Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Dario Savoca and Version 2 by Camila Xu.
Phthalic acid esters (PAEs), commonly named phthalates, are a class of dialkyl or alkyl/aryl esters of phthalic acid (1,2-benzenedicarboxylic acid) structured in one benzene ring linked with two aliphatic ester groups, most commonly in the ortho configuration.
- phthalic acid esters
- endocrine active substances
- plastic pollution
1. Introduction
Phthalic acid esters (PAEs), commonly named phthalates, are a class of dialkyl or alkyl/aryl esters of phthalic acid (1,2-benzenedicarboxylic acid) structured in one benzene ring linked with two aliphatic ester groups, most commonly in the ortho configuration [1][2][1,2]. PAEs were used for the first time as additives in plastics in the 1920s and continue to be the largest plasticiser class in the 21st century [3]. Among all the possible sources of contamination, the impact of plastics in different environmental matrices has contributed to the widespread presence of phthalates. The release of chemicals associated with plastics into the marine environment is receiving increasing attention. Phthalates are biologically active compounds that dissolve in water to varying degrees depending on the physicochemical characteristics of the side chains, particularly octanol/water partitioning (Kow). Organisms can absorb these substances by ingestion, inhalation, or contact [1].
In the organisms, PAEs are metabolised into toxic compounds that can impair vital functions. Di-2-ethylhexyl phthalate (DEHP) and di-n-butyl phthalate (DnBP) are two of the most toxic and frequently used phthalates [4].
Animal experiments have shown that phthalates interfere with normal physiological processes mediated by hormones essential for reproduction, growth, and development (e.g., decreased testis weight, spermatogenesis impairment, and external genital malformations), leading to the so-called “phthalate syndrome” [5].
Based on the concentration, the nature of the compound, the physicochemical parameters of the environment, and the organism involved, exposure to PAEs leads to different effects and levels of chronic and acute toxicity [6].
Exposure to PAEs also adversely affects the behaviour and health of adults and their offspring [7][8][7,8] causing, among others, hepatotoxicity, oxidative stress, neurodevelopmental changes, genetic aberrations, and epigenetic reprogramming [7][9][10][11][12][7,9,10,11,12]. Depending on effects and exposure levels, phthalates can be considered risk factors for many multifactorial diseases (e.g., reproductive pathologies, developmental alterations and embryogenesis, including the hatching success of eggs, metabolic syndromes, and tumours) [7][13][7,13]. These effects are symptomatic of a hormone balance disorder; therefore, phthalates are endocrine active substances (EAS) that can interact or interfere with normal hormonal action, showing effects of different types and severity. For this reason, they can be called modulators, perturbators, disruptors, or endocrine destroyers.
In general, EAS can act in several ways: (i) mimic the action of the hormone naturally produced, inducing an excessive response or at the wrong times (agonistic effect); (ii) block the receptor, preventing the hormone from binding there so that it cannot act (antagonistic effect); (iii) alter the regulation of hormones, acting “upstream” on their production; (iv) alter the transport of hormones in the blood [14].
PAEs are substances of concern, as reiterated in the 2021 UN report on plastic pollution [15]; consequently, restrictive measures have been introduced, limiting their use.
The regulations on the restrictions on the use of phthalates are different between international legislations; moreover, they consider only phthalates with high rates of application, and thus, high risk of exposure, which are listed as toxic, for example, di-methyl phthalate (DMP), benzyl butylphthalate (BBzP), DEHP, DnBP, di-iso-nonyl phthalate (DiNP), di-iso-decyl phthalate (DiDP), and di-n-octyl phthalate (DnOP). The restrictions mainly concern food contacts materials (FCM), cosmetics, toys, and childcare articles [2][16][17][18][2,16,17,18].
2. Physical, Chemical, and Environmental Properties of Phthalates
Phthalates are formed by a reaction of phthalic anhydride with various alcohols. The number of carbon atoms present will determine the length of the lateral chains R and R’, and thus, the molecular weight of the phthalate is obtained [1]. PAEs differ chemically in the substitutions of the R1 and R2 side chains (which characterise their physicochemical properties) and are slightly volatile liquids, generally colourless, odourless, and oily liquids at room temperature [6]. In addition, their solubility in fat (lipophilic property) increases with the lengthening of the side chains R and R’ (Figure 1).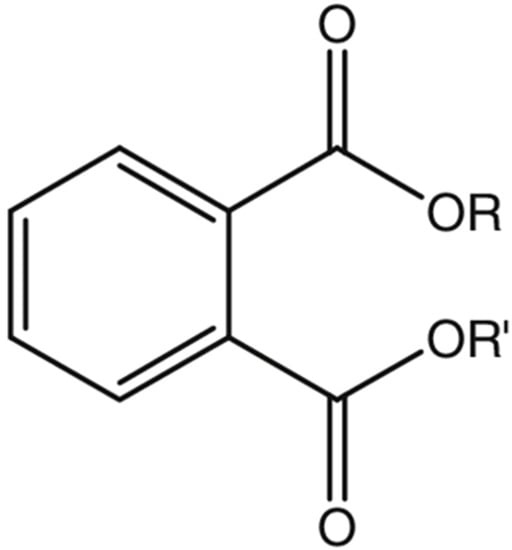
Figure 1.
General chemical structure of phthalate esters.
Table 1.
Most common phthalates with acronyms, molecular formulas, CAS, R1, and R2 chains and their log K
ow.
.
| PAE Congeners | Acronym | Molecular Formula | CAS | R1 | R2 | Log Kow |
|---|---|---|---|---|---|---|
| dimethyl phthalate | DMP | C10H10O4 | 131-11-3 | CH3 | CH3 | 1.60 |
| diethyl phthalate | DEP | C12H14O4 | 84-66-2 | CH2CH3 | CH2CH3 | 2.47 |
| diisobutyl phthalate | DiBP | C16H22O4 | 84-69-5 | CH2CH(CH3)2 | CH2CH(CH3)2 | 4.11 |
| dibutyl phthalate | DnBP | C16H22O4 | 84-74-2 | CH2CH2CH2CH3 | CH2CH2CH2CH3 | 4.50 |
| dimethylglycol phthalate | DMEP | C14H18O6 | 117-82-8 | CH2CH2OCH3 | CH2CH2OCH3 | 1.11 * |
| benzyl butyl phthalate | BBzP | C19H20O4 | 85-68-7 | CH2C6H5 | CH2C6H5 | 4.73 |
| dicyclohexyl phthalate | DCHP | C20H26O4 | 84-61-7 | CH(CH2)5 | CH(CH2)5 | 5.6 |
| di-n-pentyl phthalate | DnPP | C18H26O4 | 131-18-0 | CH2(CH2)3CH3 | CH2(CH2)3CH3 | 5.62 |
| bis (2-n-butoxyethyl) phthalate | DBEP | C20H30O6 | 117-83-9 | CH2CH2O(CH2)3CH3 | CH2CH2O(CH2)3CH3 | 4.06 * |
| diphenyl phthalate | DPhP | C24H38O4 | 84-62-8 | C6H5 | C6H5 | n.a. |
| di(2-ethylhexyl) phthalate | DEHP | C20H14O4 | 117-81-7 | CH(CH2)5(CH3)2 | CH(CH2)5(CH3)2 | 7.60 |
| di-n-octyl phthalate | DnOP | C24H38O4 | 117-84-0 | (CH2)7CH3 | (CH2)7CH3 | 8.10 |
| diisononyl phthalate | DiNP | C26H42O4 | 28553-12-0 | C9H19 | C9H19 | 8.8 |
| dinonyl phthalate | DnNP | C26H42O4 | 84-76-4 | C9H19 | C9H19 | 9.52 * |
Log Kow values were obtained from PubChem [33]; when the calculated value was not present, the estimated value was added *; n.a.: not available.
The danger of phthalates derives from their ability to interact with cell membranes, which is justified by their affinity towards organic portions. This property can be represented by the partition coefficient octanol/water, log Kow, i.e., the concentration ratio of a solute between octanol and water. Kow provides an estimate of the hydrophobicity of a given molecule and can predict the tendency of the breakdown of a chemical in water, lipids, sediments, and soil organic matter.
