
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maryse Vanderplanck | + 934 word(s) | 934 | 2020-05-26 08:55:12 | | | |
| 2 | Nora Tang | Meta information modification | 934 | 2020-06-03 05:56:35 | | | | |
| 3 | Nora Tang | -1 word(s) | 933 | 2020-11-02 02:35:57 | | |
Video Upload Options
Excessive pollen harvesting by bees can compromise the reproductive success of plants. Plants have therefore evolved different morphological structures and floral cues to narrow the spectrum of pollen feeding visitors. Among “filtering” mechanisms, the chemical and mechanical protection of pollen might shape bee-flower interactions and restrict pollen exploitation to a specific suite of visitors such as observed in Asteraceae. Asteraceae pollen is indeed only occasionally exploited by generalist bee species but plentifully foraged by specialist ones (i.e., Asteraceae paradox). During our bioassays, we observed that micro-colonies of generalist bumblebee (Bombus terrestris L.) feeding on Taraxacum pollen (Asteraceae) reduced their pollen collection and offspring production. Bees also experienced physiological effects of possible defenses in the form of digestive damage. Overall, our results suggest the existence of an effective chemical defense in Asteraceae pollen, while the hypothesis of a mechanical defense appeared more unlikely. Pre- and post-ingestive effects of such chemical defenses (i.e., nutrient deficit or presence of toxic compounds), as well as their role in the shaping of bee-flower interactions, are discussed. Our results strongly suggest that pollen chemical traits may act as drivers of plant selection by bees and partly explain why Asteraceae pollen is rare in generalist bee diets.
1. Introduction
Through pollen collection, bees act concurrently as effective pollinators and herbivores, since both larvae and adults feed exclusively on pollen and nectar [1][2]. Like other herbivorous insects, bees display a high diversity of interactions with their host plants, from strict specialization (i.e., oligolecty, where bees collect pollen from flowers of a single genus, subfamily, or family) to broad generalization (i.e., polylecty, where bees exploit flowers of more than one plant family) [3][4][5]. Regardless of floral specialization, bees display numerous adaptations to discriminate among plant species and to enhance floral resource foraging [1][6][7][8], which can compromise the reproductive success of plants. For instance, bees can remove 95.5% of the pollen produced by flowers of Campanula rapunculus (Campanulaceae) [9] and some solitary species require the entire content of more than 30 flowers, even more than 1000 flowers, to feed a single larva [7]. In response to excessive pollen harvesting, flowering plants have drawn up complex defense mechanisms and adaptations. In fact, flowers have evolved several morphological traits that can be viewed as adaptations preventing excessive pollen harvesting by bees, such as heteranthery, nototribic flowers (i.e., dorsal anthers hidden in the upper lip of the flower, such as in Lamiaceae), keel flowers (i.e., ventral anthers hidden in a boat-shaped keel formed by the fusion of the two lower petals of the flower, such as in Fabaceae), floral tubes, poricidal anthers and progressive pollen release (reviewed in [8]).
2. Asteraceae Paradox
Selection may also act on pollen traits to narrow the spectrum of pollen feeding visitors. For instance, although Asteraceae are ubiquitous in most temperate habitats [10], they are only occasionally exploited by polylectic species such as Bombus [11][12] and Colletes [4] (i.e., Asteraceae paradox [4]). This Asteraceae avoidance cannot be explained by complex floral morphology, since compound inflorescences ensure an easy access to both pollen and nectar over an extended time period [4]. The failure of several unspecialized bee species to develop on Asteraceae pollen rather suggests that it may possess unfavorable or protective properties so that bees might require physiological adaptations to use it [13][14][15][16][17][18][19]. Although Asteraceae pollens are known to have low protein content, this is seemingly not the only reason for the inadequacy of their pollen [20]. The pollen may actually lack other essential nutrients, contain toxins, or display a low digestibility [4][13][17][20][21][22]. Such pollen protections probably shape bee–flower interactions to lead to a narrowing of the spectrum of pollen-feeding visitors in Asteraceae.
It is largely assumed that the synthesis of secondary metabolites constitutes a complex system of chemical defenses in plants against herbivorous insect attacks [23][24]. Although these chemical compounds are mainly studied in vegetative parts, some obviously occur in nectar and pollen of flowering plants, with bee pollinators then exposed to their biological activities [24][25][26][27][28][29][30][31][32]. For instance, sesquiterpene lactones are characteristic secondary metabolites in Asteraceae [33], with high chemotaxonomic specificity [34]. Although they probably have evolved as defense through their deterrence to herbivores [35], they also occur in pollen [36] and may display insecticidal activities [37]. Among chemical defenses, constraints could also act through nutrient availability. Although proteins are often regarded as a reference in terms of nutritional quality, lipids are also important [38][39][40][41], including sterols, essential compounds in bee physiology (e.g., pupation, ovary development) that are exclusively exogenous [42][43]. The fact that ∂7-sterols often occur in Asteraceae pollen in higher proportions than more common and usable sterols (i.e., 24-methylenecholesterol, ß-sitosterol, and ∂5-avenasterol) may indicate a defense mechanism against excessive pollen harvesting [22]. In addition to these variations of pollen primary and secondary metabolites, pollen also varies in its wall resistance properties, which may result in incomplete digestion. Transmission electron microscopy has revealed that Asteraceae pollen possesses a thick multilayer wall [44], which might inhibit the extraction of nutrients and act as a mechanical defense [17][21]. Despite these hypotheses, it is unclear why Asteraceae pollen is unsuitable for most bee species and the Asteraceae paradox remains unsolved. In the present study, bumblebee micro-colonies (Bombus terrestris L.) were forced to feed on different diets (Figure. 1) to investigate the unfavorable properties of Taraxacum pollen. From the observed patterns in foraging behavior, larval development, and digestive damages, we infer possible mechanical or chemical protective properties of Asteraceae pollen.
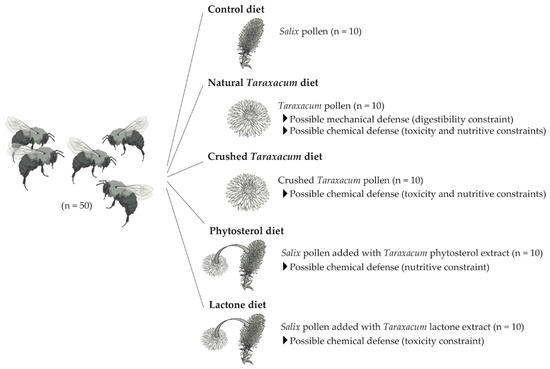
Figure 1. Experimental design and summary of diet treatments provided to B. terrestris. Each micro-colony consisted of 5 workers fed for 35 days. Mortality, offspring production, and resource collection (i.e., pollen and syrup) were monitored during or at the end of the bioassays.
References
- Westrich, P. Die Wildbienen Baden-Württembergs; Eugen Ulmer: Suttgart, Germany, 1989.
- Michener, C.D. The Bees of the World, 2nd ed.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007.
- Cane, J.H.; Sipes, S.D. Characterizing floral specialization by bees: Analytical methods and revised lexicon for oligolecty. In Plant–Pollinator Interactions from Specialization to Generalization; Waser, N.M., Ollerton, J., Eds.; The University of Chicago Press: Chicago, IL, USA, 2006; pp. 99–122.
- Müller, A.; Kuhlmann, M. Pollen hosts of western palaearctic bees of the genus Colletes (Hymenoptera: Colletidae): The Asteraceae paradox. Biol. J. Linn. Soc. 2008, 95, 719–733.
- Stefan Dötterl; Nicolas J. Vereecken; The chemical ecology and evolution of bee–flower interactions: a review and perspectivesThe present review is one in the special series of reviews on animal–plant interactions.. Canadian Journal of Zoology 2010, 88, 668-697, 10.1139/z10-031.
- Müller, A. Convergent evolution of morphogical specializations in Central European bee and honey wasp species as an adaptation to the uptake of pollen from nototribic flowers (Hymenoptera, Apoidea and Masaridae). Biol. J. Linn. Soc. 1996, 57, 235–252.
- Andreas Müller; Stefan Diener; Simone Schnyder; Katharina Stutz; Claudio Sedivy; S. Dorn; Quantitative pollen requirements of solitary bees: Implications for bee conservation and the evolution of bee–flower relationships. Biological Conservation 2006, 130, 604-615, 10.1016/j.biocon.2006.01.023.
- Westerkamp, C.; Claben-Bockhoff, R. Bilabiate Flowers: The Ultimate Response to Bees? Ann. Bot. 2007, 100, 361–374.
- Clemens Schlindwein; D. Wittmann; Celso Feitosa Martins; A. Hamm; J. Alves Siqueira; D. Schiffler; I. C. Machado; Pollination of Campanula rapunculus L. (Campanulaceae): How much pollen flows into pollination and into reproduction of oligolectic pollinators?. Österreichische Botanische Zeitschrift 2005, 250, 147-156, 10.1007/s00606-004-0246-8.
- Funk, V.A.; Bayer, R.J.; Keeley, S.; Chan, R.; Watson, L.; Gemeinholzer, B.; Schilling, E.; Panrelo, J.L.; Baldwin, B.G.; Garcia-Jacas, N.; et al. Everywhere but Antartica: Using a supertree to understand the diversity and distribution of the Compositae. Biol. Skr. 2005, 55, 343–373.
- Dave Goulson; M.E. Hanley; B. Darvill; Jonathan S. Ellis; M.E. Knight; Causes of rarity in bumblebees. Biological Conservation 2005, 122, 1-8, 10.1016/j.biocon.2004.06.017.
- David Kleijn; Ivo Raemakers; A RETROSPECTIVE ANALYSIS OF POLLEN HOST PLANT USE BY STABLE AND DECLINING BUMBLE BEE SPECIES. Ecology 2008, 89, 1811-1823, 10.1890/07-1275.1.
- M. D. Levin; M. H. Haydak; Comparative Value of Different Pollens in the Nutrition of Osmia Lignaria. Bee World 1957, 38, 221-226, 10.1080/0005772x.1957.11095007.
- Guirguis, G.N.; Brindley, W.A. Insecticide susceptibility and response to selected pollens of larval alfalfa leafcutting bees, Megachile pacifica (Panzer) (Hymenoptera: Megachilidae). Environ. Entomol. 1974, 3, 691–694.
- A. Genissel; P. Aupinel; C. Bressac; Jean-Noël Tasei; C. Chevrier; Influence of pollen origin on performance of Bombus terrestris micro-colonies. Entomologia Experimentalis et Applicata 2002, 104, 329-336, 10.1046/j.1570-7458.2002.01019.x.
- Williams, N.M. Use of novel pollen species by specialist and generalist solitary bees (Hymenoptera: Megachilidae). Oecologia 2003, 134, 228–237.
- Praz, C.J.; Müller, A.; Dorn, S. Specialized bees fail to develop on non-host pollen: Do plants chemically protect their pollen? Ecology 2008, 89, 795–804.
- Claudio Sedivy; Andreas Müller; S. Dorn; Closely related pollen generalist bees differ in their ability to develop on the same pollen diet: evidence for physiological adaptations to digest pollen. Functional Ecology 2011, 25, 718-725, 10.1111/j.1365-2435.2010.01828.x.
- Haider, M.; Dorn, S.; Müller, A. Intra- and inter- populational variation in the ability of a solitary bee species to develop on non-host-pollen: Implications for host range expansion. Funct. Ecol. 2013, 27, 255–263.
- Tasei, J.N.; Aupinel, P. Nutritive value of 15 single pollens and pollen mixes tested on larvae produced by bumble bee workers (Bombus terrestris, Hymenoptera: Apidae). Apidologie 2008, 39, 397–409.
- T’Ai H. Roulston; J. H. Cane; Pollen nutritional content and digestibility for animals. Österreichische Botanische Zeitschrift 2000, 222, 187-209, 10.1007/bf00984102.
- Vanderplanck, M.; Declèves, S.; Roger, N.; Decroo, C.; Caulier, G.; Glauser, G.; Gerbaux, P.; Lognay, G.; Richel, A.; Escaravage, N.; et al. Is non-host pollen suitable for generalist bumblebees? Insect Sci. 2018, 25, 259–272.
- Ian Woodward; Tansley reviews. New Phytologist 2004, 163, 453-453, 10.1111/j.1469-8137.2004.01141.x.
- Roberts, M.F.; Wink, M. Alkaloids—Biochemistry, Ecological Functions and Medical Applications; Plenum Press: New York, NY, USA, 1998.
- David W. Inouye; Nanette D. Favre; Judith A. Lanum; Daniel M. Levine; Jody B. Meyers; Miles S. Roberts; Florence C. Tsao; Ye-Yeng Wang; The Effects of Nonsugar Nectar Constituents on Estimates of Nectar Energy Content. Ecology 1980, 61, 992-996, 10.2307/1936767.
- Andreas Detzel; Michael Wink; Attraction, deterrence or intoxication of bees (Apis mellifera) by plant allelochemicals. Chemoecology 1993, 4, 8-18, 10.1007/bf01245891.
- Lynn S. Adler; The ecological significance of toxic nectar. Oikos 2000, 91, 409-420, 10.1034/j.1600-0706.2000.910301.x.
- Heidi E. M. Dobson; Gunnar Bergström; The ecology and evolution of pollen odors. Pollen and Pollination 2000, 222, 63-87, 10.1007/978-3-7091-6306-1_4.
- I. London-Shafir; S. Shafir; D. Eisikowitch; Amygdalin in almond nectar and pollen – facts and possible roles. Österreichische Botanische Zeitschrift 2003, 238, 87-95, 10.1007/s00606-003-0272-y.
- Kevan, P.G.; Ebert, T. Can almond nectar and pollen poison honey bees? Am. Bee J. 2005, 145, 507–509.
- Kempf, M.; Heil, S.; Haßlauer, I.; Schmidt, L.; von der Ohe, K.; Theuring, C.; Reinhard, A.; Schreier, P.; Beuerle, T.; Opinion of the SKLM on Pyrrolizidin Alkaloids in Honeys, Bee-Keeper Products, and Pollen Products. Lebensmittel und Gesundheit II / Food and Health II 2006, 54, 294-294, 10.1002/352760684x.ch38.
- Martin Heil; Nectar: generation, regulation and ecological functions. Trends in Plant Science 2011, 16, 191-200, 10.1016/j.tplants.2011.01.003.
- Funk, V.A. Sesquiterpene lactones as toxonomis characters in the Asteraceae. Taxon 1983, 32, 1–27.
- Marcus T. Scotti; Vicente Emerenciano; Marcelo J. P. Ferreira; Luciana Scotti; Ricardo Stefani; Marcelo Sobral Da Silva; Francisco Jaime B. Mendonça-Junior; Self-Organizing Maps of Molecular Descriptors for Sesquiterpene Lactones and Their Application to the Chemotaxonomy of the Asteraceae Family. Molecules 2012, 17, 4684-4702, 10.3390/molecules17044684.
- Myung-Ju Kim; Jae-Sug Lee; Seung-Hwa Baek; In vitro Biological Activity of Germacranolide sesquiterpene lactones. Oriental Pharmacy and Experimental Medicine 2009, 9, 192-199, 10.3742/opem.2009.9.2.192.
- Alen Božičević; Maria De Mieri; Christina Nassenstein; Silke Wiegand; Matthias Hamburger; Secondary Metabolites in Allergic Plant Pollen Samples Modulate Afferent Neurons and Murine Tracheal Rings. Journal of Natural Products 2017, 80, 2953-2961, 10.1021/acs.jnatprod.7b00495.
- Guillermo Padilla-Gonzalez; Felipe Antunes Dos Santos; Fernando B. Da Costa; Sesquiterpene Lactones: More Than Protective Plant Compounds With High Toxicity. Critical Reviews in Plant Sciences 2016, 35, 18-37, 10.1080/07352689.2016.1145956.
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Tooker, J.F.; Grozinger, C.M. Macronutrient ratios in polln shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc. Natl. Acad. Sci. USA 2016, 113, E4035–E4042.
- Anthony D. Vaudo; D. Stabler; H. M. Patch; J. F. Tooker; C. M. Grozinger; G. A. Wright; Bumble bees regulate their intake of essential protein and lipid pollen macronutrients. The Journal of Experimental Biology 2016, 219, 3962-3970, 10.1242/jeb.140772.
- Anthony D. Vaudo; John F. Tooker; Harland M. Patch; David J. Biddinger; Michael Coccia; Heather M. Hines; Mark Fiely; Jacob S. Francis; Heather M. Hines; Mackenzie Hodges; Stephanie W. Jackson; Denis Michez; Junpeng Mu; L. Russo; Maliheh Safari; Erin D. Treanore; Maryse Vanderplanck; Eric Yip; Anne S. Leonard; Christina M. Grozinger; Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects 2020, 11, 132, 10.3390/insects11020132.
- Fabian A. Ruedenauer; David Raubenheimer; Daniela Kessner‐Beierlein; Nils Grund‐Mueller; Lisa Noack; Johannes Spaethe; Sara D. Leonhardt; Best be(e) on low fat: linking nutrient perception, regulation and fitness. Ecology Letters 2020, 23, 545-554, 10.1111/ele.13454.
- Spencer T. Behmer; W David Nes; Insect Sterol Nutrition and Physiology: A Global Overview. Advances in Insect Physiology 2003, 31, 1-72, 10.1016/s0065-2806(03)31001-x.
- Cohen, A.C. Insect Diet: Science and Technology; CRC: Boca Raton, FL, USA, 2004.
- N. R. Meier-Melikyan; N. I. Gabaraeva; S. V. Polevova; V. V. Grigor'eva; Ya. V. Kosenko; M. V. Tekleva; Development of Pollen Grain Walls and Accumulation of Sporopollenin. Russian Journal of Plant Physiology 2003, 50, 330-338, 10.1023/a:1023818103172.





