Borrelia miyamotoi is a Gram-negative bacteria included in the genus Borrelia from the family Spirochaetaceae, within the phylum Spirochaetes and the order Spirochaetales. B. miyamotoi is spirochete from the relapsing fever (RF) group of Borrelia. In RF-Borrelia complex only B. miyamotoi is transmitted by Ixodes ticks - a vector of B. burgdorferi s.l. complex. The biological feature that distinguishes B. miyamotoi from B. burgdorferi s.l. is transovarial transmission. B. miyamotoi was first isolated from questing I. persulcatus ticks and mouse Apodemus argentus in Japan in 1994. The main vector of B. miyamotoi is I. persulcatus (Asia), I. pacificus, I. scapularis (North America), and I. ricinus (Europe). Worldwide, B. miyamotoi prevalence in questing Ixodes ticks ranges from 0.2 to 10%. A phylogenetic analysis based on selected sequences of B. miyamotoi genome revealed genetic differences between isolates from Asia, North America, and Europe, which are clearly separated into three genotypes. Human symptomatic cases of Borrelia miyamotoi disease (BMD) were first reported in 2011 in Russia and then in North America, Europe, and Asia. BMD is usually manifested by several episodes of fever and flu-like symptoms (chills, headaches, muscle, and joint aches and general fatigue). However, serious symptoms such as meningoencephalitis can be observed.
1. Introduction
In Europe, tick-borne diseases transmitted by
Ixodes ricinus are the most common zoonoses with significant medical and veterinary importance[1]. This hematophagous arthropod is a reservoir and vector of many pathogenic microorganisms, including the bacteria
are the most common zoonoses with significant medical and veterinary importance [1]. This hematophagous arthropod is a reservoir and vector of many pathogenic microorganisms, including the bacteria Borrelia burgdorferi
sensu lato (s.l.) complex—the causative agent of Lyme borreliosis (LB),
Rickettsia
spp., and
Anaplasma spp., as well as the flavivirus responsible for tick-borne encephalitis (TBE) and the etiological protozoan agents of babesiosis[2][3]. With advanced methods of molecular biology, new tick-borne microorganism species and their genetic variants with confirmed or potential pathogenicity for humans and animals are still being identified[4]. One of the emerging
spp., as well as the flavivirus responsible for tick-borne encephalitis (TBE) and the etiological protozoan agents of babesiosis [2,3]. With advanced methods of molecular biology, new tick-borne microorganism species and their genetic variants with confirmed or potential pathogenicity for humans and animals are still being identified [4]. One of the emerging Ixodes
-borne diseases in the northern temperate climate zones of the world, including Europe, is
Borrelia miyamotoi
disease (BMD), caused by spirochete from the relapsing fever (RF) group of
Borrelia[5][6]. Since 1994, when
B. miyamotoi
was first isolated from questing
I. presulcatus
ticks and mouse
Apodemus argentus in Japan[7], it was considered to be a non-pathogenic endosymbiont. However, since 2011 many symptomatic
in Japan [7], it was considered to be a non-pathogenic endosymbiont. However, since 2011 many symptomatic B. miyamotoi infections in humans have been noted in Asia, North America, and Europe [8][9][10][11][12][13][14].
infections in humans have been noted in Asia, North America, and Europe [8,9,10,11,12,13,14].
2. Recent Studies
2.1. Taxonomic Position
B. miyamotoi
is a Gram-negative bacteria included in the genus
Borrelia
from the family
Spirochaetaceae
, within the phylum
Spirochaetes
and the order
Spirochaetales
Borrelia
species are obligate parasites, transmitted by arthropod vectors to vertebrate hosts. The biological feature that distinguishes
B. miyamotoi
and several other relapsing fever species from
B. burgdorferi s.l. is transovarial transmission[16].
s.l. is transovarial transmission [16].
The
Borrelia spirochete cells are 0.2–0.5 mm in diameter by 3–30 mm in length, with 15–20 periplasmic flagella (endoflagella) located in the periplasmic space between the outer membrane and the protoplasmic cylinder. These cells can move actively with frequent reversal of direction [15][17]. Due to limited
spirochete cells are 0.2–0.5 mm in diameter by 3–30 mm in length, with 15–20 periplasmic flagella (endoflagella) located in the periplasmic space between the outer membrane and the protoplasmic cylinder. These cells can move actively with frequent reversal of direction [15,17]. Due to limited B. miyamotoi
biosynthetic potential, its in vitro culture is difficult (as other
Borrelia
species) and requires microaerophilic conditions and complex nutrition. However, it can be propagated in Kelly-Pettenkofer medium with fetal calf serum (MKP-F) [18].
Although the
Borrelia
species share spirochetal morphology, they have different biological, clinical, and epidemiological features. Based on their arthropod vectors and genetic characteristics two major groups of
Borrelia
were distinguished. The first group contains 20
Borrelia
species, including the
B. burgdorferi
s.l. complex, an agent of LB, and are transmitted by
Ixodes
hard ticks. The second group includes 25
Borrelia
species associated with human RF and mostly found in soft ticks (
Argasidae
) but also in lice (
B. recurensis
) and hard ticks (
B. miyamotoi
,
B. lonestari
,
B. theileri
). In RF-
Borrelia
complex only
B. miyamotoi
is transmitted by
Ixodes
ticks—a vector of
B. burgdorferi s.l. complex [15][19][20]. These two groups are genetically similar but form distinct, independent monophyletic clades and share a common ancestor. In 2014, Adeolu and Gupta[21] proposed splitting the spirochetes from the genus
s.l. complex [15,19,20]. These two groups are genetically similar but form distinct, independent monophyletic clades and share a common ancestor. In 2014, Adeolu and Gupta [21] proposed splitting the spirochetes from the genus Borrelia
into two separate genera: a novel genus,
Borreliella
gen. nov., containing the causative agents of Lyme disease and a revised genus
Borrelia
, with spirochetes causing RF, including
B. miyamotoi. However, the proposed change in the name of this pathogenic bacteria species proved controversial and did not receive support among scientists, clinicians or public health authorities, who felt it would lead to confusion and pose a risk to patient safety [20][22][23].
. However, the proposed change in the name of this pathogenic bacteria species proved controversial and did not receive support among scientists, clinicians or public health authorities, who felt it would lead to confusion and pose a risk to patient safety [20,22,23].
2.2. Genome Organization and Genetic Diversity
The first information about the organization of the
B. miyamotoi
genome and its differences in relation to the known species from the LB- and RF-
Borrelia groups was published in 1995[7]. Later, more advanced molecular analysis of Asian, American, and European
groups was published in 1995 [7]. Later, more advanced molecular analysis of Asian, American, and European B. miyamotoi
isolates from
Ixodes
ticks and clinical samples revealed the complexity of the genome structure typical of
Borrelia spirochetes[24][25][26][27][28][29]. However, the most information was obtained by sequencing the genome of
spirochetes [24,25,26,27,28,29]. However, the most information was obtained by sequencing the genome of B. miyamotoi Izh-4 isolate from a Russian patient[30]. The complete genome of a single
Izh-4 isolate from a Russian patient [30]. The complete genome of a single B. miyamotoi
cell consists of one linear chromosome (~900 kb) and 12 linear and two circular plasmids (from 6 to 73 kb). Two of the plasmids (lp70 and lp64) had not previously been found in other
Borrelia
species. A total of 1362 genes, including 1222 protein-coding genes, 103 pseudogenes, 31 genes for transfer RNA (tRNA), a cluster of three genes of ribosomal RNA (rRNA), and three genes of non-coding RNA (ncRNA) were identified. In
B. miyamotoi virulence, a significant role is played by plasmid lp4, which includes genes of variable membrane proteins (VMPs), necessary to mask the bacteria from the host immune system and prolong the infection[30][31][32]. A comparison of different
virulence, a significant role is played by plasmid lp4, which includes genes of variable membrane proteins (VMPs), necessary to mask the bacteria from the host immune system and prolong the infection [30,31,32]. A comparison of different B. miyamotoi isolates revealed that the number and order of VMPs genes were unique for each of them[30].
isolates revealed that the number and order of VMPs genes were unique for each of them [30].
Phylogenetic analysis based on genome sequences of
B. miyamotoi
showed genetic differences between isolates from Asia, North America and Europe which are clearly separated into three types (genotypes) and form a monophyletic clade inside the RF-
Borrelia spirochetes[30]. However, the genetic differences between the
spirochetes [30]. However, the genetic differences between the B. miyamotoi isolates are probably not connected with geographic origin, but rather with pathogenicity, vector competence, and host range[24].
isolates are probably not connected with geographic origin, but rather with pathogenicity, vector competence, and host range [16,24].
The
B. miyamotoi
genetic distance from other LB species and the relationship with the species from the RF group is evidenced by the carriage and expression of a
glpQ gene, coding the immunoreactive protein glycerophosphodiester phosphodiesterase [33][34]. The
gene, coding the immunoreactive protein glycerophosphodiester phosphodiesterase [33,34]. The glpQ
gene and GlpQ protein are conserved among the members of the genus
Borrelia
, except LB spirochetes (
). Therefore, GlpQ is usually used as a marker in molecular and serological tests to detect RF spirochete infections and to distinguish cases of LB and other tick-borne infections (e.g., anaplasmosis, babesiosis) [8][35][36][37].
). Therefore, GlpQ is usually used as a marker in molecular and serological tests to detect RF spirochete infections and to distinguish cases of LB and other tick-borne infections (e.g., anaplasmosis, babesiosis) [8,35,36,37].
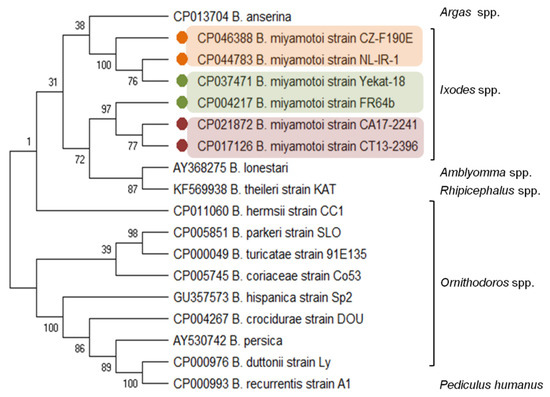
Figure 1.
Molecular relationships between
B. miyamotoi
and other RF
Borrelia
species based on the sequences of the
glpQ gene selected from GenBank. The consensus tree constructed using the neighbor-joining method and the maximum composite likelihood as the distance method; numbers at the tree nodes indicate bootstrap value from 1000 replicates; analyses were conducted in MEGA X[38]. Marks: orange—European type, green—Asian type, red—American type
gene selected from GenBank. The consensus tree constructed using the neighbor-joining method and the maximum composite likelihood as the distance method; numbers at the tree nodes indicate bootstrap value from 1000 replicates; analyses were conducted in MEGA X [38]. Marks: orange—European type, green—Asian type, red—American type of B. miyamotoi
. The genus names of the vectors were added.