The thymus gland is a primary lymphoid organ for T-cell development. T-cells are one of the white blood cells that play a critical role in adaptive immune response. Various viral infections can result in the disturbance of thymic functions.
- viruses
- enterovirus
- Coxsackievirus B
- thymus
- self-tolerance
- autoimmunity
- type diabetes
- fetal and perinatal life
1. Overview
The thymus gland is a primary lymphoid organ for T-cell development. Various viral infections can result in disturbance of thymic functions. Medullary thymic epithelial cells (mTECs) are important for the negative selection of self-reactive T-cells to ensure central tolerance. Insulin-like growth factor 2 (IGF2) is the dominant self-peptide of the insulin family expressed in mTECs and plays a crucial role in the intra-thymic programing of central tolerance to insulin-secreting islet β-cells. Coxsackievirus B4 (CVB4) can infect and persist in the thymus of humans and mice, thus hampering the T-cell maturation and differentiation process. The modulation of IGF2 expression and protein synthesis during a CVB4 infection has been observed in vitro and in vivo in mouse models. The effect of CVB4 infections on human and mouse fetal thymus has been studied in vitro. Moreover, following the inoculation of CVB4 in pregnant mice, the thymic function in the fetus and offspring was disturbed. A defect in the intra-thymic expression of self-peptides by mTECs may be triggered by CVB4. The effects of viral infections, especially CVB4 infection, on thymic cells and functions and their possible role in the pathogenesis of type 1 diabetes (T1D) are presented.
2. Enteroviruses
3. Viral Infections and the Thymus Gland
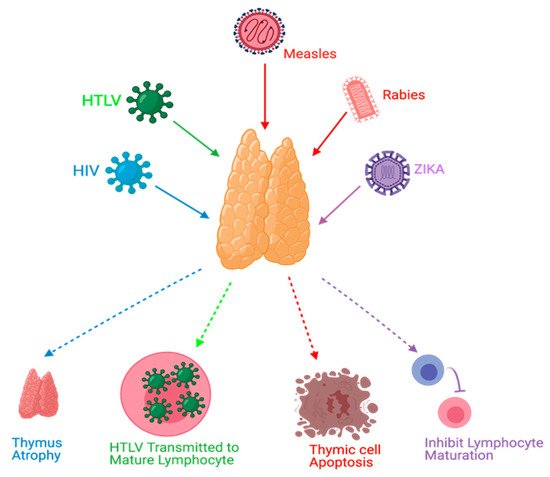
4. Conclusions
References
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV Virus Taxonomy Profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422.
- Tapparel, C.; Siegrist, F.; Petty, T.J.; Kaiser, L. Picornavirus and enterovirus diversity with associated human diseases. Infect. Genet. Evol. 2013, 14, 282–293.
- Sawyer, M.H. Enterovirus infections: Diagnosis and treatment. Semin. Pediatr. Infect. Dis. 2002, 13, 40–47.
- Alhazmi, A.; Sane, F.; Lazrek, M.; Nekoua, M.P.; Badia-Boungou, F.; Engelmann, I.; Alidjinou, E.K.; Hober, D. Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions. Microorganisms 2020, 8, 1458.
- Stewart, T.A.; Hultgren, B.; Huang, X.; Pitts-Meek, S.; Hully, J.; MacLachlan, N.J. Induction of type I diabetes by interferon-alpha in transgenic mice. Science 1993, 260, 1942–1946.
- Horwitz, M.S.; Bradley, L.M.; Harbertson, J.; Krahl, T.; Lee, J.; Sarvetnick, N. Diabetes induced by Coxsackie virus: Initiation by bystander damage and not molecular mimicry. Nat. Med. 1998, 4, 781–785.
- Filippi, C.; von Herrath, M. How viral infections affect the autoimmune process leading to type 1 diabetes. Cell. Immunol. 2005, 233, 125–132.
- Von Herrath, M.G.; Fujinami, R.S.; Whitton, J.L. Microorganisms and autoimmunity: Making the barren field fertile? Nat. Rev. Microbiol. 2003, 1, 151–157.
- Albano, F.; Vecchio, E.; Renna, M.; Iaccino, E.; Mimmi, S.; Caiazza, C.; Arcucci, A.; Avagliano, A.; Pagliara, V.; Donato, G.; et al. Insights into Thymus Development and Viral Thymic Infections. Viruses 2019, 11, 836.
- Alidjinou, E.K.; Sané, F.; Engelmann, I.; Geenen, V.; Hober, D. Enterovirus persistence as a mechanism in the pathogenesis of type 1 diabetes. Discov. Med. 2014, 18, 273–282.
- Tracy, S.; Smithee, S.; Alhazmi, A.; Chapman, N. Coxsackievirus can persist in murine pancreas by deletion of 5′ terminal genomic sequences. J. Med. Virol. 2014, 87, 240–247.
- Jaïdane, H.; Halouani, A.; Jmii, H.; Elmastour, F.; Abdelkefi, S.; Bodart, G.; Michaux, H.; Chakroun, T.; Sane, F.; Mokni, M.; et al. In-utero coxsackievirus B4 infection of the mouse thymus. Clin. Exp. Immunol. 2017, 187, 399–407.
- Ferrando-Martinez, S.; De Pablo-Bernal, R.S.; De Luna-Romero, M.; De Ory, S.J.; Genebat, M.; Pacheco, Y.M.; Parras, F.J.; Montero, M.; Blanco, J.R.; Gutierrez, F.; et al. Thymic Function Failure Is Associated With Human Immunodeficiency Virus Disease Progression. Clin. Infect. Dis. 2017, 64, 1191–1197.
- Rosado-Sánchez, I.; Herrero-Fernández, I.; Genebat, M.; Ruiz-Mateos, E.; Leal, M.; Pacheco, Y.M. Thymic Function Impacts the Peripheral CD4/CD8 Ratio of HIV-Infected Subjects. Clin. Infect. Dis. 2017, 64, 152–158.
- Fiume, G.; Scialdone, A.; Albano, F.; Rossi, A.; Tuccillo, F.M.; Rea, D.; Palmieri, C.; Caiazzo, E.; Cicala, C.; Bellevicine, C.; et al. Impairment of T cell development and acute inflammatory response in HIV-1 Tat transgenic mice. Sci. Rep. 2015, 5, 13864.
- Hartling, H.J.; Gaardbo, J.C.; Ronit, A.; Salem, M.; Laye, M.; Clausen, M.R.; Skogstrand, K.; Gerstoft, J.; Ullum, H.; Nielsen, S.D. Impaired thymic output in patients with chronic hepatitis C virus infection. Scand. J. Immunol. 2013, 78, 378–386.
- Ye, P.; Kirschner, D.E.; Kourtis, A.P. The thymus during HIV disease: Role in pathogenesis and in immune recovery. Curr. HIV Res. 2004, 2, 177–183.
- De Voeght, A.; Martens, H.; Renard, C.; Vaira, D.; Debruche, M.; Simonet, J.; Geenen, V.; Moutschen, M.; Darcis, G. Exploring the link between innate immune activation and thymic function by measuring sCD14 and TRECs in HIV patients living in Belgium. PLoS ONE 2017, 12, e0185761.
- Rb-Silva, R.; Nobrega, C.; Azevedo, C.; Athayde, E.; Canto-Gomes, J.; Ferreira, I.; Cheynier, R.; Yates, A.J.; Horta, A.; Correia-Neves, M. Thymic Function as a Predictor of Immune Recovery in Chronically HIV-Infected Patients Initiating Antiretroviral Therapy. Front. Immunol. 2019, 10, 25.
- Carvalho Barros, L.R.; Linhares-Lacerda, L.; Moreira-Ramos, K.; Ribeiro-Alves, M.; Machado Motta, M.C.; Bou-Habib, D.C.; Savino, W. HTLV-1-infected thymic epithelial cells convey the virus to CD4. Immunobiology 2017, 222, 1053–1063.
- Cardenas Palomo, L.F.; de Souza Matos, D.C.; Chaves Leal, E.; Bertho, A.L.; Marcovistz, R. Lymphocyte subsets and cell proliferation analysis in rabies-infected mice. J. Clin. Lab. Immunol. 1995, 46, 49–61.
- Marcovistz, R.; Bertho, A.L.; Matos, D.C. Relationship between apoptosis and thymocyte depletion in rabies-infected mice. Braz. J. Med. Biol. Res. 1994, 27, 1599–1603.
- Wiktor, T.J.; Doherty, P.C.; Koprowski, H. Suppression of cell-mediated immunity by street rabies virus. J. Exp. Med. 1977, 145, 1617–1622.
- Hirai, K.; Kawano, H.; Mifune, K.; Fujii, H.; Nishizono, A.; Shichijo, A.; Mannen, K. Suppression of cell-mediated immunity by street rabies virus infection. Microbiol. Immunol. 1992, 36, 1277–1290.
- Kasempimolporn, S.; Saengseesom, W.; Mitmoonpitak, C.; Akesowan, S.; Sitprija, V. Cell-mediated immunosuppression in mice by street rabies virus not restored by calcium ionophore or PMA. Asian Pac. J. Allergy Immunol. 1997, 15, 127–132.
- Godfraind, C.; Holmes, K.V.; Coutelier, J.P. Thymus involution induced by mouse hepatitis virus A59 in BALB/c mice. J. Virol. 1995, 69, 6541–6547.
- Auwaerter, P.G.; Kaneshima, H.; McCune, J.M.; Wiegand, G.; Griffin, D.E. Measles virus infection of thymic epithelium in the SCID-hu mouse leads to thymocyte apoptosis. J. Virol. 1996, 70, 3734–3740.
- Valentin, H.; Azocar, O.; Horvat, B.; Williems, R.; Garrone, R.; Evlashev, A.; Toribio, M.L.; Rabourdin-Combe, C. Measles virus infection induces terminal differentiation of human thymic epithelial cells. J. Virol. 1999, 73, 2212–2221.
- Yamanouchi, K.; Chino, F.; Kobune, F.; Kodama, H.; Tsuruhara, T. Growth of measles virus in the lymphoid tissues of monkeys. J. Infect. Dis. 1973, 128, 795–799.
- Moench, T.R.; Griffin, D.E.; Obriecht, C.R.; Vaisberg, A.J.; Johnson, R.T. Acute measles in patients with and without neurological involvement: Distribution of measles virus antigen and RNA. J. Infect. Dis. 1988, 158, 433–442.
- Gibb, T.R.; Bray, M.; Geisbert, T.W.; Steele, K.E.; Kell, W.M.; Davis, K.J.; Jaax, N.K. Pathogenesis of experimental Ebola Zaire virus infection in BALB/c mice. J. Comp. Pathol. 2001, 125, 233–242.
- Mocarski, E.S.; Bonyhadi, M.; Salimi, S.; McCune, J.M.; Kaneshima, H. Human cytomegalovirus in a SCID-hu mouse: Thymic epithelial cells are prominent targets of viral replication. Proc. Natl. Acad. Sci. USA 1993, 90, 104–108.
- Messias, C.V.; Loss-Morais, G.; Carvalho, J.B.; González, M.N.; Cunha, D.P.; Vasconcelos, Z.; Arge, L.W.P.; Farias-de-Oliveira, D.A.; Gerber, A.L.; Portari, E.A.; et al. Zika virus targets the human thymic epithelium. Sci. Rep. 2020, 10, 1378.
- Gul, K.A.; Sonerud, T.; Fjærli, H.O.; Nakstad, B.; Abrahamsen, T.G.; Inchley, C.S. Thymus activity measured by T-cell receptor excision circles in patients with different severities of respiratory syncytial virus infection. BMC Infect. Dis. 2017, 17, 18.
