Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Elena Puerta and Version 2 by Sirius Huang.
Sirtuin 2 (SIRT2), one of the seven members of the sirtuin family, has emerged as a potential regulator of aging and age-related pathologies since several studies have demonstrated that it shows age-related changes in humans and different animal models. A detailed analysis of the relevant works addressing this topic shows that the changes that occur in SIRT2 with aging seem to be opposite in the brain and in the periphery.
- aging
- brain
- epigenetics
- inflammation
- neurodegenerative diseases
- sirtuin 2
1. SIRT2 and Aging
In the last decades, with the increase of life expectancy, there has been a demographic growth in the elderly population, a tendency that is expected to continue. According to the World Health Organization, it is expected that from 2015 to 2050 the world’s population over 60 years old will almost double, raising from 12% to 22% [1]. As the tendency to disease increases with age, the focus on healthy aging and its research is becoming more relevant. Considering the aging process is very diverse, if we aim to understand it, the influence of genetics should not be ignored. Gene expression can be altered in many levels such as DNA replication, transcription, RNA translation, and post-translational modifications of proteins. In this context, epigenetics could also be a piece of the puzzle. Epigenetics is defined as “the study of changes in gene function that are mitotically and/or meiotically heritable and that do not entail a change in DNA sequence” [2]. These changes occur through several mechanisms, all of which can be modulated by environmental, physiological, and pathological processes. Some of the predominant epigenetic mechanisms are ATP-dependent chromatin-remodeling complexes, non-coding RNAs, covalent modifications of DNA bases and histone modifications [3].
In particular, histone modifications have been widely studied. Histones are octameric proteins around which DNA strands wrap, conforming the chromatin. These DNA-binding proteins provide a structural support to DNA, that can be extended or compacted depending on the modifications (methylation, acetylation, phosphorylation, and ubiquitination) that take place in their tails. Notably, histone acetylation on the lysine residues reduces chromatin condensation becoming more accessible to transcription factors, and activating the transcription of genes located in the affected region. This results in an increase gene expression. Histone acetyltransferases (HAT) and histone deacetylases (HDAC) are crucial in these epigenetic modifications since they are responsible for dynamic histone acetylation and deacetylation, respectively, tuning the level of transcripts (for a review, see [4]) (Figure 1).
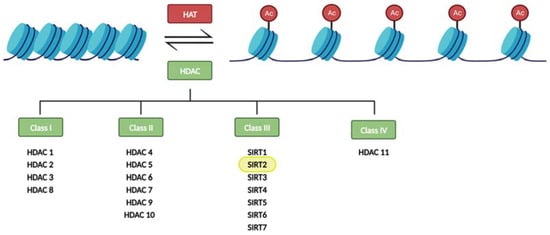
Figure 1. Regulation of histone acetylation levels by histone acetyl transferases (HATs) and histone deacetylases (HDACs). Sirtuins constitute the class III of HDACs.
Eighteen HDAC proteins are classified into four classes named I–IV. Class III HDACs, also known as sirtuins, are nicotinamide adenine nucleotide- (NAD+) dependent, while the other three classes require zinc as a cofactor. Sirtuins also differ from other HDACs in their unique ability to catalyze ADP-ribosylation [5][6][5,6].
Sirtuins are present in every living species and widely distributed along the tissues [7]. Besides deacetylating histone proteins, they are also key in the control of many physiological processes in mammals. For instance, they take part in the regulation of the cell cycle, antioxidant protection, inflammation, neurogenesis, and various metabolic pathways. Thus, their functions are often related to stress response, aging, and general homeostasis [8][9][8,9].
To date, seven isoforms of sirtuins (SIRT1-7) have been described in mammals. They all differ in their functions among other reasons due to diverse terminal regions, subcellular location, enzymatic activities, and substrates [10][11][10,11]. Regarding their subcellular location, SIRT1 is mostly located in the nucleus, with the ability to migrate to the cytosol. SIRT2, on the other hand, is the only sirtuin that is predominantly cytosolic, although it has the capacity to shuttle to the nucleus and mitochondria. SIRT3, SIRT4, and SIRT5 are mitochondrial sirtuins, being SIRT3 and SIRT5 able to migrate to the cytoplasm. SIRT6 is found in the nucleus, associated with chromatin, and SIRT7 in the nucleolus and nucleus [9][12][13][14][9,12,13,14].
Although their functions are very diverse and many studies are still needed to understand their role in each process, it has been described that SIRT1 is involved in inflammation, oxidative stress, cell proliferation, and apoptosis [15][16][15,16], while the other nuclear sirtuins, SIRT6 and SIRT7, participate in DNA repair [17] and regulation of gene transcription [18], respectively. Mitochondrial sirtuins SIRT3, SIRT4, and SIRT5 are involved in the response to oxidative stress and metabolic pathways inside this organelle [19]. SIRT2 has been involved in multiple functions regulating gene expression and many metabolic pathways [20].
Since the interest in sirtuins is growing, research is being carried out on all the members of the sirtuin family in different experimental models, tissues, and conditions, leading to new findings that intend to clarify their physiological and pathological relevance. Specifically, several recent studies have described age-related changes in SIRT2 in different organs and tissues [21][22][23][24][21,22,23,24]. This has led to the hypothesis that SIRT2 could play a key role in the aging process. However, these changes are not the same in all tissues, since an increase [25][26][27][25,26,27] or a decrease [28][29][30][28,29,30] in its expression has been described depending on the tissue analyzed. Therefore, a detailed understanding of the function of SIRT2 with aging in each cell type is necessary to determine if it is an interesting target for the treatment of diseases associated with aging.
2. CNS SIRT2 Expression in Aging
SIRT2 is the most abundant member of the sirtuin family in the CNS, notably in the cortex, striatum, hippocampus, and spinal cord [12]. In recent years, this fact has exponentially increased interest in SIRT2 in neuroscience research in order to decipher its implication in aging and age-related neurodegenerative disorders. The first study on this topic was published by Maxwell and colleagues in 2011 [25]. In this study, the authors found that three isoforms predicted for SIRT2 (SIRT2.1, SIRT2.2, and SIRT2.3) are expressed in the brain. They analyzed young adult (4–5-month-old) and aged (19–22-month-old) C57BL/6 mice and found an age-related accumulation of the isoform SIRT2.3 in spinal cord extracts and cortices. As a result, total SIRT2 levels had a modest but significant increase in aged mice. Notably, they detected an association between the local accumulation of SIRT2 protein and areas of reduced tubulin acetylation in cell bodies and neurites which could be affecting neuronal function. More recently, the behavioral and molecular consequences of the overexpression of SIRT2.3 in the hippocampus have been addressed [31][45]. According to this work, the overexpression of SIRT2.3 does not result in relevant behavioral or molecular changes in control mice. However, in a mouse model of accelerated aging, the SAMP8 model, SIRT2.3 overexpression worsened learning and memory performance and increased the expression of the pro-inflammatory cytokine IL-1β. Based on these results, the increase of SIRT2.3 in aged brains does not seem to induce or prevent senescence, but it could play a part in the progression of age-related processes together with other risk factors. In line with Maxwell’s findings, a study checked for SIRT2 level variations in various brain areas of female Wistar rats at 3, 12, and 24 months old. An increase in Sirt2 mRNA and protein levels was observed, but exclusively in the occipital lobe. This increase was paired with a significant enhancement in deacetylated FOXO3a (Forkhead Box, class O3a) transcription factor, a substrate deacetylated by SIRT2, in the same region [24]. Supporting this data, a recent study, analyzing the cortex and hippocampus of 3- and 22-month-old Wistar albino male rats, has shown an increase in SIRT2 and FOXO3a brain levels during the aging process, accompanied by an increase in oxidative stress and apoptosis [27]. In addition, authors randomly administered melatonin, which is reported to have antioxidant, anti-apoptotic, and anti-aging properties, and is physiologically reduced in aging; the SIRT2 inhibitor salermide, or DMSO as a control to both young and aged rats. They described a reduction in SIRT2 and FOXO3 protein levels in the hippocampus but not in the cortex of aged rats treated with melatonin. Interestingly, salermide administration to aged rats led to the inhibition of SIRT2 and FOXO3 in both regions. Considering these results and the functions attributed to melatonin in aging, the authors suggest that SIRT2 and FOXO3 could play a key role in oxidative stress and apoptosis [27]. Indeed, the FOXO transcription factors are regulated by post-translational modifications, and SIRT2-mediated deacetylation of FOXO3a promotes its ubiquitination and degradation [32][46]. In fact, SIRT2 deacetylates FOXO3a and stimulates its translocation to the nucleus, therefore inducing apoptosis [33][47]. In addition, exposure to oxidative stress upregulates FOXO3a in the hippocampus [34][48], enhanced levels of SIRT2 lead to cell death, and the inhibition of SIRT2 results in a reduction of oxidative stress and apoptosis [22][35][36][37][38][22,49,50,51,52]. In agreement with these observations, an increase in FOXO3a activity has been found in neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease [39][40][53,54]. However, the reason why the increase in SIRT2 is only observed in certain brain regions and the physiological consequences of these changes need to be further investigated. Supporting the notion that elevated SIRT2 levels in the CNS are deleterious, another study has shown an upregulation of SIRT2 in the brain of a D-galactose-induced aging rat model [41][55]. In fact, as a result of D-galactose administration, the expression of the pro-inflammatory cytokines interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) increased, while the autophagic marker Beclin-1 was downregulated. Noteworthy, metformin supplementation induced an anti-aging effect, downregulating the expressions of SIRT2, IL-6, and TNF-α, whereas increasing Beclin-1 expression. The authors state that metformin promotes the activation of autophagy and reduces inflammation, hence restoring the antioxidant status and improving brain aging [41][55]. In line with these results, the implication of SIRT2 in autophagy has also been described in different studies. It has been shown that upregulated SIRT2 interferes with autophagy efficiency and promotes protein accumulation under proteasome inhibition, intensifying proteinopathy-related cytotoxicity [42][56]. Consistently, lowered SIRT2 increases autophagy levels [43][57]. Taking into account that, in the context of neurodegenerative disorders, inadequate autophagy induces neuronal cell death while activated autophagy is neuroprotective, these results further justify the deleterious consequences of age-related increases in SIRT2 in the CNS. In a different publication, Diaz-Perdigon and colleagues [26] compared SIRT2 protein and mRNA levels in 2- and 9-month-old male SAMR1 and SAMP8 mice. In both models, they found a significant increase in hippocampal SIRT2 protein in aged animals, with no significant differences between both strains. Therefore, they pointed out SIRT2 as a possible biomarker of the aging process. However, this increase did not correlate with changes in Sirt2 mRNA, which according to the authors, indicates protein accumulation rather than an increase in its synthesis. In contrast, there were no significant changes in the protein expression in the frontal cortex and striatum. Interestingly, in order to understand the physiological consequences of the observed SIRT2 increase, they administered the SIRT2 inhibitor 33i to 5- and 8-month-old SAMP8 and SAMR1 mice. Authors conclude that early SIRT2 inhibition improves age-related cognitive decline and prevents neuroinflammation in SAMP8 mice. However, the inhibition of SIRT2 once the aging phenotype is well established (in 8-month-old SAMP8 mice) cannot reverse age-induced behavioral and molecular changes [26]. These results point to SIRT2 inhibition as a promising therapeutic target to prevent age-related cognitive decline. In agreement with all these studies, it has been recently published that SIRT2 protein expression increases gradually with aging in the cortex and hippocampus isolated from 3-, 6-, 12-, and 24-month-old C57BL/6 wild type (WT) mice [44][58]. Interestingly, the authors show that, at the same time, SIRT1 expression decreases gradually; thus, the SIRT2:SIRT1 ratio gradually increases with age. In an attempt to understand how the changes in SIRT1 and SIRT2 levels may affect the vulnerability of the neurons to a neurotoxic insult, SH-SY5Y neuroblastoma cells were transfected with empty vector, flag-tagged SIRT1 or SIRT2, and then treated with Aβ42 oligomers. They found that Aβ substantially increased cell death when transfecting cells with an empty vector, whereas SIRT1 overexpression largely restored the cell damage by Aβ. On the other hand, SIRT2 overexpression reduced the survival of Aβ42-treated cells compared to untreated cells [44][58]. Together, these data support the notion that SIRT1 and SIRT2 have inverse effects on neuron viability; SIRT1 protects against neurotoxicity, while SIRT2 promotes it. The findings of the studies mentioned above point out an increase of SIRT2 in the CNS during the aging process. However, another study performed by Kireev and colleagues showed differing results when testing male Wistar rats [45][59]. In this case, the researchers found a significant age-related decrease of Sirt2 mRNA accompanied by an increase in gene and protein levels of pro-apoptotic markers (Bax and Bad) in the dentate gyrus comparing 2- and 22-month-old animals [45][59]. Noteworthy, in this case, growth hormone treatment reduced the pro/anti-apoptotic ratio to levels observed in young animals and also increased SIRT2 levels, which was accompanied by a reduction in apoptosis and enhanced survival markers in this part of the hippocampus. In general terms, most of the studies collected in this section agree in concluding that SIRT2 seems to be increased in the CNS with aging (Table 1) and that this increase seems to be harmful by promoting oxidative stress and neurodegeneration. Therefore, based on these conclusions, SIRT2 inhibition or different strategies aimed at counteracting age-related increases in SIRT2 could be considered good therapeutic options for age-related diseases.Table 1.
Sirtuin 2 expression variations in the aging CNS.
| Authors and Year | Analyzed Model | Sample | Sirtuin 2 Expression with Aging | |
|---|---|---|---|---|
| Specie | Ages Compared in Months | |||
| Maxwell et al., 2011 [25] | ||||
Table 2.
Sirtuin 2 expression changes with aging in the periphery.
| Authors and Year | Analyzed Model | Sample | Sirtuin 2 Expression with Aging | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Specie | Ages Compared | ||||||||
| C57BL/6 mouse | 4–5 vs. 19–22 | Spinal cord and cortex | Increase | ||||||
| Kireev et al., 2013 [45 | |||||||||
| Chambers et al., 2007 [21] | C57BL/6 mouse | 2- vs. 21-month-old | HSCs isolated from BM | Decrease | |||||
| ][59] | |||||||||
| Yudoh et al., 2015 [46][60] | Male Wistar rat | 2 vs. 22 | Hippocampus | Human |
22- to 66-year-old(dentate gyrus) | Decrease | |||
| PBMCs | Decrease | Braidy et al., 2015 [24] | Female Wistar rat | 3 vs. 12 vs. 24 | Occipital lobe | ||||
| Luo et al., 2019 [29] | C57BL/6 mouse | Increase | |||||||
| 3- vs. 24-month-old | HSCs isolated from BM | Decrease | Garg et al., 2017 [41][55] | Male Wistar rat | 4 vs. 24 | Whole brain | Increase | ||
| Wongchitrat et al., 2019 [28] | Human | 25- to 35-year-old vs. ≥65-year-old | Peripheral blood (plasma) | Increase | Diaz-Perdigon et al., 2020 [26] | Male SAMR1 and SAMP8 mice | 2 vs. 9 | Hippocampus | Increase |
| Lehallier et al., 2019 [23] | Human | 18- to 95-year-old | Peripheral blood (plasma) | Decrease | Keskin-Atkan et al., 2022 [27] | Male Wistar rat | |||
| He et al., 2020 [ | 3 vs. 22 | 47][ | Hippocampus and cortex | Increase | |||||
| 44] | Male C57BL/6 mouse | 3- vs. 24-month-old | Macrophages isolated from BM | Decrease | Li et al., 2023 [44][58] | C57BL/6 mouse | 3 vs. 6 vs. 12 vs. 24 | Hippocampus and cortex | Increase |
3. Peripheral SIRT2 Expression in Aging
Considering that the expression of SIRT2 is very extensive in the periphery, several works have also addressed the changes that occur in its expression in different peripheral cell types (Table 2| Ye et al., 2023 | ||||
| [ | ||||
| 30 | ||||
| ] | ||||
| Cynomolgus macaque | 4- to 6- vs. 18- to 21-year-old | Cardiomyocytes | Decrease | |
| Zhang et al., 2023 [48][61] | C57BL/6 mouse | 4- vs. 24-month-old | Aorta and VSMCs | Decrease |
BM: bone marrow; HSC: hematopoietic stem cell; PBMC: peripheral blood mononuclear cell; VSMC: vascular smooth muscle cell.
