The neuropeptide oxytocin is synthesized by cells in the hypothalamic paraventricular and supraoptic nuclei and transported to the posterior pituitary for release into the blood, where it is most well-known for acting on smooth muscle to stimulate uterine contractions during labor and milk-ejection from the breast. The role of the hypothalamic neuropeptide oxytocin in influencing the brain and behavior has been the subject of widespread research due, most notably, to its reported involvement in promoting social cognition and motivation, reducing anxiety, and relieving pain. It is also increasingly being considered as an important therapeutic intervention in a variety of disorders with social dysfunction as a symptom. There is increasing evidence that many of its functional effects can be peripherally mediated via increasing its concentration in the blood. This has opened up an oromucosal administration route as an alternative, which is beneficial since the oral consumption of peptides is problematic due to their rapid breakdown in the acidic environment of the gastrointestinal system.
- oromucosal administration
- oxytocin
1. Introduction
2. Oromucosal Administration of Peptides
The oral cavity is easily accessible for the administration of peptides and the main issue is to ensure that they remain in the mouth for a sufficient time to be absorbed into the blood via the mucosae. The two main pathways by which molecules diffuse passively across mucosal membranes are paracellular (passage between adjacent cells, i.e., intercellular) and transcellular (passage through cells) routes [11,20][11][12]. The paracellular diffusion route predominates for most lipophilic and hydrophilic molecules when they cross mucosal membranes with two types of pathways within the intercellular space; a hydrophobic pathway through the lipid bilayers and a parallel hydrophilic one along the narrow aqueous regions associated with lipid polar head groups. As the lipophilicity of a molecule increases, the propensity for its diffusion across the epithelial barrier also increases, although the main influence on diffusion is molecular weight, and other factors such as charge can also be important for non-polar molecules to cross the epithelial layer more effectively [11] (see Figure 1). A key advantage of the oromucosal administration of substances, as opposed to oral administation using pills, is that they can enter the blood directly and thereby reduce first-pass metabolism depletion by the liver and gut [20][12].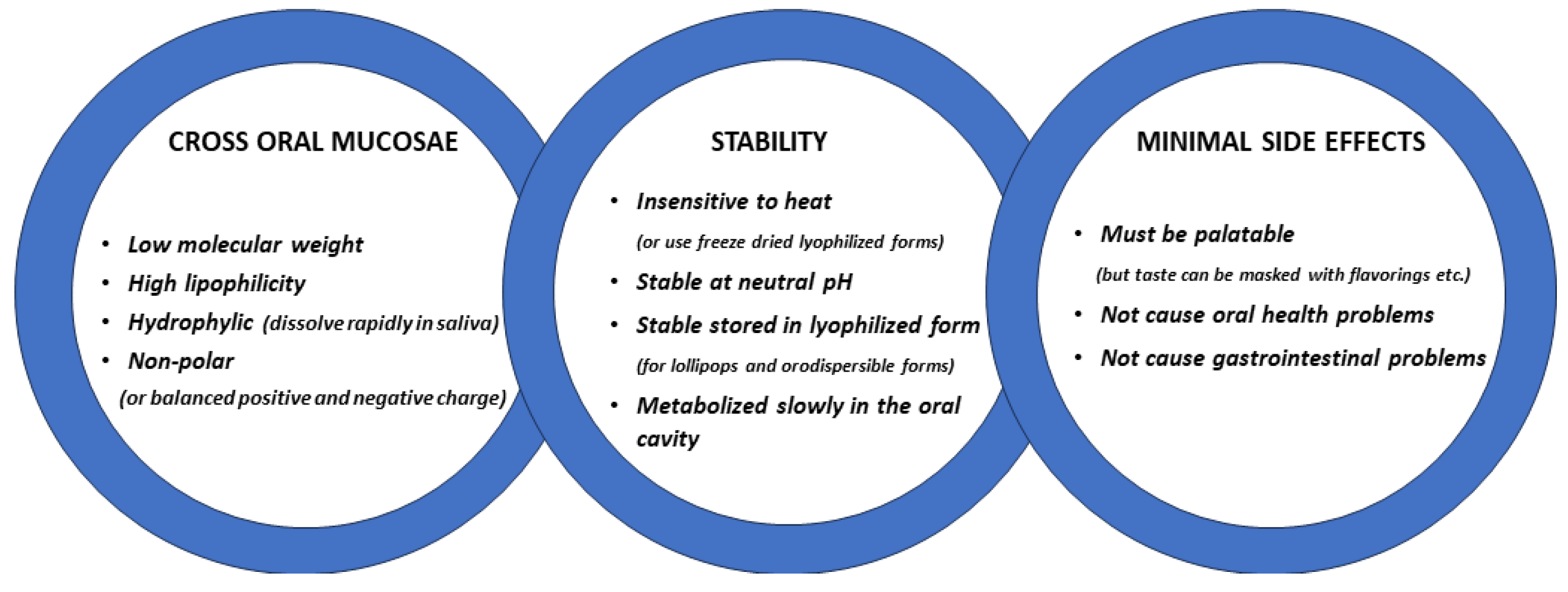
Development of Oromucosal Oxytocin Administration Using Lingual Spray and Medicated Lollipop ‘Oxipop’ Methodologies
Given that oxytocin is commercially available as an intranasal spray and licensed for use as a lactation enhancer in post-partum women (e.g., Sichuan Defeng, Pharmaceutical Co., Ltd., Chengdu, China), a first step into assessing its efficacy when given orally was to utilize a spray for administration by lingual application. The spray contains oxytocin acetate at a concentration of 4 IUs per 0.1 mL puff and is dissolved in 0.9% saline and stabilized using glycerol. Placebo sprays are identical in composition with the exception that the peptide is excluded. The same protocol was therefore adopted as for intranasal administration with 6 × 4 IU doses being self-administered under and on top of the tongue and with each spray being given at 30 s intervals to match the intranasal protocol (i.e., a total administration duration of 3 min). To maximize absorption, subjects were required to keep the spray in their mouth without swallowing until they self-administered the next dose [33,34,35][25][26][27]. Initially, in these studies, a pharmacokinetic assessment was carried out in 10 male adult male subjects with blood samples being taken at 15 min intervals before and for 105 min after the lingual administration. The absolute bioavailability was calculated using data for measurements of oxytocin concentrations in the patients’ blood following a 10 min intravenous infusion of 10 IU taken from [15][28], with repeated samples being taken over a similar time course. For lingual administration following a 24 IU dose, the AUC for the altered plasma oxytocin concentrations was significantly less than that for the intranasal administration (mean ± SD 4.79 ± 3.38 (n = 10) vs. 11.93 ± 7.68 pg/mL/h (n = 27), p = 0.008), as was the absolute bioavailability (4.45 ± 3.51% vs. 11.07 ± 7.12%, p = 0.008). The calculated bioavailability for the intranasal administration was very similar to that using data from a previous published paper where a 40 IU dose of oxytocin was used (10.59 ± 7.95%—from [15][28]). For the time to reach maximum concentrations in blood (Tmax), the lingual administration was 34.5 ± 17.4 min compared with 26.61 ± 14.34 min for the intranasal administration and did not differ significantly (p = 0.159). This obviously contrasts with profiles following intravenous infusions, where blood concentrations rise immediately and sharply [15][28]. However, lingual sprays where subjects are required to deliberately retain the liquid in their mouth for a period of time to maximize absorption are also clearly not optimal in terms of patient compliance, particularly in pediatric subjects. In recent years, medicated chocolate and candy lollipops have also been increasingly used to administer a variety of substances ranging from vitamin and mineral supplements to antibiotics and pain killers (see [36,37,38][29][30][31]). Typical lollipop formulations tend to be either hard or soft forms of sugar-free candy, with the substance for medication being incorporated into the candy during the heating stage of its formation. The candy lollipops in particular have a number of advantages for drug administration via oromucosal absorption since, during sucking, they are in constant close contact with the roof of the mouth and tongue and are pleasant to eat, especially for children. The two main disadvantages of such medicated lollipops are that, firstly, since a heating process is used in their conventional manufacture, this precludes using them for heat-sensitive drugs and, secondly, if the drug used has a strong bitter or unpalatable taste, this can also be a problem. Thus, the conventional strategy of incorporating drugs into the whole of a lollipop using a heating process is not suitable for peptides, which are typically heat-sensitive and would therefore degrade using such a strategy. To overcome this problem, the novel step was taken of freeze-drying oxytocin onto the surface of the lollipop to create ‘oxipops’ rather than trying to incorporate it into the whole lollipop. This had the dual advantage that current commercial sugar-free lollipops could be utilized and that it maximized the peptide being dissolved by the saliva as the child or adult sucked the lollipop with the whole dose being released slowly without the requirement that all of the lollipop needed to be consumed, since the oxytocin dose was only on the surface. These commercial lollipops typically use xylitol as a sweetener, which is widely accepted to be a safe form of low-calorie sweetener that does not cause insulin spikes and can actually promote oral hygiene and even immunocompetence [39][32]. Clearly, both children and adults may exhibit distinct taste preferences which could potentially influence compliance, although this can be overcome through the use of different flavor alternatives (current options have included orange, grape, pineapple, and pear). Oxytocin does not have a distinctive taste per se, so there is no problem in this respect. The ‘oxipop’ protocol involves firstly dissolving different peptide concentrations in volumes from 0.1 to 0.2 mL of 0.9% saline (with glycerol added as a preservative) and then freeze-drying the lollipops using two standard cycles lasting around a total of 48 h in a commercial freeze-dryer [40][33]. The resultant ‘oxipops’ are then sealed and placed in a fridge (2–8 deg C) until required (see Figure 2). The ‘oxipops’ can be stored stably for 3–6 months although possibly for longer periods. The freeze-drying process causes the formation of a white residue on the surface of the lollipop and so it is important for the purposes of blinding in placebo-controlled experiments to also produce placebo lollipops using application of the saline and glycerol solution without the peptide and using the same freeze-drying process. Sucking the ‘oxipops’ for a total of 3 min can achieve similar increases in plasma concentrations of oxytocin as does direct administration of the same concentration over the same time period using lingual sprays (see Figure 1). The AUC is 4.74 ± 2.41 pg/mL/h (n = 11, p = 0.004 vs. intranasal and p = 0.97 vs. lingual), with an estimated bioavailability of 4.4 ± 2.23% (p = 0.008 vs. intranasal and p = 0.97 vs. lingual), thereby confirming in vivo that a similar concentration of oxytocin is being released into the oral cavity and absorbed into the blood as with a direct lingual spray using the same dose. The Tmax was 39.5 ± 21.5, which is not significantly different than that for lingual or intranasal administration (Kruskall–Wallis ANOVA, p = 0.095).
3. Neural and Behavioral Effects of Oromucosal Administration of Oxytocin
A number of proof-of-concept studies have been conducted in human participants where the efficacy of single doses of oxytocin given by intranasal, lingual, and oral ‘oxipop’ routes was compared using both brain and behavioral measures. The first of these studies used a neural biomarker for the effects of intranasal oxytocin in adult male subjects, where it has been shown to decrease the responses of the amygdala to emotional faces [47[40][41],48], although it has been shown to increase them in females [49][42]. Findings demonstrated that the lingual administration of oxytocin increased rather than decreased amygdala responses in males and also tended to increase them in females. Additionally, lingual oxytocin increased the responses of the putamen (a brain reward region) in both males [34][26] and females [50][43], whereas intranasal oxytocin only did so in females. However, overall, lingual and intranasal oxytocin both appeared to influence neural responses to emotional faces, although with some route-dependent differences which might possibly have been due to the fact that increased oxytocin concentrations were greater after intranasal, compared to lingual, administration. Additionally, 24 IU of both intranasal and lingual oxytocin produce a similar facilitation of the perceived pleasantness of soft, caress-like touch, but not of medium pressure touch, as well as increased responses in brain reward (orbitofrontal cortex) and social cognition (superior temporal gyrus) processing regions [33][25]. Finally, a series of studies investigating the effects of oxytocin on top-down compared with bottom-up attention control found equivalent effects of 24 IU doses administered intranasally, lingually, and via ‘oxipops’ on top-down attention control, particularly in relation to social stimuli but also to some extent for non-social ones, using an anti-saccade task [35,40][27][33]. In this task, subjects were instructed to direct their eye movement (saccade) from a central point either towards (prosaccade) or away (anti-saccade) from a social or non-social stimulus. In general, subjects found it hard to look away from the social stimulus (anti-saccade), made more errors, and were slower to respond relative to when asked to look towards stimuli (pro-saccade). All three routes of oxytocin administration equivalently increased the number of errors subjects made when instructed to look away from stimuli and increased the time taken to look away from social stimuli. As such, this is a very sensitive task for measuring the facilitatory effects of oxytocin on the top-down control of attention and particularly social attention, which has important therapeutic potential in disorders where individuals exhibit impaired attention to social cues.References
- Hermesch, A.C.; Kernberg, A.S.; Layoun, V.R.; Caughey, A.B. Oxytocin: Physiology, pharmacology, and clinical application for labor management. Am. J. Obstet. Gynecol. 2023; in press.
- Gimpl, G.; Fahrenholz, F. The oxytocin receptor system: Structure, function, and regulation. Physiol. Rev. 2001, 81, 629–683.
- Kendrick, K.M.; Guastella, A.J.; Becker, B. Overview of human oxytocin research. Curr. Top. Behav. Neurosci. 2017, 35, 321–348.
- Yao, S.; Kendrick, K.M. Effects of Intranasal Administration of Oxytocin and Vasopressin on Social Cognition and Potential Routes and Mechanisms of Action. Pharmaceutics 2022, 14, 323.
- Le, J.; Zhang, L.; Zhao, W.; Zhu, S.; Lan, C.; Kou, J.; Zhang, Q.; Zhang, Y.; Li, Q.; Chen, Z.; et al. Infrequent Intranasal Oxytocin Followed by Positive Social Interaction Improves Symptoms in Autistic Children: A Pilot Randomized Clinical Trial. Psychother. Psychosom. 2022, 91, 335–347.
- Parker, K.J.; Oztan, O.; Libove, R.A.; Sumiyoshi, R.D.; Jackson, L.P.; Karhson, D.S.; Summers, J.E.; Hinman, K.E.; Motonaga, K.S.; Phillips, J.M.; et al. Intranasal oxytocin treatment for social deficits and biomarkers of response in children with autism. Proc. Natl. Acad. Sci. USA 2017, 114, 8119–8124.
- Yamasue, H.; Kojima, M.; Kuwabara, H.; Kuroda, M.; Matsumoto, K.; Kanai, C.; Inada, N.; Owada, K.; Ochi, K.; Ono, N.; et al. Effect of a novel nasal oxytocin spray with enhanced bioavailability on autism: A randomized trial. Brain 2022, 145, 490–499.
- Yatawara, C.J.; Einfeld, S.L.; Hickie, I.B.; Davenport, T.A.; Guastella, A.J. The effect of oxytocin nasal spray on social interaction deficits observed in young children with autism: A randomized clinical crossover trial. Mol. Psychiatry 2016, 21, 1225–1231.
- De Cagna, F.; Fusar-Poli, L.; Damiani, S.; Rocchetti, M.; Giovanna, G.; Mori, A.; Politi, P.; Brondino, N. The Role of Intranasal Oxytocin in Anxiety and Depressive Disorders: A Systematic Review of Randomized Controlled Trials. Clin. Psychopharmacol. Neurosci. 2019, 17, 1–11.
- Bharadwaj, V.N.; Tzabazis, A.Z.; Klukinov, M.; Manering, N.A.; Yeomans, D.C. Intranasal Administration for Pain: Oxytocin and Other Polypeptides. Pharmaceutics 2021, 13, 1088.
- Van Eyk, A.D.; Vand der Bijl, P.; Moll, L.M. Physicochemical characteristics of molecules and their diffusion across human vaginal mucosa. Eur. J. Inflamm. 2008, 2, 65–71.
- Prego, C.; Garcia, M.; Torres, D.; Alonso, M.J. Transmucosal macromolecular drug delivery. J. Control. Release 2005, 101, 151–162.
- Frokjaer, S.; Otzen, D.E. Protein drug stability: A formulation challenge. Nat. Rev. Drug Discov. 2005, 4, 298–306.
- Rathbone, M.J.; Drummond, B.K.; Tucker, I.G. The oral cavity as a site for systemic drug delivery. Adv. Drug Deliv. Rev. 1994, 13, 1–22.
- Bastos, F.; Pinto, A.C.; Nunes, A.; Simões, S. Oromucosal products—Market landscape and innovative technologies: A review. J. Control. Release 2022, 348, 305–320.
- Mehta, A.C. Buccal and oral drugs: Induction of labour. Acta. Chir. Hung. 1986, 27, 157–163.
- Westergaard, J.G.; Lange, A.P.; Pedersen, G.T.; Secher, N.J. Oral oxytocics for induction of labor. A randomized study of prostaglandin E2 tablets and demoxytocin resoriblets. Acta. Obstet. Gynecol. Scand. 1983, 62, 103–110.
- Gleeson, J.P.; Fein, K.C.; Whitehead, K.A. Oral delivery of peptide therapeutics in infants: Challenges and opportunities. Adv. Drug Deliv. Rev. 2021, 173, 112–124.
- Sharman, A.; Low, J. Vasopressin and its role in critical care. Cont. Ed. Anaesth. Crit. Care Pain 2008, 8, 134–137.26.
- van Kerrebroeck, P.; Nørgaard, J.P. Desmopressin for the treatment of primary nocturnal enuresis. Pediatr. Health 2009, 3, 311–327.
- Schiele, J.T.; Quinzler, R.; Klimm, H.-D.; Pruszydlo, M.G.; Haefeli, W.E. Difficulties swallowing solid oral dosage forms in a general practice population: Prevalence, causes, and relationship to dosage forms. Eur. J. Clin. Pharmacol. 2013, 69, 937–948.
- FDA. 2020. Available online: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/ferring-us-issues-voluntary-nationwide-recall-ddavpr-nasal-spray-10-mcg01ml-desmopressin-acetate (accessed on 8 May 2020).
- Kottke, D.; Burckhardt, B.B.; Knaab, T.C.; Breitkreutz, J.; Fischer, B. Development and evaluation of a composite dosage form containing desmopressin acetate for buccal administration. Int. J. Pharm. X 2021, 9, 100082.
- Hoffmann, E.M.; Breitenbach, A.; Breitkreutz, J. Advances in orodispersible films for drug delivery. Exp. Op. Drug Deliv. 2011, 8, 299–316.
- Chen, Y.; Zou, H.; Hou, X.; Lan, C.; Wang, J.; Qing, Y.; Chen, W.; Yao, S.; Kendrick, K.M. Oxytocin administration enhances pleasantness and neural responses to gentle stroking but not moderate pressure social touch by increasing peripheral concentrations. Elife 2023, 12, e85847.
- Kou, J.; Lan, C.; Zhang, Y.; Wang, Q.; Zhou, F.; Zhao, Z.; Montag, C.; Yao, S.; Becker, B.; Kendrick, K.M. In the nose or on the tongue? Contrasting motivational effects of oral and intranasal oxytocin on arousal and reward during social processing. Transl. Psychiatry 2021, 11, 94.
- Zhuang, Q.; Zheng, X.; Yao, S.; Zhao, W.; Becker, B.; Xu, X.; Kendrick, K.M. Oral Administration of Oxytocin, Like Intranasal Administration, Decreases Top-Down Social Attention. Int. J. Neuropsychopharmacol. 2022, 25, 912–923.
- Martins, D.A.; Mazibuko, N.; Zelaya, F.; Vasilakopoulou, S.; Loveridge, J.; Oates, A.; Maltezos, S.; Mehta, M.; Wastling, S.; Howard, M.; et al. Effects of route of administration on oxytocin-induced changes in regional cerebral blood flow in humans. Nat. Commun. 2020, 11, 1160.
- Pawar, P.G.; Darekar, A.B.; Saudagar, R.B. Medicated chocolate and lollipops: A novel drug delivery system for pediatric patients. Pharma. Sci. Monitor. 2018, 9, 677–696.
- Shetty, S.; Kamath, K.; Shabaraya, R.; Miranda, F.C. Design and development of medicated lollipop containing albendazole. Am. J. Pharmatech. Res. 2019, 9, 275–285.
- Tangso, K.J.; Ho, Q.P.; Boyd, B.J. Confectionery-based dose forms. Curr. Drug Deliv. 2015, 12, 56–62.
- Gasmi Benahmed, A.; Gasmi, A.; Arshad, M.; Shanaida, M.; Lysiuk, R.; Peana, M.; Pshyk-Titko, I.; Adamiv, S.; Shanaida, Y.; Bjørklund, G. Health benefits of xylitol. Appl. Microbiol. Biotechnol. 2020, 104, 7225–7237.
- Xu, D.; Li, Q.; Zhuang, Q.; Zhang, Y.; Yao, S.; Zhao, W.; Kendrick, K.M. Oro-mucosal administration of oxytocin using medicated lollipops alters social attention, similar to intranasal and lingual routes: Implications for therapeutic use. Front. Neurosci. 2022, 16, 1022101.
- Daughters, K.; Manstead, A.S.R.; Hubble, K.; Rees, A.; Thapar, A.; van Goozen, S.H.M. Salivary Oxytocin Concentrations in Males following Intranasal Administration of Oxytocin: A Double-Blind, CrossOver Study. PLoS ONE 2015, 10, e0145104.
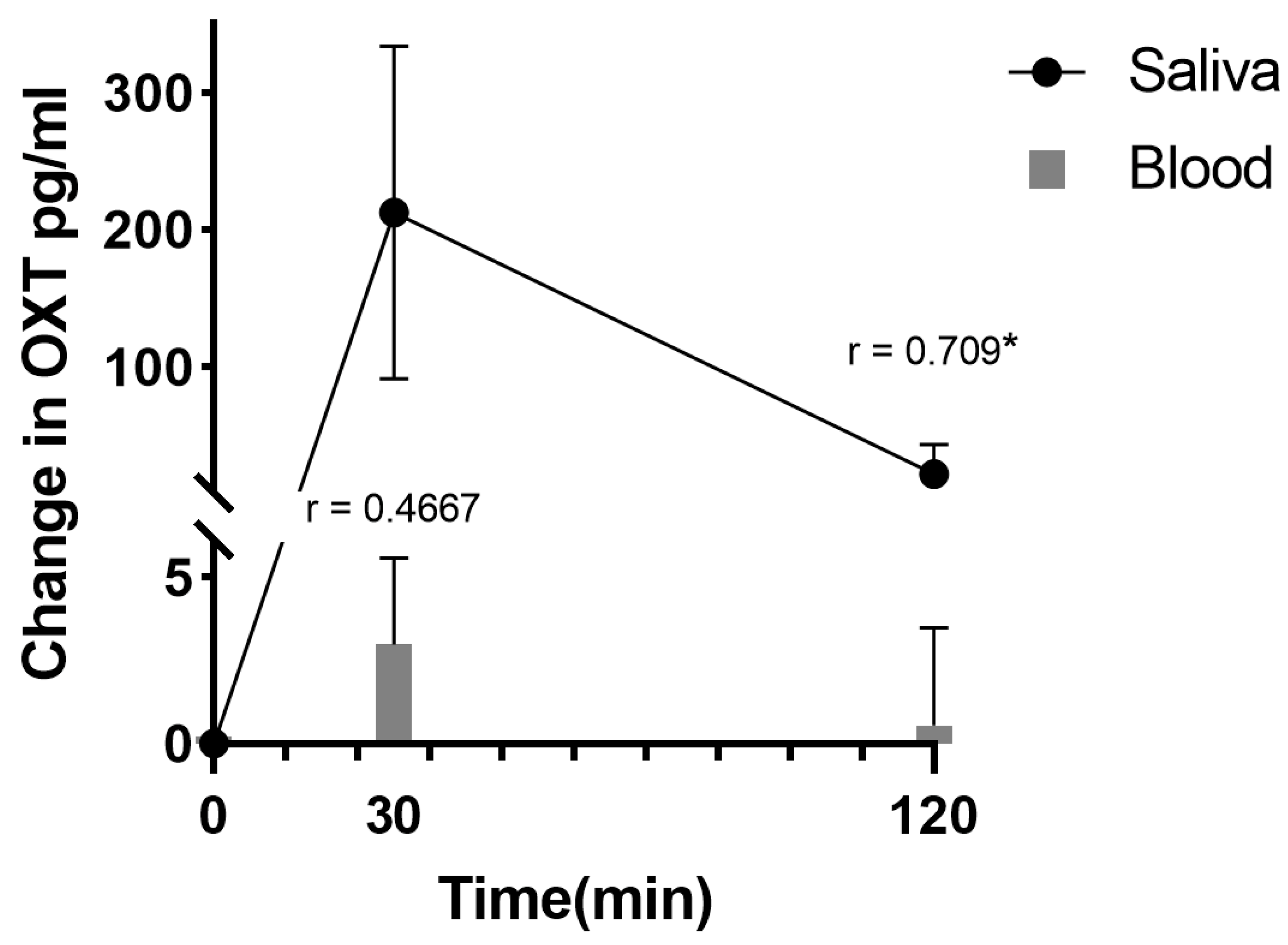
- van Ijzendoorn, M.H.; Bhandari, R.; van der Veen, R.; Grewen, K.M.; Bakermans-Kranenburg, M.J. Elevated Salivary Levels of Oxytocin Persist More than 7 h after Intranasal Administration. Front. Neurosci. 2012, 6, 174.
- Weisman, O.; Zagoory-Sharon, O.; Feldman, R. Intranasal oxytocin administration is reflected in human saliva. Psychoneuroendocrinology 2012, 37, 1582–1586.
- Weisman, O.; Schneiderman, I.; Zagoory-Sharon, O.; Feldman, R. Salivary vasopressin increases following intranasal oxytocin administration. Peptides 2013, 40, 99–103.
- Martins, D.; Gabay, A.S.; Mehta, M.; Paloyelis, Y. Salivary and plasmatic oxytocin are not reliable trait markers of the physiology of the oxytocin system in humans. Elife 2020, 9, e62456.
- Striepens, N.; Kendrick, K.M.; Hanking, V.; Landgraf, R.; Wüllner, U.; Maier, W.; Hurlemann, R. Elevated cerebrospinal fluid and blood concentrations of oxytocin following its intranasal administration in humans. Sci. Rep. 2013, 3, 3440.
- Kou, J.; Zhang, Y.; Zhou, F.; Sindermann, C.; Montag, C.; Becker, B.; Kendrick, K.M. A randomized trial shows dose-frequency and genotype may determine the therapeutic efficacy of intranasal oxytocin. Psychol. Med. 2022, 52, 1959–1968.
- Spengler, F.B.; Schultz, J.; Scheele, D.; Essel, M.; Maier, W.; Heinrichs, M.; Hurlemann, R. Kinetics and Dose Dependency of Intranasal Oxytocin Effects on Amygdala Reactivity. Biol. Psychiatry 2017, 82, 885–894.
- Lieberz, J.; Scheele, D.; Spengler, F.B.; Matheisen, T.; Schneider, L.; Stoffel-Wagner, B.; Kinfe, T.M.; Hurlemann, R. Kinetics of oxytocin effects on amygdala and striatal reactivity vary between women and men. Neuropsychopharmacology 2020, 45, 1134–1140.
- Lan, C.; Chen, Y.; Zhang, Y.; Kou, J.; Huang, L.; Xu, T.; Yang, X.; Xu, D.; Yang, W.; Kendrick, K.M.; et al. Oral Oxytocin Facilitates Responses to Emotional Faces in Reward and Emotional-Processing Networks in Females. Neuroendocrinology 2023, 113, 957–970.
