in the diagnosis, treatment decision, and follow-up of patients with primary MGN.
- M-type phospholipase A2
- thrombospondin type containing domain A7
- retinol binding protein
- beta-2 microglobulin
- membranous glomerulonephritis
- neutral endopeptidase
1. Introduction
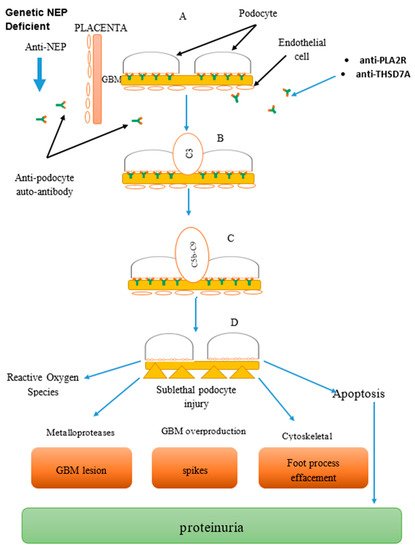
2. Pathogenesis
Immune complex formation in the subepithelial surface of the glomerular complex membrane is central to the formation of membranous glomerulonephritis [12]. Three major mechanisms have been proposed so far [1][2]. The first hypothesis emphasizes on passive entrapment of preformed immune complex due to higher intraglomerular pressure and negatively charged capillary which force the protein across the glomerular capillary wall. An example is in lupus nephritis [13][14]. The second hypothesis involves the localization of the circulating antigens in the subepithelial aspect of the glomerular basement membrane, as such, the antigen forms in-situ immune complex deposit with the antibodies. This is seen in hepatitis B [15][16], hepatitis C [17][18], Helicobacter pylori [19], and patients diagnosed with MGN [20][21]. The third mechanism focuses on the binding of autoantibodies to antigens, as well as to podocyte membrane, leading to immune complex deposition. Numerous studies were conducted to describe the pathogenesis of MGN, ranging from the Heymann rat model of 1959 where membranous glomerulonephritis was induced using Lewis rats with crude kidney extract [22]. The use of megalin in the 1980s by Kerjaschki and Farquhar showed oxygen radicals responsible for glomerular damage resulting in proteinuria [23]. No anti-megalin antibody was recorded to have been found in patients with MGN. Therefore, megalin cannot be associated with proteinuria in human [12][24].3. The Biomarkers
Neutral endopeptidase (NEP) is the first human biomarker for MGN identified in early 2000s and is seen in patients with alloimmune neonatal MGN. This involves vertical transmission from a mother to her offspring following sensitization during previous pregnancy [25]. Therefore, it is very important to screen families with a history of membranous nephropathy as part of their antenatal care [25][26]. Beck et al. (2009) conducted a breakthrough study where a M-type phospholipase A2 receptor (PLA2R) was detected as a biomarker for MGN using Western blot technique. More recently, another biomarker, thrombospondin domain containing 7A (THSD7A) was discovered to complement PLA2R. This biomarker is detected in seronegative anti-PLA2R primary MGN patients except for some rare conditions where anti-PLA2R and anti-THSD7A can be detected [27][28]. Anti-THSD7A cannot be detected in a normal individual or patients with secondary MGN [29][30]. Anti-THSD7A occur in 2.5–5% of patients with idiopathic MGN and does not appear in secondary MGN. Except for few where both PLA2R and THSD7A were found positive, THSD7A is only detected among those MGN patients who are PLA2R negative [31][32].3.1. Clinical Feature
3.2. PLA2R-Related Sarcoidosis and Hepatitis B Virus (HBV) Infection
3.3. THSD7A and Malignancy
3.4. Role of Biomarkers in Kidney Transplant
References
- Larsen, C.P.; Messias, N.C.; Silva, F.G.; Messias, E.; Walker, P.D. Determination of primary versus secondary membranous glomerulopathy utilizing phospholipase A2 receptor staining in renal biopsies. Modern Pathol. 2012, 26, 709–715.
- Fogo, A.B.; Lusco, M.A.; Najafian, B. AJKD atlas of renal pathology: Membranous nephropathy. Nephrol. Dial. Transplant. 2011, 26, 3425–3426.
- Salant, D.J. Membranous nephropathy. Port. J. Nephrol. Hypert. 2015, 33, 239–251.
- Cattran, D.C.; Brenchley, P.E. Membranous nephropathy: Integrating basic science into improved clinical management. Kidney Int. 2017, 566–574.
- Cattran, D.; Brenchley, P. Membranous nephropathy: Thinking through the therapeutic options. Nephrol. Dial. Transpl. 2017, 32, i22–i29.
- Seitz-Polski, B.; Dolla, G.; Payre, C.; Girard, C.A.; Polidori, J.; Zorzi, K.; Birgy-Barelli, E.; Jullien, P.; Courivaud, C.; Krummel, T.; et al. Epitope Spreading of Autoantibody Response to PLA2R Associates with Poor Prognosis in Membranous Nephropathy. J. Am. Soc. Nephrol. 2016, 27, 1517–1533.
- Feng, Z.; Wang, S.; Huang, Y.; Liang, X.; Shi, W.; Zhang, B. A follow-up analysis of positron emission tomography/computed tomography in detecting hidden malignancies at the time of diagnosis of membranous nephropathy. Oncotarget 2016, 7, 9645.
- Zeng, C.H.; Chen, H.M.; Wang, R.S.; Chen, Y.; Zhang, S.H.; Liu, L.; Li, L.S.; Liu, Z.H. Etiology and Clinical Characteristics of Membranous Nephropathy in Chinese Patients. Am. J. Kidney Dis. 2008, 52, 691–698.
- Lefaucheur, C.; Stengel, B.; Nochy, D.; Martel, P.; Hill, G.S.; Jacquot, C.; Rossert, J. Membranous nephropathy and cancer: Epidemiologic evidence and determinants of high-risk cancer association. Kidney Int. 2006, 70, 1510–1517.
- KDIGO Clinical Practice Guideline for Glomerulonephritis. Available online: (accessed on 28 March 2014).
- Mastroianni-Kirsztajn, G.; Hornig, N.; Schlumberger, W. Autoantibodies in renal diseases-clinical significance and recent developments in serological detection. Front. Immunol. 2015, 6, 1–6.
- Ling, W.; Hao, T.; Chen, P.M.; Chan, C.K.; Chiang, W.C.; Chen, Y.M.; Wu, K.D. Science Direct Membranous nephropathy: A review on the pathogenesis, diagnosis, and treatment. J. Formos. Med. Assoc. 2015, 114, 102–111.
- Shlomchik, M.J.; Madaio, M.P. The role of antibodies and B cells in the pathogenesis of lupus nephritis. Springer Semin. Immunopathol. 2003, 24, 363–375.
- Wilcox, T.; Hirshkowitz, A. The role of B cells in Lupus pathogenesis. Int. J. Biochem. Cell Biol. 2010, 42, 543–550.
- Bhimma, R.; Coovadia, H.M. Hepatitis B virus-associated nephropathy. Am. J. Nephrol. 2004, 24, 198–211.
- Moon, J.Y.; Lee, S.H. Treatment of Hepatitis B Virus-Associated Membranous Nephropathy: Lamivudine Era versus Post-Lamivudine Era. Korean J. Intern. Med. 2012, 27, 394–396.
- Sandri, A.M.; Elewa, U.; Poterucha, J.J.; Fervenza, F.C. Treatment of hepatitis C-mediated glomerular disease. Nephron. Clin. Pract. 2011, 119, 121–130.
- Morales, J.M.; Kamar, N.; Rostaing, L. Hepatitis C and renal disease: Epidemiology, diagnosis, pathogenesis. Hepat. C Ren. Dis. Hemodial. Transplant. 2012, 176, 10–23.
- Dede, F.; Ayli, D.; Gonul, I.; Yuksel, O.; Ozturk, R.; Yildiz, A.; Yenigun, E.; Piskinpasa, S.; Turgut, D.; Koc, E.; et al. The effect of Helicobacter pylori eradication on proteinuria in patients with primary glomerulonephritis. Arch. Med. Sci. 2015, 11, 764–769.
- Lien, Y.H. Pathogenesis, diagnosis and management of paraneoplastic glomerulonephritis. Nat. Rev. Nephrol. 2011, 7, 85–95.
- Beck, L.H.; Salant, D.J. Membranous nephropathy: Recent travels and new roads ahead. Kidney Int. 2010, 77, 765–770.
- Heymann, W.; Hackel, D.B.; Harwood, S.; Wilson, S.G. Production of nephrotic syndrome in rats by Freund’s adjuvants and rat kidney suspensions. Proc. Soc. Exp. Biol. Med. 1959, 100, 660–664.
- Farquhar, M.G.; Saito, A.; Kerjaschki, D. The Heymann nephritis antigenic complex: Megalin (gp330) and RAP. J. Am. Soc. Nephrol. 1995, 6, 35–47.
- Ronco, P.; Debiec, H. Pathophysiological advances in membranous nephropathy: Time for a shift in patient’s care. Lancet 2015, 385, 1983–1992.
- Hu, P.; Xuan, Q.; Hu, B.; Lu, L.; Qin, Y.H. Anti-neutral endopeptidase natriuretic peptides, disarrangement and proteinuria onset in membranous nephropathy. Mol. Biol. Rep. 2013, 40, 2963–2967.
- Debiec, H.; Nauta, J.; Coulet, F.; van der, B.M.; Guigonis, V.; Schurmans, T. Role of truncating mutations in MME gene in fetomaternal alloimmunisation and antenatal glomerulopathies. Lancet 2004, 364, 1252–1259.
- Larsen, C.P.; Cossey, L.N.; Beck, L.H. THSD7A staining of membranous glomerulopathy in clinical practice reveals cases with dual autoantibody positivity. Mod. Pathol. 2016, 29, 421–426.
- Gödel, M.; Grahammer, F.; Huber, T.B. Thrombospondin Type-1 Domain-Containing 7A in Idiopathic Membranous Nephropathy. N. Engl. J. Med. 2015, 372, 1073–1075.
- Hofstra, J.M.; Beck, L.H.; Beck, D.M.; Wetzels, J.F.; Salant, D.J. Anti-phospholipase a2 receptor antibodies correlatewith clinical status in idiopathic membranous nephropathy. Clin. J. Am. Soc. Nephrol. 2011, 6, 1286–1291.
- Prunotto, M.; Carnevali, M.L.; Candiano, G.; Murtas, C.; Bruschi, M.; Corradini, E.; Trivelli, A.; Magnasco, A.; Petretto, A.; Santucci, L.; et al. Autoimmunity in membranous nephropathy targets aldose reductase and SOD2. J. Am. Soc. Nephrol. 2010, 21, 507–519.
- Liu, L.Y.; Lin, M.; Lai, Z.; Jiang, J.; Huang, Y.; Jao, L. Motor neuron-derived Thsd7a is essential for zebrafish vascular development via the Notch-dll4 signaling pathway. J. Biomed. Sci. 2016, 23, 59.
- Novel Nephrological Markers: Anti-PLA2R, anti-THSD7A and Uromodulin. Available online: (accessed on June 2016).
- Kemp, W.L.; Burns, D.K.; Brown, T.G. Pathology of the Kidney and Bladder. In Pathology: The Big Picture; McGraw-Hill Medical: New York, NY, USA, 2008.
- Thompson, A.; Cattran, D.C.; Blank, M. Complete and partial remission as surrogate end points in membranous nephropathy. J. Am. Soc. Nephrol. 2015, 2930–2937.
- Barbour, S.; Reich, H. Short-term complication of membranous nephropathy. Contrib. Nephrol. 2013, 143–151.
- Ponticelli, C.; Passerini, P. A randomized pilot trial comparing methylprednisolone plus a cytotoxic agent versus synthetic adrenocorticotropic hormone in idiopathic membranous nephropathy. Am. J. Kidney 2006, 47, 233–240.
- Nachman, P.H.; Jennette, J.C. Primary glomerular disease. In Brenner and Rector’s the Kidney, 9th ed.; Elsevier: London, UK, 2012; pp. 1100–1191.
- 5 rd Report of the Malaysian Registry of Renal Biopsy. Available online: (accessed on June 2012).
- Ren, S.; Wu, C.; Zhang, Y.; Wang, A.Y.; Li, G.; Wang, L.; Hong, D. An update on clinical significance of use of THSD7A in diagnosing idiopathic membranous nephropathy: A systematic review and meta-analysis of THSD7A in IMN. Ren. Fail. 2018, 40, 306–313.
- Guerry, M.J.; Vanhille, P.; Ronco, P. Serum anti-PLA2R antibodies may be present before clinical manifestations of membranous nephropathy. Kidney Int. 2016, 89, 1399.
- Stanescu, H.C.; Arcos-Burgos, M.; Medlar, A.; Bockenhauer, D.; Kottgen, A.; Ragomirescu, L. Risk HLA-DQA1 and PLA2R1 alleles in idiopathic membranous nephropathy. N. Engl. J. Med. 2011, 364, 616–626.
- Hladunewich, M.A.; Troyanov, S.; Calafati, J. The natural history of the non-nephrotic membranous nephropathy patient. Clin. J. Am. Soc. Nephrol. 2009, 4, 1417–1422.
- Stehlé, T.; Joly, D.; Vanhille, P.; Boffa, J.J.; Rémy, P.; Mesnard, L.; Hoffmann, M.; Grimbert, P.; Choukroun, G.; Vrtovsnik, F.; et al. Clinicopathological study of glomerular diseases associated with sarcoidosis: A multicenter study. Orphanet. J. Rare. Dis. 2013, 8, 65.
- Stehlé, T.; Audard, V.; Ronco, P.; Debiec, H. Phospholipase A2 receptor and sarcoidosis-associated membranous nephropathy. Nephrol. Dial. Transplant. 2015, 30, 1047–1050.
- Iannuzzi, M.C.; Rybicki, B.A. Sarcoidosis. N. Engl. J. Med. 2007, 357, 2153–2165.
- Debiec, H.; Ronco, P. Immunopathogenesis of membranous nephropathy: An update. Semin. Immunopathol. 2014, 36, 381–397.
- Xie, Q.; Li, Y.; Xue, J.; Xiong, Z.; Wang, L.; Sun, Z.; Ren, Y.; Zhu, X.; Hao, C.M. Renal Phospholipase A2 Receptor in Hepatitis B Virus-Associated Membranous Nephropathy. Am. J. Nephrol. 2015, 41, 345–353.
- Sasaki, Y.; Nagai, Y.; Mikami, T.; Akasaka, Y.; Shibuya, K.; Urita, Y. Anti-phospholipase A2 receptor antibody positive hepatitis B virus-associated membranous nephropathy remitted with entecavir after relapse with lamivudine. J. Nephropathol. 2018, 7, 93–97.
- Xu, X.; Zhu, X.; Yuan, S.; Jiang, W.; Xia, Y.; Liu, H.; Li, J.; Sun, L.; Peng, Y.; Liu, F. Role of M-Type phospholipase A2 receptor and its antibody in hepatitis B virus-Associated membranous nephropathy. J. Cent. South. Univ. Med. Sci. 2016, 41, 1064–1068.
- Tomas, N.M. Autoantibodies against thrombospondin type 1 domain–containing 7A induce membranous nephropathy. J. Clin. Investig. 2016, 126, 2519–2532.
- Hoxha, E.; Wiech, T.; Stahl, P.R. A mechanism for cancer-associated membranous nephropathy. N. Engl. J. Med. 2016, 374, 1995–1996.
- Hoxha, E.; Beck, L.H.; Wiech, T. An indirect immunofluorescence method facilitates detection of thrombospondin type 1 domain-containing 7A-specific antibodies in membranous nephropathy. J. Am. Soc. Nephrol. 2016, 28, 520–531.
- Xian, L.; Dong, D.; Luo, J.; Zhuo, L.; Li, K.; Zhang, P.; Wang, W.; Xu, Y.; Xu, G.; Wang, L.; et al. Expression of THSD7A in neoplasm tissues and its relationship with proteinuria. BMC Nephrol. 2019, 20, 1–6.
- Quintana, L.F.; Miguel, B.; Miguel, S.; Perez, N.S.; Lopez-Hoyos, M.; Patricia, V.; Emillio, R.; Odette, V.; Guadalupe, E.; Fritz, D.; et al. Antiphospholipase A2 antibody predict the risk of post transplantation recurrence of membranous nephropathy. Transplantation 2015, 99, 1709–1714.
- Gupta, G.; Fattah, H.; Ayalon, R.; Kidd, J.; Gehr, T.; Quintana, L.F. Pretransplant phospholipase A2 receptor autoantibody concentration is associated with clinically significant recurrence of membranous nephropathy post-kidney transplantation. Clin. Transpl. 2016, 30, 461–469.
- Leon, J.; Pérez-Sáez, M.J.; Batal, I.; Beck, L.H.; Rennke, H.G.; Canaud, G.; Legendre, C.; Pascual, J.; Riella, L.V. Membranous Nephropathy Post-Transplantation. Transplantation 2019, 103, 1.
- Debiec, H.; Lefeu, F.; Kemper, M.J.; Niaudet, P.; Deschênes, G.; Remuzzi, G.; Ulinski, T.; Ronco, P. Early-childhood membranous nephropathy due to cationic bovine serum albumin. N. Engl. J. Med. 2011, 364, 2101–2110.
- Tomas, N.M.; Beck, L.H.; Meyer-Schwesinger, C.; Seitz-Polski, B.; Ma, H.; Zahner, G.; Dolla, G.; Hoxha, E.; Helmchen, U.; Dabert-Gay, A.-S.; et al. Thrombospondin Type-1 Domain-Containing 7A in Idiopathic Membranous Nephropathy. N. Engl. J. Med. 2014, 371, 2277–2287.
- Wai, H.; Lim, M.S.G.W. Recurrent and de novo glomerulonephritis after kidney transplantation. Core Concepts Ren. Transplant. 2019, 10, 211–231.
