
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Michael Wassenegger | + 1838 word(s) | 1838 | 2021-05-20 04:51:51 | | | |
| 2 | Nora Tang | Meta information modification | 1838 | 2021-05-20 09:41:32 | | | | |
| 3 | Dalakouras Athanasios | -52 word(s) | 1786 | 2021-05-20 13:26:53 | | |
Video Upload Options
Viroids are plant pathogenic, circular, non-coding, single-stranded RNAs (ssRNAs). Members of the Pospiviroidae family replicate in the nucleus of plant cells through double-stranded RNA (dsRNA) intermediates, thus triggering the host’s RNA interference (RNAi) machinery. In plants, the two RNAi pillars are Post-Transcriptional Gene Silencing (PTGS) and RNA-directed DNA Methylation (RdDM), and the latter has the potential to trigger Transcriptional Gene Silencing (TGS). Over the last three decades, the employment of viroid-based systems has immensely contributed to our understanding of both of these RNAi facets.
1. Viroids and RNAi
Viroids, the smallest known infectious agents, were discovered in 1971 by Theodor Diener [1]. Fifty years of ongoing research into these minimalistic pathogens has shed significant light on their biology. It is now well established that viroids are non-encapsidated, non-coding, circular ssRNAs, with a size of approximately 250–400 nucleotides (nt) and are classified into two major families, the Pospiviroidae and the Avsunviroidae, whose members replicate in the nucleus and the chloroplast, respectively [2][3][4][5]. The Pospiviroidae species, a potato spindle tuber viroid (PSTVd), has a rod-like structured 359-nucleotide genome that can be functionally and structurally divided into the following five domains: terminal left (TL), pathogenicity (P), central (C), variable (V) and terminal right (TR) [6][7]. When a PSTVd enters a plant cell, it is directed into the nucleus, most likely through the interaction of its TR domain with the host’s bromodomain-containing viroid RNA-binding protein 1 (VIRP1) [8][9]. In the nucleus, a PSTVd replicates via an asymmetric rolling circle mechanism [10]. A circular monomeric viroid (defined as having a plus polarity) is transcribed by a DNA-dependent RNA polymerase II (POL II) into linear oligomeric ssRNAs of a minus polarity [11]. These serve as a template for POL II, resulting in the accumulation of linear oligomeric ssRNAs of a plus polarity. The latter is cleaved by a type III RNase into fragments that are finally ligated by DNA ligase 1 into unit length circular RNAs [12] (Figure 1). A mature viroid then exits the nucleus into the cytoplasm, moves to neighboring cells through plasmodesmata and then to distant parts of the plant through the phloem [13][14]. The most important features of viroids that make them a very specific and unique pathogen are the fact that they do not encode for proteins and that their infection cycle does not involve DNA intermediates, although their RNAs are produced by POL II, an enzyme that generally transcribes DNA.
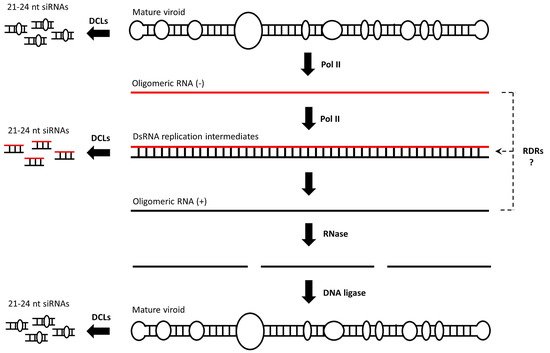
Figure 1. Pospiviroidae replicates through an asymmetric rolling circle mechanism. POL II is involved in all of the steps of viroid replication, generating dsRNA replication intermediates, but whether additional polymerases such as RDRs are also involved is not clear. DCLs may process both dsRNAs and the rod-shaped ssRNA mature molecule into siRNAs.
It has been suggested that dsRNAs are generated during viroid replication [15][16][17]. The source of these dsRNAs is not clear, but they are most probably generated upon POL II transcription of an oligomeric plus from an oligomeric minus transcript and their kinetic interaction. Whether the RNA-directed RNA polymerase (RDR) transcription of these oligomeric transcripts or even of the mature viroid contributes to dsRNA generation is not clear but cannot be excluded. In any case, viroid replication triggers the host’s RNAi mechanism [18][19][20]. At least in Nicotiana benthamiana, all four dicer-like endonucleases (DCLs) seem to process a PSTVd into a small interfering RNA (siRNA) [21][22]. It is most likely that dsRNA replication intermediates are preferentially processed by DCL2/3/4 into 21–24-nucleotide siRNAs, but it cannot be ruled out that a microRNA (miRNA) precursor-like, rod-shaped, mature viroid is also processed by a DCL1 into 21-nucleotide siRNAs, although in the latter case the occurring siRNAs would contain bulges (Figure 1). Whatever their source may be, DCL4-produced 21-nucleotide siRNAs are generally loaded onto Argonaute 1 (AGO1), leading to the formation of an RNA-induced silencing complex (RISC) [23]. An RISC recognizes the complementary transcripts for cleavage in a process termed PTGS [24]. DCL2-produced 22-nucleotide siRNAs are also loaded onto AGO1 and are suggested to change the conformation of AGO1. This alteration is assumed to affect the target RNA degradation. Thus, instead of RNA cleavage, 22-nucleotide siRNA-containing AGO1 complexes seem to either recruit an RDR6 to initiate transitivity and secondary siRNA formation or to inhibit the translation of the targeted mRNA [25][26]. Finally, DCL3-produced 24-nucleotide siRNAs are predominantly loaded onto AGO4 and are involved in the RdDM of cognate sequences [27]. Viroid siRNAs are 5′-phosphorylated and 3′-methylated, as all plant siRNAs are [28]. Small RNA deep sequencing revealed that viroid siRNAs are of both polarities and map preferentially, in the same way virus- and transgene-derived siRNAs do, to specific hotspot regions [29]. At least the 21-nucleotide viroid siRNAs are biologically active, since they trigger the degradation of mRNA’s containing regions that are complementary to viroid siRNAs [30][31]. Interestingly, the mature circular viroid is considered to be resistant to RISC-mediated degradation, most likely due to its extensive secondary structure [29]. Nevertheless, tomato plants expressing a hairpin PSTVd RNA transgene construct were found to be resistant to viroid infection, indicating that the RNAi machinery could target PSTVd-specific RNA molecules that are essential for viroid infection [32].
2. RNA-Directed DNA Methylation
In eukaryotes, DNA methylation refers to the addition of a methyl group to the fifth atom in the six-atom ring of cytosine residues (Cs). As a chemical modification, DNA methylation was discovered in 1948 by Rollin Hotchkiss, almost simultaneous to the identification of DNA as genetic material by Avery, MacLeold and McCarty. Almost thirty years later, in 1975, Holliday and Pugh proposed that DNA methylation is an important epigenetic mark [33]. However, the mechanism by which DNA methylation was induced remained elusive for many years. In plants, DNA methylation was widely considered to be mediated by DNA–DNA interactions. However, in 1994, a study using viroid-infected tobacco plants showed that de novo DNA methylation is mediated by RNA molecules, thus aptly termed ‘RNA-directed DNA methylation’ (RdDM) [34]. Today, almost 30 years after the RdDM discovery, the widely accepted model suggests that 24-nucleotide siRNAs (canonical RdDM) or, in exceptional cases, 21/22-nucleotide siRNAs (non-canonical RdDM), are loaded onto AGO4 and guide domains-rearranged methyltransferases (DRMs) to methylate cognate DNA, most likely through a process wherein siRNAs directly interact with DNA or interact with the nascent transcripts produced by RNA polymerase V (POL V) [35][36][37]. However, a growing body of evidence challenges the validity of this model. To begin with, it has been suggested that POL V is recruited to an already methylated DNA template, thus it can hardly be involved in the very first step of de novo methylation of a completely unmethylated DNA [38]. More importantly, RdDM is not eliminated in an Arabidopsis thaliana quadruple dcl1 dcl2 dcl3 dcl4 mutant, suggesting that DCL-produced siRNAs are dispensable for RdDM [39]. In addition, AGO4, which is thought to be involved in both canonical and non-canonical RdDM, is not always required for RdDM. AGO4 contains the DDH motif and has a slicer activity [40]. During sense RdDM (S-RdDM), a single-stranded RNA is cleaved by the siRNAs loaded onto AGO4 and the cleaved transcripts are copied by RDRs into dsRNAs [40]. However, during inverted repeat RdDM (IR-RdDM), the generation of a dsRNA would not rely on AGO4 activity, but on the mere transcription of hairpin RNA-producing DNA. Indeed, AGO4 is not required in IR-RdDM, at least not in Arabidopsis thaliana [41]. Collectively, these data suggest that RdDM is triggered not by siRNAs, but by long dsRNAs (>50 bp) [42][43][44][45]. According to our model, long dsRNAs appear to define the DNA region that will be methylated in a ruler-like fashion (Step 1, Figure 2). The dsRNA–DNA interaction may involve the formation of triple helices, as has been shown to take place in mammals between long non-coding RNAs (lncRNAs) and DNA [46]. Such aberrant structures could attract DRMs to impose a first (perhaps incomplete) wave of de novo methylation on fully unmethylated DNA (Step 2, Figure 2). The resulting (and incompletely) methylated DNA seems to recruit POL IV and POL V. POL IV generates short transcripts (40–50 nt) that are copied by RDR2 into 40–50 bp dsRNAs that are cleaved by DCL3 into 24-nucleotide siRNAs [47]. The AGO4-loaded 24-nucleotide siRNAs will now hybridize the newly produced POL V transcript, and this re-initiates the recruitment of DRMs to impose a second wave of methylation marks (Step 3, Figure 2) [48][49]. As siRNAs are amplified by this self-reinforcing loop mechanism, methylation will sequentially increase until nearly all of the Cs are methylated to CG, CHG and CHH sequence contexts in both of the DNA strands [50]. Importantly, upon cell division and in the absence of dsRNAs/siRNAs, CG methylation can be maintained through the action of methyltransferase 1 (MET1) (Step 4, Figure 2) [51]. To a lesser extent, CHG methylation will be maintained by chromomethylase 3 (CMT3) [52]. However, CHH methylation cannot be maintained, as it always needs to be de novo established and requires the presence of dsRNAs/siRNAs [45].
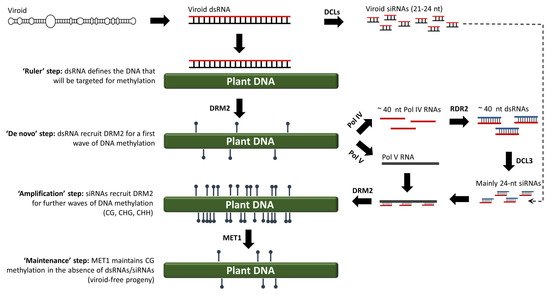
Figure 2. An alternative RdDM model for both viroid and non-viroid systems, wherein the very first de novo methylation step is triggered by dsRNAs rather than siRNAs.
3. Discovery of RdDM in Viroid-Infected Plants
RdDM was discovered in a study involving viroid-infected tobacco plants [34]. In that study, transgenic tobacco plants carried an expression cassette containing three head-to-tail linked, plus-oriented, full length PSTVd359 cDNA copies (Nt-tri-PSTVd359). The transcription of this transgene construct led to the formation of mature, circular and infectious PSTVd molecules. However, at that time it was not known that replicating PSTVd and the concomitant accumulation of viroid dsRNAs/siRNAs could lead to the DNA methylation of the cognate sequences. Thus, it was puzzling that the PSTVd359 cDNA proved severely resistant to methylation-sensitive restriction endonucleases when genotyping the transgenic lines using Southern blot analysis. In order to exclude the possibility that methylation was induced by the DNA–DNA interactions, transgenic tobacco plants were generated expressing a head-to-tail linked dimeric PSTVd333 cDNA (Nt-di-PSTVd333), where each PSTVd333 fragment carried a 26-bp deletion compared to the full length PSTVd359 (Figure 3A). Upon transcription of the dimeric PSTVd333, a 666-nucleotide RNA was produced but, due to the 26-nucleotide deletion, it could not be processed into a mature PSTVd. Thus, these transgenic lines were not PSTVd-infected. Moreover, no methylation of the PSTVd333 was detected, suggesting that the agent of DNA methylation is something other than the mere DNA–DNA interactions that were suggested to occur upon the presence of tandem cDNA repeats (Figure 3B). Indeed, when the Nt-di-PSTVd333 plants were mechanically-infected with PSTVd RNA, the PSTVd333 cDNA exhibited dense methylation, demonstrating that an RNA agent directed the DNA methylation in the cognate DNA sequences (Figure 3C) [34].
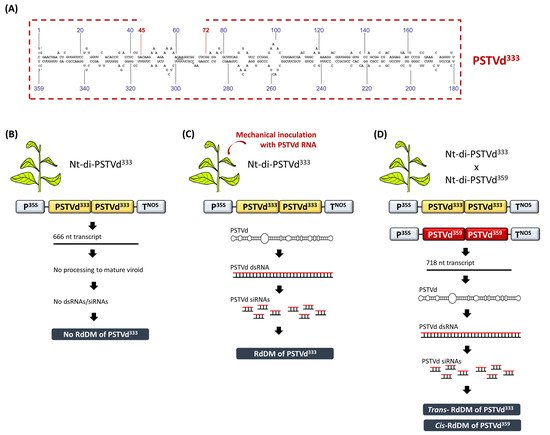
Figure 3. The discovery of RdDM in viroid-infected tobacco plants. (A) Schematic representation of the PSTVd333 containing a 26-nucleotide deletion compared to a full length PSTVd359. (B) Nt-di-PSTVd333 plants are free of methylation. (C) Upon viroid infection, Nt-di-PSTVd333 plants exhibit dense RdDM. (D) The crossing of Nt-di-PSTVd333 with Nt-di-PSTVd359 induced both cis and trans RdDM in CG, CHG and CHH contexts. Upon the segregation of the PSTVd359 infectious transgene, the PSTVd333 progeny maintains CG methylation for at least two generations.
References
- Diener, T.O. Potato spindle tuber “virus”: IV. A replicating, low molecular weight RNA. Virology 1971, 45, 411–428.
- Flores, R.; Gago-Zachert, S.; Serra, P.; Sanjuan, R.; Elena, S.F. Viroids: Survivors from the RNA world? Annu. Rev. Microbiol. 2014, 68, 395–414.
- Tabler, M.; Tsagris, M. Viroids: Petite RNA pathogens with distinguished talents. Trends Plant Sci. 2004, 9, 339–348.
- Wang, Y. Current view and perspectives in viroid replication. Curr. Opin. Virol. 2021, 47, 32–37.
- Flores, R.; Hernandez, C.; de Alba, A.E.M.; Daros, J.A.; di Serio, F. Viroids and viroid-host interactions. Annu. Rev. Phytopathol. 2005, 43, 117–139.
- Owens, R.A.; Hammond, R.W. Viroid pathogenicity: One process, many faces. Viruses 2009, 1, 298–316.
- Tsagris, E.M.; de Alba, A.E.M.; Gozmanova, M.; Kalantidis, K. Viroids. Cell. Microbiol. 2008, 10, 2168–2179.
- Gozmanova, M.; Denti, M.A.; Minkov, I.N.; Tsagris, M.; Tabler, M. Characterization of the RNA motif responsible for the specific interaction of potato spindle tuber viroid RNA (PSTVd) and the tomato protein Virp1. Nucleic Acids Res. 2003, 31, 5534–5543.
- Kalantidis, K.; Denti, M.A.; Tzortzakaki, S.; Marinou, E.; Tabler, M.; Tsagris, M. Virp1 is a host protein with a major role in potato spindle tuber viroid infection in Nicotiana plants. J. Virol. 2007, 81, 12872–12880.
- Branch, A.D.; Robertson, H.D. A replication cycle for viroids and other small infectious RNA’s. Science 1984, 223, 450–455.
- Mudiyanselage, S.D.D.; Wang, Y. Evidence supporting that RNA polymerase II catalyzes de novo transcription using potato spindle tuber viroid circular RNA templates. Viruses 2020, 12, 371.
- Nohales, M.A.; Flores, R.; Daros, J.A. Viroid RNA redirects host DNA ligase 1 to act as an RNA ligase. Proc. Natl. Acad. Sci. USA 2012, 109, 13805–13810.
- Ding, B.; Kwon, M.O.; Hammond, R.; Owens, R. Cell-to-cell movement of potato spindle tuber viroid. Plant J. 1997, 12, 931–936.
- Zhu, Y.; Qi, Y.; Xun, Y.; Owens, R.; Ding, B. Movement of potato spindle tuber viroid reveals regulatory points of phloem-mediated RNA traffic. Plant Physiol. 2002, 130, 138–146.
- Owens, R.A.; Diener, T.O. RNA intermediates in potato spindle tuber viroid replication. Proc. Natl. Acad. Sci. USA 1982, 79, 113–117.
- Grill, L.K.; Semancik, J.S. RNA sequences complementary to citrus exocortis viroid in nucleic acid preparations from infected Gynura aurantiaca. Proc. Natl. Acad. Sci. USA 1978, 75, 896–900.
- Hadidi, A.; Hashimoto, J.; Diener, T.O. Potato spindle tuber viroid-specific double-stranded RNA in extracts from infected tomato leaves. Annales de l’Institut Pasteur Virologie 1982, 133, 15–31.
- Dalakouras, A.; Dadami, E.; Wassenegger, M. Engineering viroid resistance. Viruses 2015, 7, 634–646.
- Itaya, A.; Folimonov, A.; Matsuda, Y.; Nelson, R.S.; Ding, B. Potato spindle tuber viroid as inducer of RNA silencing in infected tomato. Mol. Plant Microbe Interact. 2001, 14, 1332–1334.
- Wang, M.B.; Bian, X.Y.; Wu, L.M.; Liu, L.X.; Smith, N.A.; Isenegger, D.; Wu, R.M.; Masuta, C.; Vance, V.B.; Watson, J.M.; et al. On the role of RNA silencing in the pathogenicity and evolution of viroids and viral satellites. Proc. Natl. Acad. Sci. USA 2004, 101, 3275–3280.
- Dadami, E.; Boutla, A.; Vrettos, N.; Tzortzakaki, S.; Karakasilioti, I.; Kalantidis, K. Dicer-like 4 but not dicer-like 2 may have a positive effect on potato spindle tuber viroid accumulation in Nicotiana benthamiana. Mol. Plant 2013, 6, 232–234.
- Katsarou, K.; Mavrothalassiti, E.; Dermauw, W.; van Leeuwen, T.; Kalantidis, K. Combined activity of DCL2 and DCL3 is crucial in the defense against potato spindle tuber viroid. PLoS Pathog. 2016, 12, e1005936.
- Minoia, S.; Carbonell, A.; di Serio, F.; Gisel, A.; Carrington, J.C.; Navarro, B.; Flores, R. Specific argonautes selectively bind small RNAs derived from potato spindle tuber viroid and attenuate viroid accumulation in vivo. J. Virol. 2014, 88, 11933–11945.
- Hamilton, A.J.; Baulcombe, D.C. A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 1999, 286, 950–952.
- Chen, H.M.; Chen, L.T.; Patel, K.; Li, Y.H.; Baulcombe, D.C.; Wu, S.H. 22-nucleotide RNAs trigger secondary siRNA biogenesis in plants. Proc. Natl. Acad. Sci. USA 2010, 107, 15269–15274.
- Wu, H.; Li, B.; Iwakawa, H.O.; Pan, Y.; Tang, X.; Ling-Hu, Q.; Liu, Y.; Sheng, S.; Feng, L.; Zhang, H.; et al. Plant 22-nt siRNAs mediate translational repression and stress adaptation. Nature 2020, 581, 89–93.
- Chan, S.W.; Zilberman, D.; Xie, Z.; Johansen, L.K.; Carrington, J.C.; Jacobsen, S.E. RNA silencing genes control de novo DNA methylation. Science 2004, 303, 1336.
- Martin, R.; Arenas, C.; Daros, J.A.; Covarrubias, A.; Reyes, J.L.; Chua, N.H. Characterization of small RNAs derived from citrus exocortis viroid (CEVd) in infected tomato plants. Virology 2007, 367, 135–146.
- Itaya, A.; Zhong, X.; Bundschuh, R.; Qi, Y.; Wang, Y.; Takeda, R.; Harris, A.R.; Molina, C.; Nelson, R.S.; Ding, B. A structured viroid RNA serves as a substrate for dicer-like cleavage to produce biologically active small RNAs but is resistant to RNA-induced silencing complex-mediated degradation. J. Virol. 2007, 81, 2980–2994.
- Adkar-Purushothama, C.R.; Brosseau, C.; Giguere, T.; Sano, T.; Moffett, P.; Perreault, J.P. Small RNA derived from the virulence modulating region of the potato spindle tuber viroid silences callose synthase genes of tomato plants. Plant Cell 2015, 27, 2178–2194.
- Vogt, U.; Pelissier, T.; Putz, A.; Razvi, F.; Fischer, R.; Wassenegger, M. Viroid-induced RNA silencing of GFP-viroid fusion transgenes does not induce extensive spreading of methylation or transitive silencing. Plant J. 2004, 38, 107–118.
- Schwind, N.; Zwiebel, M.; Itaya, A.; Ding, B.; Wang, M.B.; Krczal, G.; Wassenegger, M. RNAi-mediated resistance to potato spindle tuber viroid in transgenic tomato expressing a viroid hairpin RNA construct. Mol. plant Pathol. 2009, 10, 459–469.
- Holliday, R.; Pugh, J.E. DNA modification mechanisms and gene activity during development. Science 1975, 187, 226–232.
- Wassenegger, M.; Heimes, S.; Riedel, L.; Sanger, H.L. RNA-directed de novo methylation of genomic sequences in plants. Cell 1994, 76, 567–576.
- Gallego-Bartolome, J. DNA methylation in plants: Mechanisms and tools for targeted manipulation. New Phytol. 2020, 227, 38–44.
- Matzke, M.A.; Kanno, T.; Matzke, A.J. RNA-directed DNA Methylation: The evolution of a complex epigenetic pathway in flowering plants. Annu. Rev. Plant Biol. 2015, 66, 243–267.
- Matzke, M.A.; Mosher, R.A. RNA-directed DNA methylation: An epigenetic pathway of increasing complexity. Nat. Rev. Genet. 2014, 15, 394–408.
- Liu, Z.W.; Shao, C.R.; Zhang, C.J.; Zhou, J.X.; Zhang, S.W.; Li, L.; Chen, S.; Huang, H.W.; Cai, T.; He, X.J. The SET domain proteins SUVH2 and SUVH9 are required for pol V occupancy at RNA-directed DNA methylation loci. PLoS Genet. 2014, 10, e1003948.
- Yang, D.L.; Zhang, G.; Tang, K.; Li, J.; Yang, L.; Huang, H.; Zhang, H.; Zhu, J.K. Dicer-independent RNA-directed DNA methylation in arabidopsis. Cell Res. 2016, 26, 1264.
- Qi, Y.; He, X.; Wang, X.J.; Kohany, O.; Jurka, J.; Hannon, G.J. Distinct catalytic and non-catalytic roles of ARGONAUTE4 in RNA-directed DNA methylation. Nature 2006, 443, 1008–1012.
- Zilberman, D.; Cao, X.; Johansen, L.K.; Xie, Z.; Carrington, J.C.; Jacobsen, S.E. Role of arabidopsis ARGONAUTE4 in RNA-directed DNA methylation triggered by inverted repeats. Curr. Biol. 2004, 14, 1214–1220.
- Dalakouras, A.; Dadami, E.; Wassenegger, M. Viroid-induced DNA methylation in plants. Biomol. Concepts 2013, 4, 557–565.
- Dalakouras, A.; Papadopoulou, K.K. Epigenetic modifications: An unexplored facet of exogenous RNA application in plants. Plants 2020, 9, 673.
- Dalakouras, A.; Wassenegger, M. Revisiting RNA-directed DNA methylation. RNA Biol. 2013, 10, 453–455.
- Dalakouras, A.; Ganopoulos, I. Induction of promoter DNA methylation upon high-pressure spraying of double-stranded RNA in plants. Agronomy 2021, 11, 789.
- Li, Y.; Syed, J.; Sugiyama, H. RNA-DNA triplex formation by long noncoding RNAs. Cell Chem. Biol. 2016, 23, 1325–1333.
- Zhai, J.; Bischof, S.; Wang, H.; Feng, S.; Lee, T.F.; Teng, C.; Chen, X.; Park, S.Y.; Liu, L.; Gallego-Bartolome, J.; et al. A one precursor one siRNA model for Pol IV-pependent siRNA biogenesis. Cell 2015, 163, 445–455.
- Wierzbicki, A.T.; Haag, J.R.; Pikaard, C.S. Noncoding transcription by RNA polymerase Pol IVb/Pol V mediates transcriptional silencing of overlapping and adjacent genes. Cell 2008, 135, 635–648.
- Wierzbicki, A.T.; Ream, T.S.; Haag, J.R.; Pikaard, C.S. RNA polymerase V transcription guides ARGONAUTE4 to chromatin. Nat. Genet. 2009, 41, 630–634.
- Pelissier, T.; Thalmeir, S.; Kempe, D.; Sanger, H.L.; Wassenegger, M. Heavy de novo methylation at symmetrical and non-symmetrical sites is a hallmark of RNA-directed DNA methylation. Nucleic Acids Res. 1999, 27, 1625–1634.
- Aufsatz, W.; Mette, M.F.; Matzke, A.J.; Matzke, M. The role of MET1 in RNA-directed de novo and maintenance methylation of CG dinucleotides. Plant Mol. Biol. 2004, 54, 793–804.
- Lindroth, A.M.; Cao, X.; Jackson, J.P.; Zilberman, D.; McCallum, C.M.; Henikoff, S.; Jacobsen, S.E. Requirement of CHROMOMETHYLASE3 for maintenance of CpXpG methylation. Science 2001, 292, 2077–2080.





