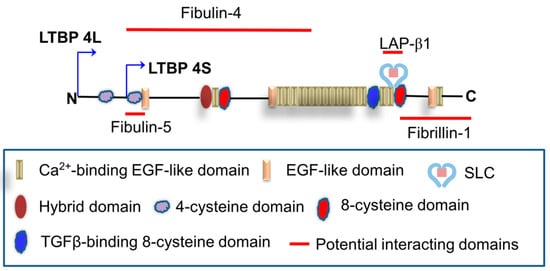
LTBP–TGFβ complexes are required for LTBP to incorporate into the ECM. If the TGFβ–LLC complex does not incorporate into the ECM, the levels of active TGFβ may be reduced. LTBP4 has been reported to enable latent TGFβ secretion and regulate latent TGFβ activation
[12]. TGFβ is a potent pleiotropic cytokine, the synthesis and activation of which are associated with LTBPs. Sequestration of the latent form of the TGFβ complex in the ECM is important for proper TGFβ activation. Although incorporated into the matrix in the form of LLC, TGFβ remains inert. Activation of latent TGFβ in different tissues is initiated by several biological processes, such as hypoxia and stretch, dependent on TGFβ signaling, thereby leading to the release of TGFβ from the complex to bind to its specific TGFβ receptors.
Once TGFβ is activated from its propeptide dimer form, it binds to a heteromeric transmembrane TGFβ receptor complex, formed by the dimeric TGFβ1 receptor Alk5 (also known as TβRI) and the dimeric TGFβ type II receptor
[14][15][14,15]. For downstream signaling, the canonical TGFβ family ligand signaling begins with phosphorylation of TβRI on serine residues of human receptor-regulated/activated (R-) Smad proteins. Smad1, Smad5, and Smad8 are phosphorylated by Alk1- and Alk2-containing receptors, whereas Smad2 and Smad3 are phosphorylated by Alk5/TβRI in the TGFβ receptor. In contrast, the two inhibitory (I-) Smads, Smad6 and Smad7, provide negative feedback control of canonical signaling
[16]. Inhibitory Smad7 antagonizes TGFβ signaling by inhibiting R-Smad binding to TβRI
[17]. Moreover, the activated Smad2/3 complex cooperates with co-Smad4 and translocates into the nucleus, leading to increased gene expression regulated by co-activators and cell-specific transcription factors. In addition to canonical Smad signaling, TGFβ can activate various other pathways, including JNK, p38, ERK, MAPK, and Rho/ROCK, to influence multiple cellular signaling systems such as the actin cytoskeleton, tight-junction strands, and transcriptional regulation
[18]. TGFβ signaling is part of a complex network of intracellular signaling, with crucial connection points in development, immunity, and malignancy
[19]. Smad complexes bind DNA with low affinity and act in connection with other transcription factors that define binding sites and repressor/activator activities
[20][21][22][20,21,22].
Because of its involvement in context-dependent signaling pathways, TGFβ1 can act in opposing manners, which includes its impact on different cell types even in similar environments. Such context-dependent signaling (i.e., dependent on the signaling state of the target cell) is a factor affecting the opposing effects of TGFβ1 on different cells in the same environment
[17]. The same cancer cells inhibited by TGFβ1 to reduce their growth and metastatic potential in the early phases of tumor development progressively escape this suppressive effect and use growth factors to drive metastasis
[23].