Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Andrea Ehrmann and Version 4 by Catherine Yang.
Exchange bias (EB) is a unidirectional anisotropy occurring in exchange-coupled ferromagnetic/antiferromagnetic systems, such as thin films, core–shell particles, or nanostructures. In addition to a horizontal shift of the hysteresis loop, defining the exchange bias, asymmetric loops and even vertical shifts can often be found.
- exchange bias (EB)
- hysteresis loop shift
- coercivity
- ferromagnet
- antiferromagnet
1. Introduction
The exchange bias (EB), a unidirectional magnetic anisotropy, was first reported by Meiklejohn and Bean for Co/CoO core–shell particles [1][2][1,2] and has since been extensively investigated. The main effect is a horizontal shift of the hysteresis loop in a system consisting of a ferromagnet (FM) exchange-coupled to an antiferromagnet (AFM), similar to a frozen internal magnetic field applied to the FM by the AFM’s fixed magnetic moments. Nevertheless, this simple explanation and corresponding naïve models cannot fully simulate the value of the horizontal shift, nor the other correlated changes in the hysteresis loop as compared to pure ferromagnets, i.e., an often-visible asymmetry of the loop as well as a potential vertical shift [3].
In addition to the first investigations of core–shell particles, usually with a ferromagnetic core and oxidized antiferromagnetic shell, experiments have evolved rapidly towards thin film systems [4][5][4,5] and further to nanostructured systems [6][7][8][6,7,8]. The materials under investigation are often ferromagnets coupled with antiferromagnets or ferrimagnets, such as Co/CoO [9], Fe/FeF2 [10], or Fe/MnF2 [11][12][11,12]. Recently, more sophisticated systems have become part of experimental and theoretical studies, e.g., Fe/LaAlO3 [13] or Pr0.67Sr0.33MnO3/SrTiO3 [14].
In addition to developing new materials for innovative EB systems with an enhanced effect size, more asymmetric loops, or other technically useful effects, e.g., applications for hard disk read/write heads and spintronics devices, the magnetic properties of well-known material systems can also be manipulated by creating nanostructures with different shapes and dimensions. In their comprehensive review from 2005, Nogués et al. described the effects in detail of the former state of research [6]. However, research activities dealing with exchange bias in general and exchange bias in nanostructures in particular have considerably increased since then, as depicted in Figure 1. It is intriguing that including the word “nano” in the search causes the bibliographic data to pass through a maximum value of around 2015. This indicates the achievement of some kind of technological excellence in the preparation of exchange-biased structures.
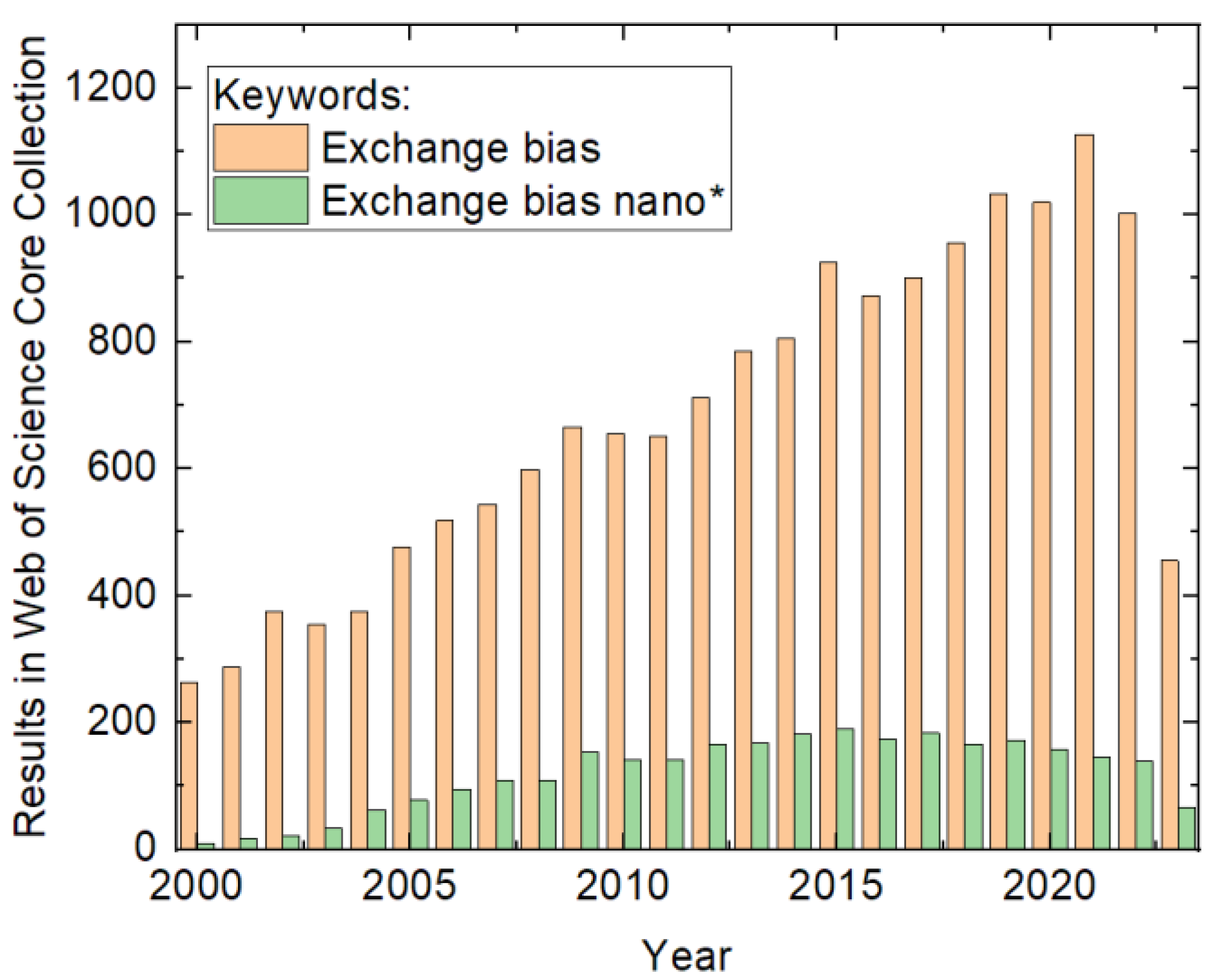

Figure 1. Numbers of results in the Web of Science Core Collection for the keywords provided in the inset, counted on 15 July 2023.
Numbers of results in the Web of Science Core Collection for the keywords provided in the inset, counted on 15 July 2023.
2. Properties of Exchange-Biased Nanostructures
Generally, exchange bias systems consisting of a ferromagnet and an antiferromagnet show a horizontal shift of their magnetization hysteresis loop when they are cooled through the Néel temperature of the AFM (Figure 2a). This is often accompanied by a broadening of the loop (Figure 2b), a vertical loop shift, or an asymmetry of the loop, which are attributed to unidirectional exchange bias anisotropy [6].
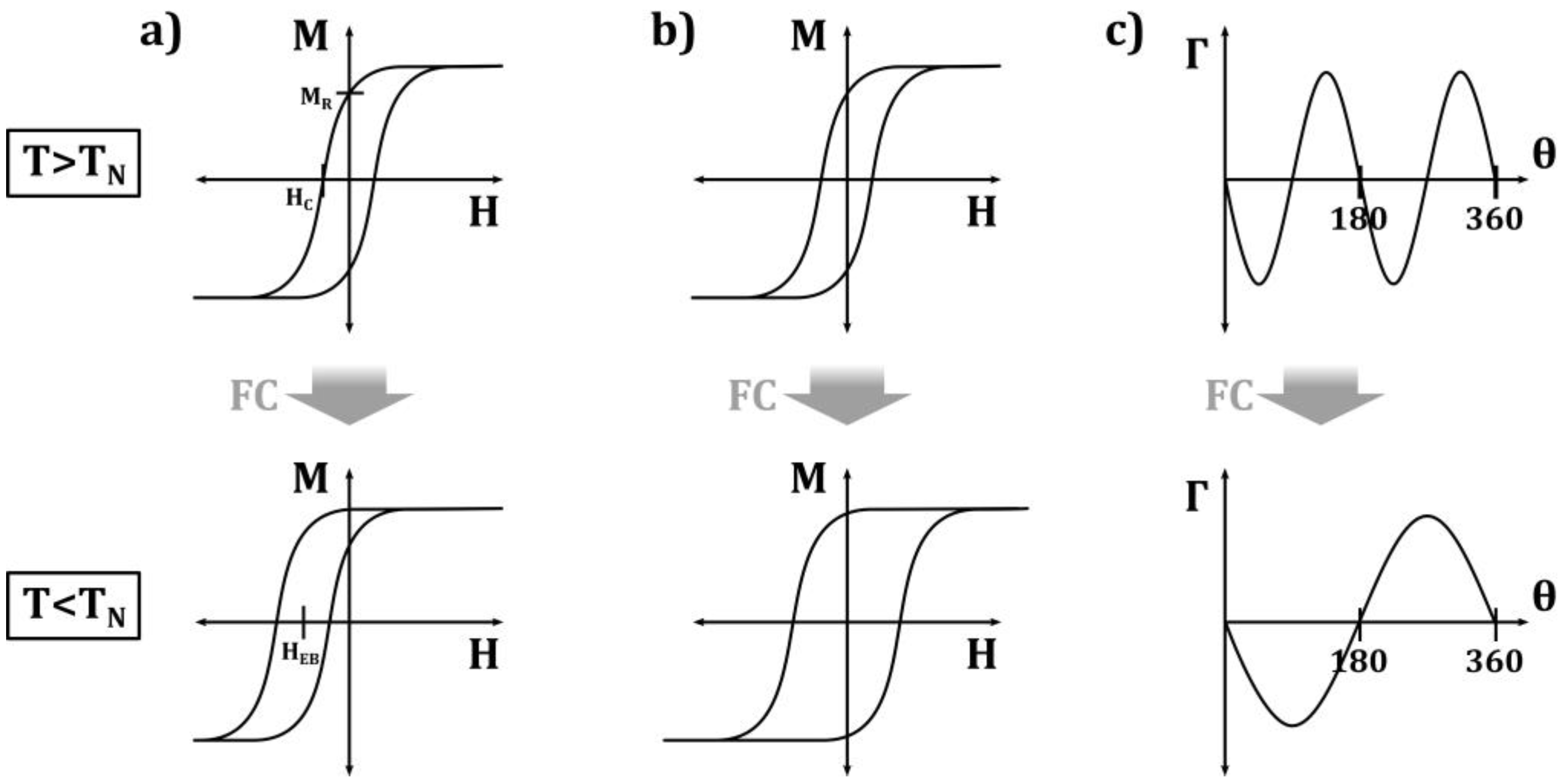

Figure 2. Schematic representation of the main effects induced by the FM–AFM exchange coupling causing (a) loop shift, (b) coercivity enhancement, and (c) unidirectional anisotropy. Redrawn illustration from [6].

All these effects are temperature-dependent, with a larger EB typically at lower temperatures, while a sign change in the loop shift near the Néel temperature is also possible [15][16][15,16]. The blocking temperature TB, above which no EB is visible, can be significantly lower than the Néel temperature.
Other parameters affecting the exchange bias are the thickness of the layers or shells [5], the cooling field [17], the roughness of the interface between the FM and AFM [18], as well as the AFM orientation and crystallinity [5][19][5,19].
Especially in nanostructured EB systems, other effects may occur. Generally, a size reduction in a single FM or AFM may lead to a change in magnetic properties, as compared to bulk or even thin film materials [20][21][22][23][24][20,21,22,23,24]. The superposition of the EB-induced anisotropy with the shape anisotropy, which becomes increasingly important at smaller scales, can lead to even more interesting and partly counterintuitive results [25][26][27][25,26,27].
As the reader can recognize, there are no clear dependencies of the exchange bias on the material, structural, or other parameters—a thicker AFM can increase or decrease the EB field, a larger cooling field can increase or decrease the EB or even switch its sign, another crystal orientation can completely change the temperature-dependent asymmetry of the hysteresis loop, etc.
3. Ni/NiO Nanostructures
Similar to Co/CoO, Ni/NiO can also be found in the form of core/shell particles. Querejeta-Fernández et al. described the preparation of such nanoparticles with an average diameter of 10 nm by the thermal decomposition of a medium containing a Ni2+ salt, followed by a reduction step to yield Ni crystallization and finally the oxidation of the shell [28][115]. They observed large EB shifts for small- and middle-core diameters and smaller EB fields for larger cores with thin NiO shells. Johnston-Peck et al. used solution chemistry with subsequent solution-phase oxidation instead of preparing Ni/NiO core–shell nanoparticles with shell thicknesses of 2–3 nm and core diameters of 8–24 nm [29][116]. While the temperature-dependence of the sample magnetization depended on the core and shell diameters, these samples generally showed no horizontal EB shifts, but small increases in coercivity indicating a weak EB. For core–shell particles prepared by a sol-gel route with diameters of 8–27 nm, Thakur et al. investigated the cooling field dependence and observed a slightly reduced EB for cooling fields larger than 20 kOe [30][117]. Rinaldi-Montes et al. prepared Ni/NiO core/shell nanoparticles by the pyrolysis of an inorganic precursor in the pores of an active carbon matrix, followed by oxidation in air [31][118]. These nanoparticles showed a shell thickness of 2 nm and varying core diameters, depending on the pyrolysis temperature. The authors reported that the shell froze into a spin glass state below approximately 40 K, correlated to an EB shift of the measured hysteresis loops below this temperature, which was far below the bulk Néel temperature of NiO of 523 K. While most studies of Ni/NiO nanostructures are based on core/shell structures, a few other exchange-biased Ni/NiO nanostructures were investigated. Kremenovic et al. prepared nanocomposites of 62% NiO with crystallite sizes of about 11 nm and much larger crystallite sizes of 278 nm for Ni [32][119]. Using thermal annealing in air, the NiO content and crystallite sizes increased, while the Ni crystallite sizes decreased. However, high-energy ball milling resulted in a reduction in the NiO content and overall decreased the crystallite size. An EB was found in milled samples with particle sizes of 10 nm for NiO and 11 nm for Ni, while larger crystallites resulted in a reduced coupling area and correspondingly vanishing EB.4. Other Exchange-Biased Nanostructures Containing Nickel Oxides
Similar to cobalt oxide nanostructures exchange-coupled to ferromagnets other than cobalt, there are also few reports about NiO combined with other ferro- or ferrimagnets. Tsopoe et al. prepared core–shell nanoparticles combining NiO with the ferrimagnet Fe3O4, testing the AFM as a core and shell, respectively [33][120]. They observed rod-shaped NiO nanoparticles, while pure Fe3O4 nanoparticles and both sorts of core–shell nanoparticles were spherical, as shown in Figure 39. All diameters were in the range of 30–50 nm. For both sorts of core–shell nanoparticles, the blocking temperature was around 200–250 K, with the highest EB of 330 Oe at 60 K observed for NiO@Fe3O4 core–shell nanoparticles. Interestingly, the authors observed an EB shift along the positive x-axis, i.e., opposite to the common direction, for Fe3O4@NiO core–shell particles, which they explained by more pinning of down-spins at the core–shell interface. The coercive fields of all nanoparticles, both pure and core–shell, decreased with the increasing temperature. Embedding NiFe2O4 ferrimagnetic nanoparticles in a NiO matrix, Tian et al. also observed a blocking temperature of approximately 250 K [34][121]. The authors explained the EB by the exchange interaction between the ferrimagnetic nanoparticles and the spin glass-like interface phase.
Figure 39. Scanning electron microscope images of (a) pure Fe3O4 and (b) NiO nanoparticles, as well as core–shell nanoparticles of (c) Fe3O4@NiO and (d) NiO@Fe3O4. Adapted from [33][120], originally published under a CC-BY license.
Such a spin glass state, formed below 10 K, was also mentioned by Rinaldi-Montes et al. who prepared NiO nanoparticles [35][122]. Similar to the aforementioned CoO or Co3O4 nanoparticles, they observed an EB for nanoparticles larger than a 2 nm diameter, which they attributed to the magnetic coupling between the AFM core and spin glass shell. Winkler et al. reported the spin glass state of 3 nm NiO nanoparticles to occur below 15 K [36][123]. Makhlouf et al. investigated the temperature dependence of the EB in NiO nanoparticles depending on the NP diameter and observed a lower blocking temperature and also smaller exchange bias shift for smaller nanoparticles, while the greatest EB was achieved for a nanoparticle diameter of 26 nm [37][124].
